Advanced Catalytic Route for High-Purity p-Chlorophenethylamine Production
Introduction to Patent CN110156608B
The global pharmaceutical landscape is increasingly focused on effective treatments for metabolic disorders, with Lorcaserin standing out as a pivotal anti-obesity agent approved by the FDA. At the heart of manufacturing this critical medication lies the efficient production of its key precursor, p-chlorophenethylamine. Patent CN110156608B, published in early 2022, introduces a groundbreaking synthetic methodology that addresses longstanding safety and scalability issues associated with traditional routes. This innovation leverages a modified Ullmann-type coupling reaction, utilizing cuprous iodide and a specialized solid base system to facilitate the connection between p-halochlorobenzene and protected haloethylamine derivatives. For industry stakeholders, this represents a significant leap forward in process chemistry, offering a pathway that avoids highly toxic reagents while delivering exceptional yields exceeding 98%. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advancements is crucial for securing a competitive edge in the production of complex amine intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of p-chlorophenethylamine has been plagued by reliance on hazardous reduction systems and cumbersome purification techniques. Prior art, such as the methods described in CN18010766 and CN18010765, typically employs p-chlorobenzonitrile or p-chlorophenylacetonitrile as starting materials, requiring reduction via potassium borohydride and Raney Nickel. While these routes can achieve yields around 80%, the use of Raney Nickel introduces severe safety liabilities due to its pyrophoric nature and classification as a carcinogen. Furthermore, the resulting crude products often necessitate purification through column chromatography, a technique that is notoriously difficult to scale for industrial manufacturing and significantly drives up operational costs. The environmental burden of disposing of heavy metal catalysts and the complexity of managing exothermic hydrogenation reactions further diminish the commercial viability of these legacy processes for cost reduction in API manufacturing.
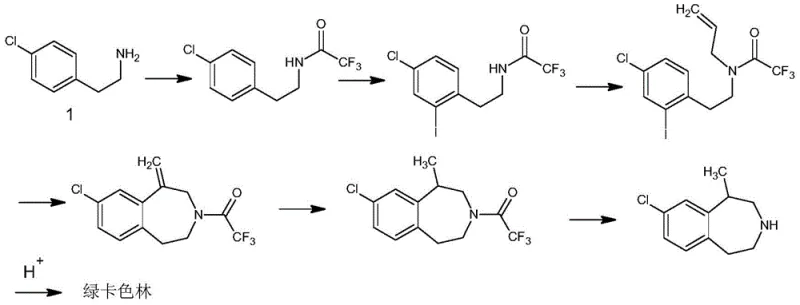
The Novel Approach
In stark contrast, the methodology disclosed in CN110156608B presents a streamlined, catalytic alternative that fundamentally reshapes the production workflow. By shifting the synthetic strategy to a direct coupling of p-halochlorobenzene with haloethylamine, the process eliminates the need for nitrile reductions entirely. The core innovation lies in the use of a nano-silicon dioxide-based solid base combined with cuprous iodide, which activates the aryl halide under controlled high-temperature and high-pressure conditions. This approach not only simplifies the reaction setup but also ensures high selectivity, thereby removing the absolute necessity for column chromatography. The result is a robust process capable of delivering high-purity p-chlorophenethylamine through standard workup procedures like extraction and distillation. This transition from batch-wise chromatographic purification to continuous flow-friendly coupling reactions exemplifies the kind of process intensification that modern chemical enterprises seek for commercial scale-up of complex pharmaceutical intermediates.
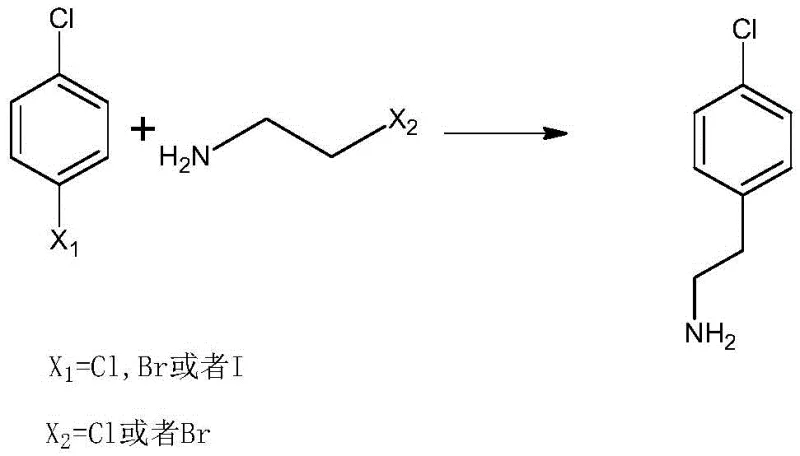
Mechanistic Insights into Cu-Catalyzed Cross-Coupling
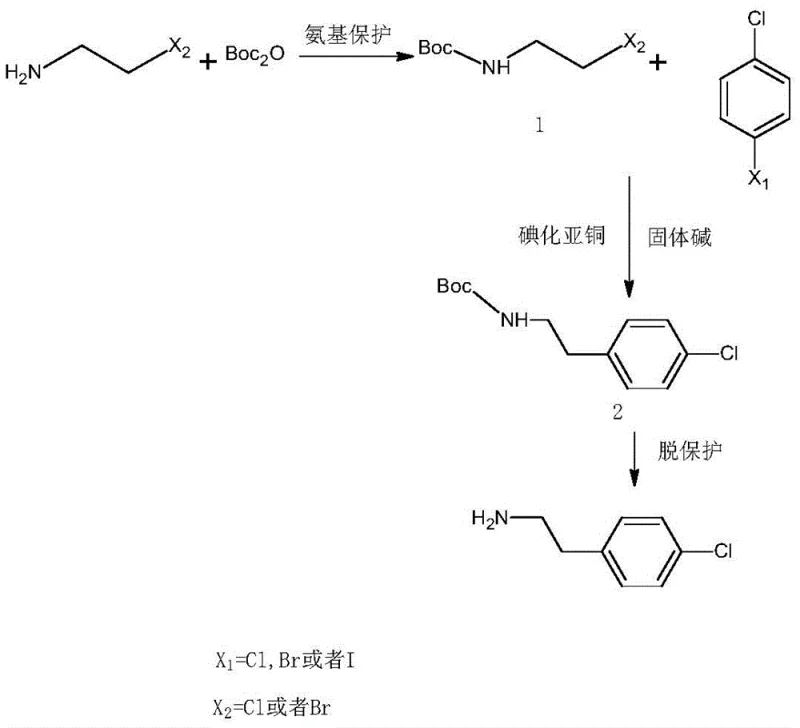
The success of this novel synthesis hinges on a sophisticated interplay between protective group chemistry and transition metal catalysis. The process initiates with the protection of the reactive amino group in haloethylamine using di-tert-butyl dicarbonate (Boc2O), forming a stable carbamate intermediate. This protection step is critical as it prevents the nucleophilic amine from interfering with the subsequent coupling reaction or undergoing self-polymerization. Once protected, the haloethylamine derivative enters the catalytic cycle where cuprous iodide acts as the central mediator. Under the influence of the solid base and elevated temperatures ranging from 130°C to 170°C, the copper catalyst facilitates the oxidative addition into the carbon-halogen bond of the p-halochlorobenzene. This generates an organocopper species that is highly reactive towards the alkyl halide moiety of the protected amine.
Following the coupling event, the reaction mixture undergoes a carefully controlled deprotection sequence using aqueous hydrochloric acid. The acidic conditions cleave the Boc group, regenerating the free amine while simultaneously forming a water-soluble ammonium salt. This phase behavior is exploited during the workup, where pH adjustment allows for the selective partitioning of the product into the organic phase, leaving inorganic salts and catalyst residues behind. The use of a nano-silicon dioxide-supported base further enhances the reaction kinetics by providing a high surface area for the base to interact with the reactants, effectively scavenging the hydrogen halide byproduct generated during the coupling. This mechanistic elegance ensures that impurity profiles remain minimal, directly contributing to the high GC purity observed in the final product without the need for extensive downstream purification.
How to Synthesize p-Chlorophenethylamine Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and safety. The protocol begins with the preparation of the protected amine in tetrahydrofuran, followed by the introduction of the aryl halide and catalyst system in a separate solvent phase like benzene or toluene. The reaction is conducted under an inert atmosphere to prevent catalyst oxidation, with pressure carefully ramped up to 5-7 atmospheres during the coupling phase to drive the equilibrium forward. Detailed operational guidelines regarding temperature ramps, addition rates, and quenching procedures are essential for reproducibility. For a comprehensive breakdown of the exact molar ratios and specific thermal profiles required to achieve the reported 98% yields, please refer to the standardized synthesis protocol below.
- Protect the amino group of haloethylamine using di-tert-butyl dicarbonate and base in THF at 50-70°C to form mixture M1.
- React p-halochlorobenzene with cuprous iodide and solid base in solvent S2 at 130-170°C under pressure, then add M1 to form mixture M2.
- Deprotect mixture M2 using hydrochloric acid at 80-90°C, adjust pH to 4, and isolate the final product via extraction and concentration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers substantial strategic benefits beyond mere technical feasibility. The elimination of Raney Nickel removes a major regulatory and safety hurdle, significantly reducing the costs associated with hazardous material handling, storage, and specialized waste disposal. Furthermore, the avoidance of column chromatography translates directly into reduced solvent consumption and shorter batch cycle times, which are critical factors for enhancing throughput in large-scale facilities. The reliance on commodity starting materials like p-dichlorobenzene and chloroethylamine ensures that the supply chain remains resilient against raw material shortages, providing a stable foundation for long-term production planning. These factors collectively contribute to a more sustainable and economically viable manufacturing model for high-volume pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing expensive and difficult-to-handle reduction catalysts with a reusable copper-based system. By eliminating the need for column chromatography, the method drastically reduces solvent usage and labor hours associated with purification. The high yield reported in the patent examples means less raw material is wasted, directly lowering the cost of goods sold (COGS) for the final intermediate. Additionally, the simplified workup procedure reduces energy consumption related to solvent recovery and drying operations.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as p-halochlorobenzenes and simple haloalkylamines mitigates the risk of supply disruptions often seen with specialized reagents. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is less sensitive to minor variations in input quality compared to sensitive hydrogenation reactions. This stability ensures consistent delivery timelines for downstream API manufacturers, reinforcing the reliability of the supply chain for critical obesity treatment medications.
- Scalability and Environmental Compliance: The transition from batch chromatography to a catalytic coupling process inherently supports scalability, allowing production volumes to be increased from pilot scale to multi-ton capacity with minimal re-engineering. The removal of toxic heavy metals like nickel from the process stream simplifies environmental compliance and wastewater treatment, aligning with increasingly stringent global green chemistry regulations. This eco-friendly profile enhances the marketability of the intermediate to environmentally conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is vital for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term value proposition of this manufacturing method.
Q: Why is this new method safer than traditional Raney Nickel reductions?
A: Traditional methods often rely on Raney Nickel, a pyrophoric and highly toxic catalyst that poses significant storage and disposal hazards. This patented process utilizes cuprous iodide and a solid base system, eliminating the need for hazardous hydrogenation catalysts and simplifying waste management.
Q: How does the process achieve high purity without column chromatography?
A: The process employs a selective Boc protection strategy and optimized reaction conditions (temperature and pressure control) that minimize byproduct formation. The final workup involves simple acid-base extraction and crystallization, achieving GC purity over 98% without the need for expensive and difficult-to-scale column chromatography.
Q: What are the key raw materials for this synthesis?
A: The primary starting materials are p-halochlorobenzene (such as p-dichlorobenzene or p-chlorobromobenzene) and haloethylamine (such as 2-chloroethylamine). These are commodity chemicals that are readily available globally, ensuring a stable supply chain for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Chlorophenethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications like Lorcaserin. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of p-chlorophenethylamine meets the highest industry standards. Our facility is equipped to handle the specific pressure and temperature requirements of this advanced catalytic process, delivering a product that is ready for immediate downstream cyclization.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to demonstrate how our optimized manufacturing capabilities can support your project goals and accelerate your time to market.
