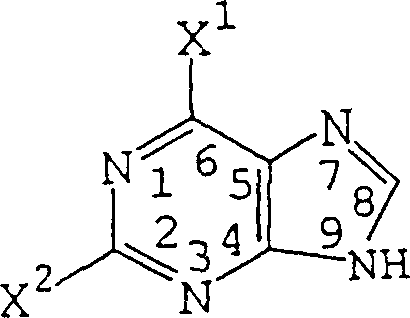
Advanced Synthesis of 2,6-Dihalopurine: Technical Breakthroughs for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of potent antiviral and oncology therapeutics has placed 2,6-dihalopurines at the center of synthetic attention, serving as critical scaffolds for next-generation nucleoside analogs. Patent CN1314685C introduces a transformative preparation method that addresses long-standing inefficiencies in producing these vital intermediates, offering a pathway that combines high yield with operational simplicity. Historically, the synthesis of 2,6-dihalopurines was plagued by harsh reaction conditions, multi-step sequences, and difficult purification protocols that hindered commercial viability. This innovative approach leverages a unique combination of halosilane compounds and diazotization reagents to achieve selective halogenation under remarkably mild conditions. By redefining the synthetic landscape for purine derivatives, this technology provides a robust foundation for the reliable pharmaceutical intermediate supplier seeking to optimize their production pipelines. The method not only enhances chemical efficiency but also aligns with modern green chemistry principles by reducing energy consumption and hazardous waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for synthesizing 2,6-dihalopurines have historically imposed significant burdens on manufacturing infrastructure and safety protocols. One prevalent method involves the chlorination of xanthines using pyrophosphoryl chloride, which necessitates the complex and hazardous in-situ preparation of the chlorinating agent from phosphorus oxychloride. This process demands extreme reaction temperatures reaching 165°C and requires specialized corrosion-resistant reaction vessels to withstand the aggressive chemical environment over prolonged periods of approximately 19 hours. Alternative routes utilizing barbituric acid as a starting material involve cumbersome four-step sequences that accumulate impurities and reduce overall atom economy. Furthermore, strategies employing 9-alkylated purine precursors often result in low yields, such as the 40% yield reported in older literature, and face the critical bottleneck of removing the alkyl group at the 9-position to obtain the desired unsubstituted product. These legacy methods collectively represent a substantial barrier to efficient cost reduction in pharmaceutical intermediate manufacturing due to their operational complexity and resource intensity.
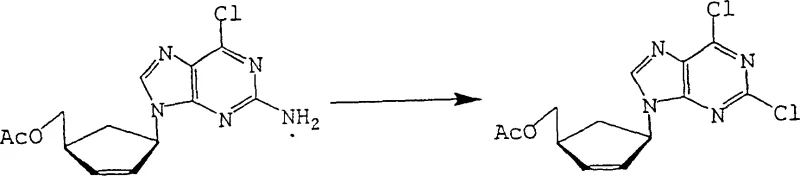
The Novel Approach
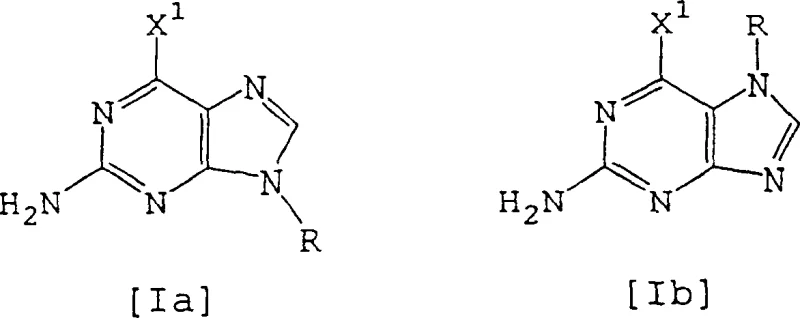
In stark contrast to these archaic techniques, the novel approach detailed in the patent utilizes a direct and elegant transformation of 2-amino-6-halopurine derivatives into the target 2,6-dihalopurines. By reacting compounds of formula [Ia] or [Ib], where the nitrogen atoms may be protected by acyl groups, with halosilane compounds and nitrite reagents, the process achieves high conversion rates without the need for extreme thermal energy. This method effectively bypasses the need for pyrophosphoryl chloride, replacing it with more manageable reagents like trimethylchlorosilane or dimethyldichlorosilane that function efficiently at temperatures between 20°C and 60°C. The strategic use of an acyl protecting group at the 7 or 9 position not only directs the regioselectivity of the reaction but also facilitates the isolation of the final product through simple filtration and crystallization. This paradigm shift allows for the commercial scale-up of complex pharmaceutical intermediates with significantly reduced lead times and enhanced process safety profiles.
Mechanistic Insights into Halosilane-Mediated Diazotization
The core of this technological advancement lies in the sophisticated interplay between the halosilane reagent and the diazotization agent, which orchestrates the replacement of the amino group with a halogen atom. The reaction initiates with the formation of a diazonium species at the 2-position of the purine ring, driven by the nitrite reagent such as isoamyl nitrite in the presence of the halosilane. The halosilane acts as both a source of the halogen atom and a silylating agent that stabilizes intermediates, ensuring that the substitution proceeds cleanly without degrading the sensitive purine skeleton. The presence of a phase transfer catalyst, specifically quaternary ammonium salts like tetraethylammonium chloride, further accelerates the reaction kinetics by facilitating the interaction between reagents in the organic phase. This mechanistic pathway avoids the formation of stable N-oxides or other stubborn byproducts that typically complicate purification in traditional chlorination methods. Consequently, the reaction mixture yields the target 2,6-dihalopurine in a form that is easily separable, often precipitating directly from the reaction solvent upon cooling.
Impurity control is inherently built into this mechanism through the careful selection of the acyl protecting group, typically an acetyl group, which modulates the electronic properties of the purine ring. By temporarily blocking the N9 or N7 position, the synthesis prevents unwanted polymerization or side-reactions that could occur at these nucleophilic sites during the harsh diazotization conditions. The acyl group is subsequently removed or retained depending on the specific downstream requirements, but its presence during the critical halogenation step ensures a cleaner impurity profile. This level of control is crucial for meeting the stringent purity specifications required for active pharmaceutical ingredients, as it minimizes the formation of regioisomers and over-halogenated byproducts. The ability to tune the reaction by selecting specific halosilanes, such as switching from chlorosilanes to bromosilanes, further demonstrates the versatility of this mechanism in generating diverse halogenated purine libraries for drug discovery.
How to Synthesize 2,6-Dihalopurine Efficiently
The synthesis of 2,6-dihalopurine via this patented route represents a significant optimization of process chemistry, designed to maximize yield while minimizing operational hazards. The procedure begins with the preparation of the acylated starting material, where 2-amino-6-chloropurine is treated with acetic anhydride to install the acetyl group, a step that proceeds with nearly quantitative yield. Following this protection step, the key transformation occurs by combining the acylated intermediate with a halosilane and isoamyl nitrite in a solvent system such as heptane or o-dichlorobenzene. The reaction is maintained at mild temperatures, typically around 50-60°C, for a duration of 10 to 20 hours, allowing the diazotization and halogen exchange to reach completion without thermal degradation. Detailed standardized synthesis steps see the guide below.
- Prepare the starting material by introducing an acyl group (preferably acetyl) to the 7 or 9 position of 2-amino-6-halopurine to enhance reactivity and isolation properties.
- React the acylated intermediate with a halosilane compound (e.g., trimethylchlorosilane) and a diazotization reagent (e.g., isoamyl nitrite) in a hydrocarbon or halogenated solvent.
- Conduct the reaction at mild temperatures (20-60°C) in the presence of a phase transfer catalyst, followed by pH adjustment and filtration to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method translates into tangible improvements in cost structure and supply reliability. The elimination of pyrophosphoryl chloride removes the need for specialized corrosion-resistant reactors and the associated maintenance costs, leading to substantial capital expenditure savings over the lifecycle of the production facility. Furthermore, the ability to operate at near-ambient pressures and moderate temperatures drastically reduces energy consumption compared to the 165°C requirements of conventional methods. The simplified workup procedure, which often involves mere filtration and washing rather than complex distillation or chromatography, significantly shortens the batch cycle time. These factors combine to create a more resilient supply chain capable of responding rapidly to market demand fluctuations without the bottlenecks associated with hazardous chemical handling.
- Cost Reduction in Manufacturing: The process eliminates the expensive and dangerous preparation of pyrophosphoryl chloride, replacing it with commercially abundant halosilanes that are easier to handle and store. By avoiding the multi-step sequences associated with barbituric acid routes, the overall material throughput is increased, reducing the cost of goods sold per kilogram of active intermediate. The high yield achieved in the acylation step ensures that raw material waste is minimized, contributing to a leaner and more economical manufacturing process. Additionally, the reduced need for extensive purification equipment lowers the barrier to entry for contract manufacturing organizations looking to produce these intermediates.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, such as isoamyl nitrite and trimethylchlorosilane, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to interruptions caused by equipment failure or utility fluctuations, ensuring consistent delivery schedules. The stability of the acylated intermediates allows for potential stockpiling or semi-finished goods storage, providing a buffer against sudden spikes in demand for nucleoside analog precursors. This reliability is critical for maintaining the continuity of supply for downstream pharmaceutical customers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the absence of exothermic runaways or high-pressure requirements, making it safer for large reactor volumes. The use of hydrocarbon solvents like heptane simplifies solvent recovery and recycling systems, aligning with increasingly strict environmental regulations regarding volatile organic compound emissions. The reduction in hazardous waste generation, particularly the avoidance of phosphorus-containing byproducts, streamlines waste disposal logistics and reduces environmental compliance costs. This environmentally friendly profile enhances the corporate sustainability metrics of manufacturers adopting this technology, appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 2,6-dihalopurine synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows. The insights provided here clarify the operational parameters and expected outcomes for teams considering this advanced synthetic route.
Q: What are the primary advantages of the halosilane method over traditional phosphorus oxychloride chlorination?
A: The halosilane method operates at significantly lower temperatures (20-60°C vs 165°C) and avoids the preparation of corrosive pyrophosphoryl chloride, resulting in safer operations and reduced equipment maintenance costs.
Q: How does the acyl protection strategy improve the isolation of 2,6-dihalopurines?
A: Introducing an acyl group at the N9 or N7 position prevents unwanted side reactions and modifies the solubility profile of the intermediate, allowing for simple filtration and crystallization rather than complex chromatographic purification.
Q: Is this process suitable for large-scale manufacturing of nucleoside analog precursors?
A: Yes, the process utilizes commercially available reagents like isoamyl nitrite and trimethylchlorosilane, avoids extreme pressure or temperature conditions, and offers high yields with straightforward workup procedures ideal for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dihalopurine Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality intermediates play in the development of life-saving medications, and we are committed to delivering excellence in every batch. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that our 2,6-dihalopurine products meet the exacting standards required by global regulatory bodies. Our commitment to quality is matched only by our dedication to customer partnership, providing the stability and consistency needed for long-term pharmaceutical projects.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this superior manufacturing process. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the performance of our materials in your downstream applications. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and securing a competitive advantage in the global marketplace.
