Advanced Synthetic Strategy for High-Purity 7-Oxoacetate Abiraterone Impurity Standards
The pharmaceutical industry's relentless pursuit of safety and efficacy in oncology treatments has placed a premium on rigorous impurity profiling, particularly for potent agents like Abiraterone Acetate (ZYTIGA). As detailed in patent CN111253461A, a significant breakthrough has been achieved in the synthesis of 7-oxoacetate abiraterone, a critical degradation product and process-related impurity identified during the stability studies of this CYP17 inhibitor. Historically, the lack of a reported synthetic method for this specific steroid derivative created a bottleneck for regulatory compliance and quality assurance teams who require authentic reference standards to validate analytical methods. This patent discloses a robust, multi-step synthetic pathway that not only fills this literature gap but also establishes a reproducible protocol for generating high-purity material essential for toxicological assessment and batch release testing.
The strategic importance of this synthesis extends beyond mere academic curiosity; it addresses a fundamental need in the supply chain for reliable pharmaceutical intermediate supplier capabilities. By enabling the directional synthesis of 7-oxoacetate abiraterone, manufacturers can now implement stricter control strategies over their Abiraterone Acetate production lines. The ability to quantify this impurity accurately ensures that the final drug product meets the stringent safety profiles required by global health authorities. Furthermore, understanding the formation mechanism of this 7-oxo species allows process chemists to tweak upstream fermentation or chemical synthesis parameters to minimize its generation, thereby enhancing overall yield and reducing waste in the manufacturing of the active pharmaceutical ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in this patent, the synthesis of complex steroid impurities like 7-oxoacetate abiraterone was fraught with challenges related to regioselectivity and functional group tolerance. Conventional approaches often attempted to introduce the 7-oxo moiety late in the synthesis or through non-selective oxidation of the final API, which invariably led to complex mixtures of over-oxidized byproducts. The steroid nucleus, with its multiple chiral centers and sensitive olefinic bonds, is notoriously difficult to manipulate without affecting other parts of the molecule. Traditional oxidation methods lacked the precision to target the C7 position exclusively while leaving the C3 acetate and the C17 pyridyl substituent intact. This lack of specificity resulted in low yields, difficult purification processes requiring extensive chromatography, and ultimately, an inability to secure sufficient quantities of the impurity for comprehensive safety studies.
The Novel Approach
The novel approach outlined in the patent revolutionizes this landscape by employing a convergent strategy that prioritizes the installation of the 7-oxo group prior to the introduction of the sensitive pyridine ring. This sequence begins with the acetylation of a readily available iodo-androstadienol precursor, followed by a controlled oxidation step using chromium trioxide to establish the ketone functionality at the C7 position. Only after the steroid backbone is fully functionalized with the oxygenated groups is the pyridine ring installed via a palladium-catalyzed cross-coupling reaction. This logical progression ensures that the harsh oxidative conditions do not compromise the heteroaromatic system. 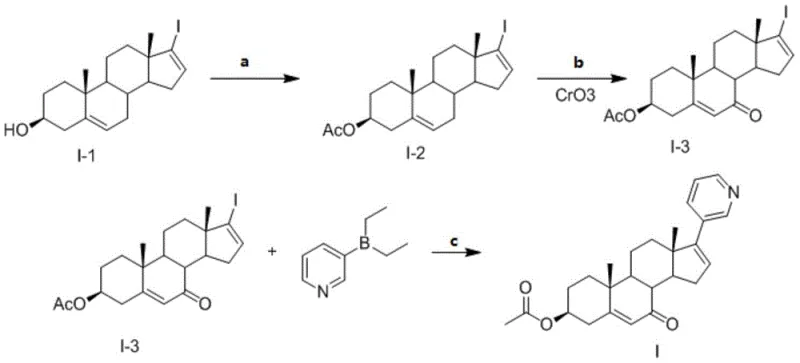
This methodology represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing by simplifying the purification workflow. Because the reaction sequence is designed to minimize side products, the reliance on expensive preparative HPLC is significantly reduced in favor of standard crystallization techniques. The use of common reagents such as acetic anhydride, chromium trioxide, and diethyl(3-pyridyl)borane ensures that the supply chain remains resilient and cost-effective. Moreover, the final recrystallization step from ethanol yields the target compound with a purity exceeding 98%, demonstrating that high quality does not necessarily require exotic or prohibitively expensive processing conditions.
Mechanistic Insights into Suzuki-Miyaura Coupling and Steroid Oxidation
The cornerstone of this synthetic route is the final Suzuki-Miyaura coupling reaction, which joins the sterically hindered C17 position of the steroid nucleus with the 3-pyridyl borane species. Mechanistically, this transformation relies on a catalytic cycle involving a palladium(0) species, generated in situ from precursors like bis(triphenylphosphine)palladium(II) chloride. The cycle initiates with the oxidative addition of the palladium catalyst into the carbon-iodine bond at the C17 position of the steroid D-ring. This step is critical because the C17 center is quaternary and highly congested, requiring a catalyst system with sufficient electronic richness and steric bulk to facilitate bond activation. Following oxidative addition, transmetallation occurs with the diethyl(3-pyridyl)borane, activated by the inorganic base such as tripotassium phosphate. 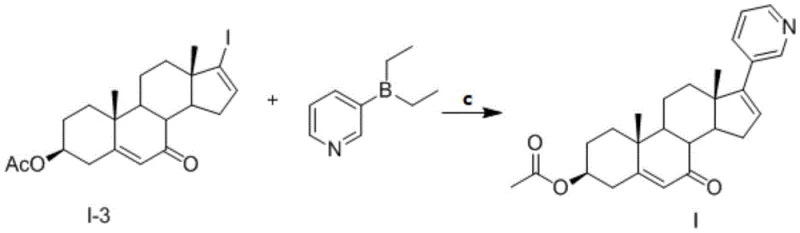
The subsequent reductive elimination releases the final 7-oxoacetate abiraterone product and regenerates the active palladium catalyst. The choice of solvent, specifically N-methylpyrrolidone (NMP), plays a vital role in stabilizing the polar transition states and ensuring the solubility of the bulky steroid intermediate. In parallel, the oxidation step utilizing chromium trioxide (CrO3) in the presence of N-hydroxyphthalimide (NOP) demonstrates high regioselectivity for the allylic C7 position. This selectivity is driven by the electronic activation of the C7 hydrogen atoms adjacent to the C5-C6 double bond. By carefully controlling the stoichiometry and temperature (50-70°C), the process avoids over-oxidation or epoxidation of the double bond, preserving the integrity of the steroid skeleton. This precise control over reaction parameters is what enables the production of high-purity steroid intermediates suitable for regulatory submission.
How to Synthesize 7-Oxoacetate Abiraterone Efficiently
The synthesis of this critical impurity standard is designed to be operationally simple, leveraging standard laboratory equipment and widely available chemical reagents to ensure reproducibility across different manufacturing sites. The process flow is linear and robust, beginning with the protection of the 3-hydroxyl group to prevent unwanted side reactions during the subsequent oxidation phase. The detailed protocol emphasizes the importance of temperature control during the acetylation and oxidation steps to maximize yield and minimize the formation of regioisomers. For research and development teams looking to implement this pathway, the patent provides specific molar ratios and solvent systems that have been optimized to balance reaction rate with selectivity. The detailed standardized synthesis steps are provided in the guide below.
- Acetylation of 17-iodoandrosta-5,16-dien-3β-ol using acetic anhydride in pyridine to protect the 3-hydroxyl group.
- Selective oxidation of the protected intermediate using chromium trioxide (CrO3) to introduce the 7-oxo functionality.
- Suzuki coupling of the 7-oxo-iodo steroid with diethyl(3-pyridyl)borane followed by recrystallization to achieve >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic method offers substantial advantages by decoupling the production of impurity standards from the complexities of the main API synthesis. The reliance on commodity chemicals such as pyridine, acetic anhydride, and ethanol means that raw material sourcing is straightforward and less susceptible to market volatility compared to processes requiring specialized ligands or rare earth catalysts. The operational simplicity, characterized by standard filtration and crystallization unit operations, translates directly into lower capital expenditure requirements for manufacturing facilities. This accessibility allows for a more diversified supplier base, reducing the risk of single-source dependency and ensuring a continuous supply of critical quality control materials for global pharmaceutical companies.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification steps in favor of crystallization significantly lowers the cost of goods sold (COGS) for this intermediate. By achieving high purity (>98%) through recrystallization alone, the process avoids the high solvent consumption and low throughput associated with preparative chromatography. Furthermore, the use of catalytic amounts of palladium, which can potentially be recovered or used at low loadings (1.0-10.0% Wt), minimizes the impact of precious metal costs on the overall budget. This economic efficiency makes the routine production of reference standards financially viable for quality control laboratories.
- Enhanced Supply Chain Reliability: The synthetic route utilizes robust chemistry that is less sensitive to minor fluctuations in reaction conditions, thereby increasing the reliability of batch-to-batch consistency. The starting materials, specifically the iodo-androstadienol derivatives, are accessible through established supply chains for steroid intermediates. This availability ensures that production schedules can be maintained without long lead times for custom synthesis of exotic building blocks. For supply chain managers, this translates to predictable delivery timelines and the ability to maintain adequate inventory levels of critical impurity standards without excessive safety stock.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram-scale production without fundamental changes to the reaction engineering. The solvents used, such as ethanol and acetone, are environmentally benign and easily recyclable, aligning with modern green chemistry initiatives. While the use of chromium trioxide requires appropriate waste management protocols, the overall atom economy of the coupling step is favorable. The ability to scale this process ensures that as regulatory requirements for impurity identification become more stringent, the supply of necessary reference materials can expand in tandem with API production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 7-oxoacetate abiraterone. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical decision-makers evaluating this technology for adoption. Understanding these nuances is crucial for integrating this impurity standard into a comprehensive quality management system.
Q: What is the primary advantage of this synthetic method for 7-oxoacetate abiraterone?
A: The primary advantage is the ability to directionally synthesize this specific impurity with high purity (>98%), which was previously unreported in literature, enabling precise quality control for Abiraterone Acetate API manufacturing.
Q: Why is the Suzuki coupling performed as the final step in this sequence?
A: Performing the Suzuki coupling last prevents the sensitive pyridine ring from being exposed to the harsh oxidative conditions required to install the 7-oxo group, thereby preserving structural integrity and minimizing side reactions.
Q: Can this process be scaled for commercial production of reference standards?
A: Yes, the process utilizes readily available raw materials and standard unit operations such as filtration and recrystallization, making it highly suitable for scalable production of pharmaceutical reference standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Oxoacetate Abiraterone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your pharmaceutical products depends on the quality of every component, including the reference standards used to test them. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your demand for 7-oxoacetate abiraterone regardless of volume. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and purity of every batch. Our commitment to excellence ensures that the impurity standards we provide are fit for purpose, supporting your regulatory filings and quality control operations with unwavering reliability.
We invite you to collaborate with us to optimize your supply chain for Abiraterone Acetate impurities. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your operational efficiency. Let us be your partner in achieving the highest standards of pharmaceutical quality and safety.
