Advanced Manufacturing Process for Pyridylamino Pyrimidine Derivatives and Commercial Scalability
Introduction to Advanced Synthesis of EGFR Inhibitor Intermediates
The pharmaceutical industry continuously seeks robust manufacturing pathways for potent oncology therapeutics, particularly Epidermal Growth Factor Receptor (EGFR) inhibitors. Patent CN110606842B introduces a groundbreaking process for the preparation of N-{2-{[2-(dimethylamino)ethyl](methyl)amino}-6-(2,2,2-trifluoroethoxy)-5-{[4-(1-methyl-1H-indol-3-yl)pyrimidin-2-yl]amino}pyridin-3-yl}acrylamide, designated as Formula I. This compound exhibits remarkable inhibitory activity against drug-resistant EGFR T790M mutations, positioning it as a critical candidate for non-small cell lung cancer treatment. The disclosed methodology addresses longstanding challenges in organic synthesis by providing an environment-friendly, low-cost, and high-yield route that is explicitly suitable for industrial production. By optimizing reaction conditions and reagent selection, this patent offers a viable solution for producing high-purity pharmaceutical intermediates at a commercial scale.
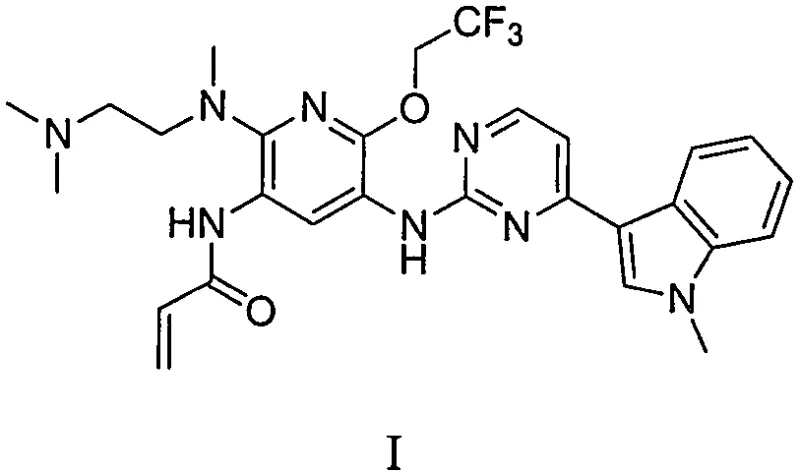
The significance of this technology lies in its ability to overcome the limitations of previous synthetic strategies, which often suffered from low overall yields and environmental hazards. For procurement managers and supply chain heads, understanding the technical nuances of this process is essential for evaluating potential suppliers who can deliver consistent quality. The patent details a comprehensive sequence involving condensation, substitution, reduction, acylation, and elimination reactions, all meticulously optimized to maximize efficiency. As a reliable pharmaceutical intermediates supplier, leveraging such advanced intellectual property ensures that the final active pharmaceutical ingredient meets stringent regulatory standards while maintaining cost-effectiveness throughout the value chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in patent application CN105315259A, present significant hurdles for scalable manufacturing. These conventional routes typically rely on the reduction of 6-chloro-2-trifluoroethoxy-3-nitropyridine using iron powder and ammonium chloride. This approach generates substantial waste residues, creating a burdensome post-treatment process that is detrimental to environmental compliance. Furthermore, the reliance on heavy metal catalysts in both coupling and hydrogenation reduction reactions introduces the risk of toxic metal residues in the final product, necessitating expensive and time-consuming purification steps. The economic viability of these older methods is further compromised by a remarkably low yield in the final acylation reaction, reported at only 23 percent, and a total route yield of merely 2.3 percent. Such inefficiencies render these processes unsuitable for modern industrial production where cost reduction in pharmaceutical manufacturing is paramount.
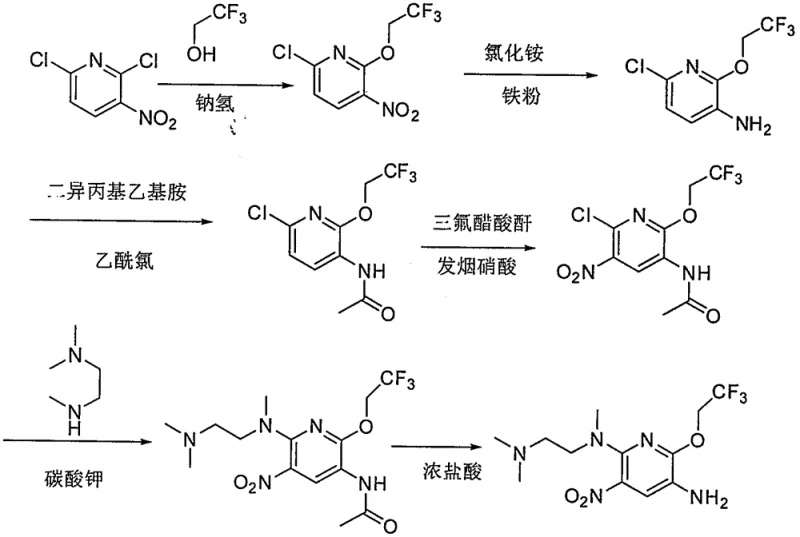
The Novel Approach
In stark contrast, the novel approach disclosed in CN110606842B utilizes a streamlined synthetic strategy that eliminates the need for problematic heavy metal catalysts and iron powder reductions. The new route employs sodium hydrosulfite for nitro reduction, a reagent that is not only environmentally friendly but also ensures high product purity without heavy metal contamination. A key innovation involves the formation of stable acid salts of intermediates, which precipitate as solids, allowing for simple filtration and removal of impurities dissolved in the organic solvent. This shift from complex chromatographic separations to straightforward crystallization and filtration drastically simplifies operations. The result is a process characterized by mild conditions, high total yield, and exceptional purity, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
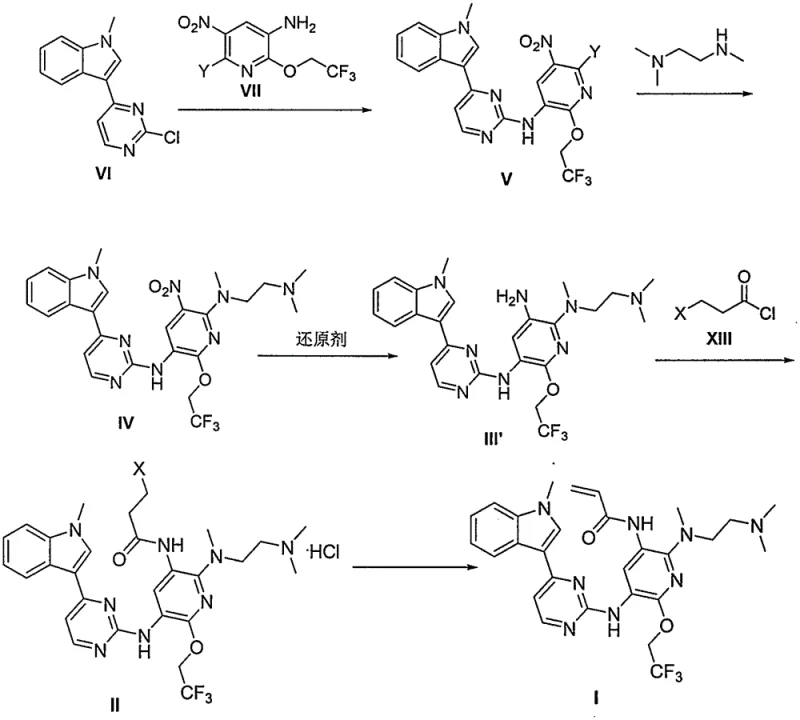
Mechanistic Insights into Sodium Hydrosulfite Reduction and Acylation
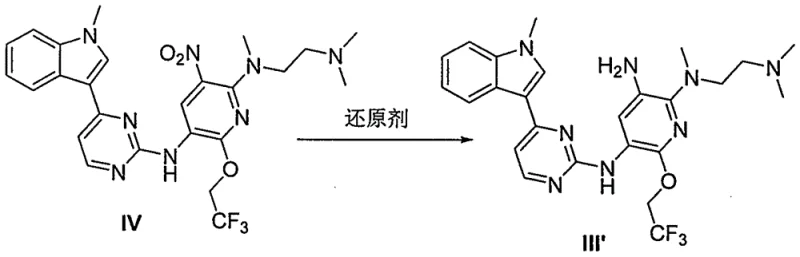
The core of this technological advancement lies in the mechanistic optimization of the reduction and acylation steps. Specifically, the reduction of the nitro group in compound IV to the amino group in compound III' is achieved using sodium hydrosulfite, potentially in the presence of an acid like hydrochloric acid. This acidic environment promotes the conversion of intermediate states to the final product, driving the reaction to completion with high efficiency. Unlike catalytic hydrogenation which requires specialized high-pressure equipment and precious metals, this chemical reduction operates under mild conditions, reducing capital expenditure and operational risks. The mechanism ensures that byproducts are minimized, directly contributing to the high purity observed in the final API intermediate. This attention to mechanistic detail is crucial for R&D directors focused on impurity profiles and process robustness.
Furthermore, the acylation reaction leading to intermediate II is engineered to exploit solubility differences for purification. By conducting the reaction in specific organic solvents such as dichloromethane or acetonitrile, the resulting hydrochloride salt of Formula II becomes insoluble and precipitates out of the solution. This phenomenon allows for the direct filtration of the product, effectively washing away soluble impurities without the need for additional workup procedures. The subsequent elimination reaction to form the acrylamide moiety in Formula I proceeds with high yield due to the high purity of the precursor. This integrated approach to reaction design, where purification is built into the synthesis steps themselves, exemplifies a sophisticated understanding of process chemistry that translates directly into commercial advantages.
How to Synthesize Pyridylamino Pyrimidine Derivative Efficiently
The synthesis of this complex molecule requires precise control over reaction parameters to ensure reproducibility and quality. The process begins with the condensation of specific precursors followed by a series of functional group transformations. Each step, from the initial etherification to the final elimination, is optimized for maximum conversion and ease of isolation. The detailed standardized synthesis steps see the guide below, which outlines the specific reagents, temperatures, and molar ratios required to achieve the reported high yields. Adhering to these protocols is essential for maintaining the integrity of the supply chain and ensuring that the final product meets the rigorous specifications demanded by the pharmaceutical industry.
- Condense compound VI with compound VII in the presence of acid to form intermediate V, followed by substitution with N,N,N'-trimethylethylenediamine to yield compound IV.
- Reduce the nitro group of compound IV using sodium hydrosulfite to obtain the amino intermediate III', optionally forming a stable salt (Formula III).
- Perform acylation with compound XIII to form intermediate II, followed by an elimination reaction under basic conditions to yield the final Formula I product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical superiority. The elimination of heavy metal catalysts and iron powder significantly reduces the complexity of waste management and lowers the cost associated with hazardous material disposal. This environmental friendliness aligns with global sustainability goals and reduces regulatory friction, ensuring smoother operations across international borders. Moreover, the simplified workup procedures, which replace column chromatography with filtration and crystallization, drastically reduce processing time and solvent consumption. These operational efficiencies translate into substantial cost savings and enhanced supply chain reliability, allowing manufacturers to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The replacement of expensive heavy metal catalysts and the avoidance of complex purification techniques like column chromatography lead to significant operational cost reductions. By utilizing cost-effective reagents such as sodium hydrosulfite and designing reactions where products precipitate naturally, the process minimizes material loss and energy consumption. This lean manufacturing approach ensures that the cost of goods sold is optimized, providing a competitive edge in the pricing of high-purity pharmaceutical intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent production output, mitigating the risk of supply disruptions. The use of stable intermediates, such as the acid salts of Formula III, allows for safer storage and transportation, reducing the likelihood of degradation during logistics. Additionally, the high yield and purity of the process mean that less raw material is required to produce the same amount of final product, securing the supply chain against fluctuations in raw material availability and ensuring timely delivery to downstream partners.
- Scalability and Environmental Compliance: Designed with industrial production in mind, this process scales efficiently from laboratory to commercial volumes without losing efficacy. The mild reaction conditions reduce the need for specialized high-pressure or high-temperature equipment, lowering capital investment barriers. Furthermore, the generation of fewer waste residues and the absence of toxic heavy metals simplify compliance with environmental regulations, facilitating faster approval processes and fostering a sustainable manufacturing ecosystem that is resilient to evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial aspects of this synthesis process. These insights are derived directly from the patent data to provide clarity on how this technology impacts production capabilities and product quality. Understanding these details helps stakeholders make informed decisions regarding sourcing and partnership opportunities in the pharmaceutical sector.
Q: How does the new process improve upon prior art methods for Formula I synthesis?
A: The new process eliminates the use of iron powder and heavy metal catalysts found in prior art, replacing them with environmentally friendly sodium hydrosulfite reduction. This avoids heavy metal residues, simplifies post-treatment by avoiding column chromatography, and significantly increases total yield from 2.3% to industrially viable levels.
Q: What are the key advantages of the salt formation step in this synthesis route?
A: Forming the HA acid salt of intermediate III' converts the free base into a stable solid that precipitates easily. This enhances storage stability, prevents deterioration, and facilitates filtration to remove impurities, ensuring higher purity and yield in the subsequent acylation and elimination steps.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is designed for industrial scalability. It utilizes mild reaction conditions, avoids complex purification techniques like column chromatography in favor of crystallization and filtration, and employs cost-effective reagents, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridylamino Pyrimidine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to meet the evolving needs of the global pharmaceutical market. Our expertise aligns perfectly with the innovations described in CN110606842B, allowing us to offer superior manufacturing solutions for EGFR inhibitor intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of volume requirements. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of Pyridylamino Pyrimidine Derivative meets the highest industry standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
