Advanced Grignard Carboxylation Strategy for Commercial Scale-up of 2,5-Dimethylphenylacetic Acid
Introduction to Patent CN110305010B Technology
The global demand for high-purity agrochemical intermediates continues to surge, driven by the need for efficient pest control solutions like spirotetramat. A pivotal breakthrough in this sector is documented in Chinese patent CN110305010B, which discloses a novel preparation method for 2,5-dimethylphenylacetic acid. This compound serves as a critical building block in the pharmaceutical and agrochemical industries, yet its traditional synthesis has long been plagued by environmental hazards and economic inefficiencies. The patented technology introduces a streamlined Grignard carboxylation strategy that fundamentally alters the production landscape by replacing toxic cyanating agents and costly noble metal catalysts with readily available magnesium and carbon dioxide. This shift not only addresses stringent regulatory pressures regarding hazardous waste but also offers a robust pathway for cost reduction in agrochemical intermediate manufacturing. By leveraging standard unit operations and benign reagents, this method ensures a reliable supply chain for high-purity 2,5-dimethylphenylacetic acid, positioning it as a superior choice for large-scale industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,5-dimethylphenylacetic acid has relied on several distinct pathways, each carrying significant drawbacks that hinder efficient commercial production. One prevalent method involves the cyanation of 2,5-dimethylbenzyl chloride followed by hydrolysis, a process depicted in early literature which suffers from a low total yield of approximately 38 percent and utilizes extremely toxic sodium cyanide reagents. 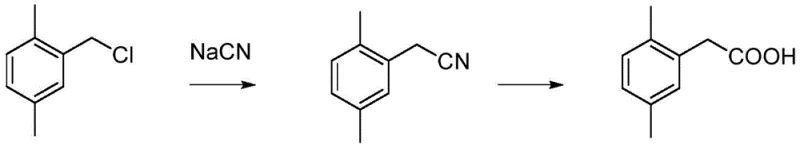 Another approach, disclosed in Bayer patents, employs a multi-step sequence involving chloroacetylation, ketalization, rearrangement, and hydrolysis, resulting in an overly complicated process flow that increases operational complexity and capital expenditure. Furthermore, alternative routes utilizing sulfur-containing coupling reagents generate substantial amounts of foul-smelling sulfide waste, creating severe environmental compliance challenges for manufacturing facilities. Perhaps most critically for cost-sensitive applications, carbonylation methods utilizing p-xylene derivatives require expensive palladium or rhodium catalysts, such as tetraphenylphosphonium palladium, which not only inflate raw material costs but also introduce risks of heavy metal contamination in the final product.
Another approach, disclosed in Bayer patents, employs a multi-step sequence involving chloroacetylation, ketalization, rearrangement, and hydrolysis, resulting in an overly complicated process flow that increases operational complexity and capital expenditure. Furthermore, alternative routes utilizing sulfur-containing coupling reagents generate substantial amounts of foul-smelling sulfide waste, creating severe environmental compliance challenges for manufacturing facilities. Perhaps most critically for cost-sensitive applications, carbonylation methods utilizing p-xylene derivatives require expensive palladium or rhodium catalysts, such as tetraphenylphosphonium palladium, which not only inflate raw material costs but also introduce risks of heavy metal contamination in the final product.
The Novel Approach
In stark contrast to these legacy technologies, the method described in CN110305010B utilizes a direct Grignard carboxylation route that simplifies the synthetic architecture to just two primary chemical transformations. The process begins with the formation of a Grignard reagent from 2,5-dimethylbenzyl halide and magnesium, followed immediately by reaction with carbon dioxide to yield the target acid. 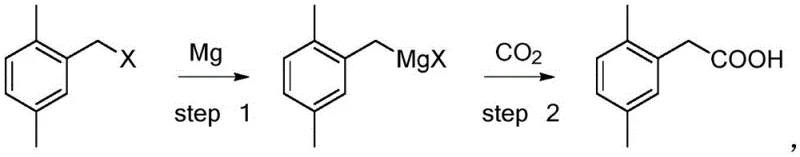 This innovative pathway completely circumvents the need for virulent cyaniding reagents and precious metal catalysts, thereby eliminating the associated procurement costs and disposal liabilities. Experimental data from the patent demonstrates that this approach achieves mass yields exceeding 92 percent with product purity consistently above 99 percent, significantly outperforming the 38 percent yield of the cyanation route. By shortening the reaction steps and utilizing environment-friendly reagents like magnesium and CO2, this novel approach offers a scalable, economically viable, and sustainable solution for the commercial scale-up of complex agrochemical intermediates.
This innovative pathway completely circumvents the need for virulent cyaniding reagents and precious metal catalysts, thereby eliminating the associated procurement costs and disposal liabilities. Experimental data from the patent demonstrates that this approach achieves mass yields exceeding 92 percent with product purity consistently above 99 percent, significantly outperforming the 38 percent yield of the cyanation route. By shortening the reaction steps and utilizing environment-friendly reagents like magnesium and CO2, this novel approach offers a scalable, economically viable, and sustainable solution for the commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into Grignard Carboxylation
The core of this technological advancement lies in the precise control of the Grignard reagent formation and its subsequent nucleophilic attack on carbon dioxide. The reaction initiates with the oxidative insertion of magnesium into the carbon-halogen bond of 2,5-dimethylbenzyl halide, a process facilitated by initiators such as iodine or 1,2-dibromoethane to overcome the induction period. The patent specifies a critical molar ratio of halide to magnesium ranging from 1:1.05 to 1:1.2, ensuring complete consumption of the organic halide while minimizing the formation of Wurtz-type coupling byproducts (dimers) that often plague Grignard reactions. The use of anhydrous ether solvents like tetrahydrofuran (THF) or diethyl ether is essential to stabilize the organomagnesium species through coordination, maintaining the reactivity required for the subsequent carboxylation step. Temperature control during the addition of the halide is also paramount; the protocol dictates a slow, batched addition to manage the exothermic nature of the Grignard formation, preventing thermal runaway and ensuring a steady concentration of the active reagent.
Following the generation of the Grignard intermediate, the mechanism proceeds via the nucleophilic attack of the carbanion equivalent on the electrophilic carbon of carbon dioxide. This step forms a magnesium carboxylate salt, which is stable under the reaction conditions until acidic workup. The patent highlights the flexibility of using either gaseous CO2 or dry ice, with a molar excess of CO2 (up to 10 equivalents) driving the equilibrium towards the product and suppressing side reactions. The subsequent quenching with saturated ammonium chloride solution effectively protonates the carboxylate to release the free acid while dissolving magnesium salts into the aqueous phase. This mechanistic clarity allows for rigorous impurity control; by optimizing the stoichiometry and maintaining an inert nitrogen atmosphere throughout, the process minimizes oxidation byproducts and ensures the final crystalline solid meets the stringent purity specifications of 99.1 percent to 99.3 percent required for downstream pesticide synthesis.
How to Synthesize 2,5-Dimethylphenylacetic Acid Efficiently
The implementation of this Grignard-based synthesis requires strict adherence to anhydrous conditions and controlled addition rates to maximize yield and safety. The process is designed to be operationally simple, utilizing standard reactor setups common in fine chemical manufacturing, yet it demands precision in reagent ratios and temperature management to replicate the high efficiencies reported in the patent literature. Operators must ensure the complete removal of moisture from the system prior to magnesium addition, as water acts as a potent quencher for the Grignard reagent, leading to significant yield losses. The following guide outlines the standardized operational parameters derived directly from the patent examples, providing a clear roadmap for technical teams to validate this route in pilot or production scales. For detailed procedural instructions, please refer to the structured synthesis guide below.
- Formation of Grignard Reagent: React 2,5-dimethylbenzyl halide with magnesium chips in anhydrous ether or THF solvent using iodine as an initiator at 30-60°C.
- Carboxylation Reaction: Introduce dry carbon dioxide gas or dry ice to the Grignard solution at 20°C to form the carboxylate salt.
- Workup and Purification: Quench with saturated ammonium chloride, extract with organic solvents like dichloromethane or ethyl acetate, and distill to obtain high-purity acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this Grignard carboxylation methodology represents a strategic opportunity to optimize both cost structures and supply reliability. The elimination of noble metal catalysts such as palladium and rhodium removes a major source of price volatility and supply risk, as these metals are subject to fluctuating global market prices and geopolitical constraints. Furthermore, the avoidance of highly toxic sodium cyanide simplifies the regulatory compliance landscape, reducing the administrative burden and insurance costs associated with handling Schedule 1 chemicals. This shift towards benign reagents aligns perfectly with modern ESG (Environmental, Social, and Governance) goals, facilitating smoother audits and fostering stronger relationships with sustainability-focused downstream partners. The simplified process flow, characterized by fewer unit operations and shorter reaction times, inherently enhances throughput capacity, allowing manufacturers to respond more agilely to market demand fluctuations without requiring massive capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the substitution of expensive catalytic systems with commodity-grade magnesium and carbon dioxide. By removing the requirement for ligands like triphenylphosphine and precious metals, the direct material cost per kilogram of product is significantly lowered. Additionally, the high yield of over 92 percent reduces the consumption of starting materials per unit of output, further driving down the variable cost of goods sold. The simplified workup procedure, which avoids complex distillation or chromatography steps often needed to remove metal residues, also translates into reduced utility consumption and labor hours, contributing to substantial overall cost savings in the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals like magnesium turnings and industrial grade CO2 ensures a robust and resilient supply chain that is less susceptible to disruptions compared to specialized catalyst supply lines. Magnesium is produced globally in vast quantities for various metallurgical and chemical applications, guaranteeing consistent availability and competitive pricing. This stability allows for long-term procurement planning and inventory optimization, reducing the need for safety stock buffers that tie up working capital. Moreover, the use of standard solvents like THF and ethyl acetate, which are staples in the chemical industry, further mitigates the risk of raw material shortages that could otherwise halt production lines.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this process offers distinct advantages by eliminating the generation of sulfur-containing waste streams and cyanide-laden effluents. The primary byproduct is magnesium salts, which are relatively benign and easier to treat or dispose of in accordance with environmental regulations. This reduction in hazardous waste volume lowers disposal costs and minimizes the environmental footprint of the facility. The process is inherently scalable, as Grignard chemistry is a well-understood unit operation in the fine chemical sector, allowing for seamless translation from laboratory bench scale to multi-ton commercial production without the need for exotic reactor designs or specialized containment systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, based on the specific data and claims found within the patent documentation. These insights are intended to clarify the operational feasibility and strategic benefits for stakeholders evaluating this technology for potential adoption or outsourcing. Understanding these nuances is critical for making informed decisions about process validation and supplier qualification in the competitive agrochemical intermediate market.
Q: Why is the Grignard route preferred over cyanation for 2,5-dimethylphenylacetic acid?
A: The Grignard route avoids the use of highly toxic sodium cyanide (NaCN) required in traditional cyanation-hydrolysis methods, significantly improving operational safety and reducing hazardous waste treatment costs.
Q: What represents the main cost advantage of this synthesis method?
A: This method eliminates the need for expensive noble metal catalysts such as palladium or rhodium complexes, which are required in carbonylation routes, thereby drastically reducing raw material costs.
Q: What purity levels can be achieved with this Grignard carboxylation process?
A: According to patent data, the process consistently yields 2,5-dimethylphenylacetic acid with purity exceeding 99%, suitable for direct use in sensitive agrochemical applications like spirotetramat synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Dimethylphenylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Grignard carboxylation route for producing high-quality 2,5-dimethylphenylacetic acid. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing environments. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for spirotetramat synthesis. We are committed to delivering not just a chemical product, but a secure and efficient supply solution that aligns with your long-term strategic goals.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the superior quality and reliability of our 2,5-dimethylphenylacetic acid supply before making any commitments.
