Optimizing Valaciclovir Hydrochloride Production: A Novel Transfer Hydrogenation Strategy for Commercial Scale-up
Optimizing Valaciclovir Hydrochloride Production: A Novel Transfer Hydrogenation Strategy for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with operational safety, particularly for antiviral agents like Valaciclovir Hydrochloride. Patent CN109651371B introduces a refined preparation method that addresses the limitations of traditional high-pressure hydrogenation techniques. This innovation leverages catalytic transfer hydrogenation to convert protected intermediates into the final active pharmaceutical ingredient under remarkably mild conditions. By shifting away from hazardous gaseous hydrogen environments, this methodology not only enhances workplace safety but also simplifies the engineering requirements for large-scale manufacturing facilities. The process ensures that the final product achieves stringent pharmacopoeia standards, making it an ideal candidate for reliable pharmaceutical intermediate suppliers aiming to optimize their production lines.
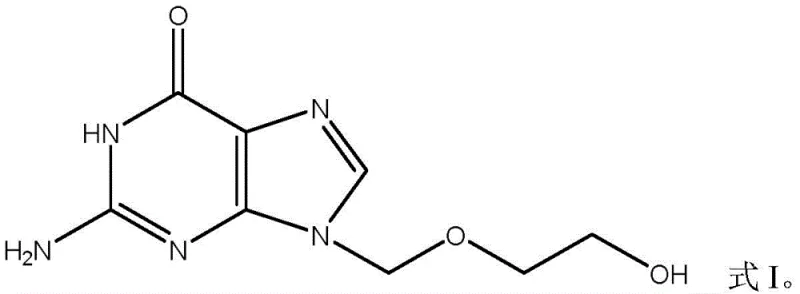
Valaciclovir serves as the L-valyl ester prodrug of acyclovir, designed specifically to overcome the poor oral absorption characteristics of its parent compound. As illustrated in the structural comparison, the addition of the valine moiety significantly enhances bioavailability, allowing for reduced dosing frequencies and improved patient compliance in treating herpes virus infections. The chemical transformation from the base structure to the esterified prodrug requires precise control over stereochemistry and functional group protection to prevent racemization or degradation. Understanding these structural nuances is critical for procurement managers evaluating the complexity of the supply chain and the potential for cost reduction in pharmaceutical manufacturing.
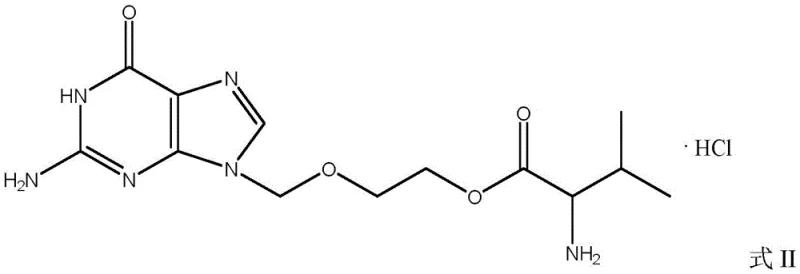
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Valaciclovir Hydrochloride has relied heavily on catalytic hydrogenation using molecular hydrogen gas, as disclosed in earlier patents such as US4957924. This conventional approach necessitates the use of specialized high-pressure equipment, such as autoclaves, which introduces significant capital costs and safety hazards related to hydrogen storage and handling. Furthermore, the requirement for strict pressure control often complicates the scale-up process, leading to potential bottlenecks in continuous manufacturing workflows. Alternative methods involving Boc-protection strategies, while avoiding high pressure, often require harsh acidic conditions for deprotection that can compromise the integrity of the sensitive purine ring system. These legacy processes frequently result in lower overall yields and generate complex waste streams that challenge environmental compliance teams.
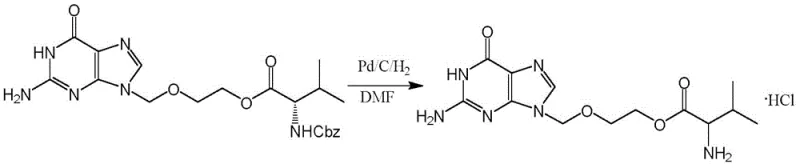
The Novel Approach
The methodology outlined in CN109651371B represents a paradigm shift by employing catalytic transfer hydrogenation using formic acid as the hydrogen donor. This technique effectively bypasses the need for gaseous hydrogen, allowing the reaction to proceed at atmospheric pressure and moderate temperatures ranging from 20°C to 40°C. The elimination of high-pressure infrastructure drastically reduces the barrier to entry for commercial scale-up of complex pharmaceutical intermediates. Additionally, the use of formic acid generates carbon dioxide as a benign byproduct, aligning with green chemistry principles and simplifying waste treatment protocols. This novel approach ensures a smoother transition from laboratory benchtop to industrial reactor without compromising the stereochemical purity of the L-valine moiety.
Mechanistic Insights into Catalytic Transfer Hydrogenation and Coupling
The core of this synthetic strategy lies in the efficient coupling of acyclovir with N-benzyloxycarbonyl-L-valine followed by a sophisticated deprotection sequence. The initial esterification utilizes N,N'-dicyclohexylcarbodiimide (DCC) and 4-dimethylaminopyridine (DMAP) in dimethylformamide (DMF) to activate the carboxylic acid. A critical mechanistic feature is the split-addition protocol, where 90-95% of the reagents are added initially, followed by a supplementary dose to drive the reaction to completion. This kinetic control minimizes the formation of N-acylurea byproducts and ensures high conversion rates. Subsequently, the removal of the Cbz protecting group is achieved via palladium-catalyzed transfer hydrogenation, where formic acid decomposes on the metal surface to provide nascent hydrogen for the reduction, avoiding the mass transfer limitations often seen with gaseous hydrogen.
Impurity control is rigorously managed through temperature regulation and solvent selection throughout the reaction pathway. Maintaining the coupling reaction between 5°C and 15°C prevents thermal degradation of the activated ester intermediate, while the subsequent deprotection at 40°C ensures rapid kinetics without promoting hydrolysis of the newly formed ester bond. The final crystallization step utilizes a methanol and acetone solvent system to selectively precipitate the hydrochloride salt while keeping organic impurities in solution. This precise manipulation of solubility parameters is essential for achieving the high-purity specifications required for regulatory approval and demonstrates the depth of process optimization embedded in this patent.
How to Synthesize Valaciclovir Hydrochloride Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for producing high-quality Valaciclovir Hydrochloride suitable for clinical applications. The process begins with the activation of the amino acid followed by nucleophilic attack from the hydroxyl group of acyclovir, creating the protected intermediate. This is immediately followed by a one-pot deprotection and salt formation sequence that streamlines the workflow. For R&D teams looking to implement this technology, the detailed standardized synthesis steps provided below offer a reproducible framework for optimizing reaction parameters and maximizing yield.
- Coupling Reaction: React Acyclovir with N-benzyloxycarbonyl-L-valine using DCC and DMAP in DMF at 5-15°C, utilizing a split-addition strategy for reagents to maximize yield.
- Deprotection and Salt Formation: Perform catalytic transfer hydrogenation using Pd/C and formic acid in methanol at 20-40°C, followed by acidification with hydrochloric acid.
- Purification: Recrystallize the crude product from a methanol and acetone solvent system to achieve pharmacopoeia-grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this transfer hydrogenation methodology offers substantial strategic benefits beyond mere technical feasibility. By removing the dependency on high-pressure hydrogenation infrastructure, manufacturers can significantly reduce capital expenditure and maintenance costs associated with specialized reactors. This shift allows for greater flexibility in production scheduling and reduces the risk of downtime caused by equipment failure or safety inspections. Furthermore, the use of readily available reagents like formic acid and standard palladium catalysts ensures a stable supply chain that is less susceptible to geopolitical fluctuations affecting specialty gas supplies.
- Cost Reduction in Manufacturing: The elimination of high-pressure autoclaves and the associated safety systems leads to a drastic simplification of the production facility requirements. Without the need for expensive hydrogen gas handling systems, operational overheads are significantly lowered, allowing for more competitive pricing structures in the global market. The mild reaction conditions also reduce energy consumption for heating and cooling, contributing to long-term operational savings and a smaller carbon footprint for the manufacturing site.
- Enhanced Supply Chain Reliability: Utilizing liquid-phase hydrogen donors like formic acid mitigates the logistical challenges and safety risks associated with transporting and storing compressed hydrogen gas. This change enhances the continuity of supply by removing a critical bottleneck often found in traditional hydrogenation processes. Additionally, the robustness of the reaction conditions means that production can be maintained consistently even with minor variations in raw material quality, ensuring reliable delivery timelines for downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable due to the absence of gas-liquid mass transfer limitations that typically hinder large-scale hydrogenation reactions. The generation of carbon dioxide as the primary byproduct simplifies waste management and aligns with increasingly stringent environmental regulations regarding volatile organic compounds and hazardous emissions. This environmental compatibility facilitates faster regulatory approvals and supports the sustainability goals of modern pharmaceutical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on yield expectations and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for commercial production.
Q: Why is transfer hydrogenation preferred over high-pressure hydrogenation for Valaciclovir synthesis?
A: Transfer hydrogenation using formic acid eliminates the need for specialized high-pressure autoclaves and gaseous hydrogen, significantly reducing capital expenditure and safety risks associated with industrial scale-up.
Q: How does the split-addition of coupling reagents improve the process?
A: Adding the majority of N-benzyloxycarbonyl-L-valine and DCC initially, followed by a supplementary addition, drives the equilibrium towards completion and minimizes side reactions, resulting in higher purity and yield of the intermediate.
Q: What are the critical quality attributes controlled in this synthesis?
A: The process strictly controls residual solvents, heavy metals (from Pd/C catalyst), and specific impurities related to incomplete deprotection, ensuring the final product meets the latest pharmacopoeia standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valaciclovir Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the production of high-value antiviral intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and compliant. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Valaciclovir Hydrochloride meets the highest international standards, providing our partners with the confidence they need to bring life-saving medications to market.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can drive value and efficiency for your organization.
