Advanced Synthesis of Polyfluoronaphthalene Liquid Crystals for High-Performance Displays
The rapid evolution of display technology demands liquid crystal materials with increasingly sophisticated electro-optical properties, driving the need for innovative molecular architectures. Patent CN113881442B introduces a groundbreaking liquid crystal compound containing polyfluoronaphthalene, specifically designed to enhance the performance of modern display devices. This novel compound, characterized by the general structure shown in Formula (I), incorporates a unique polyfluoronaphthalene core that significantly improves molecular polarity and dielectric anisotropy. The introduction of this specific structural motif allows for a wider nematic phase temperature range and superior optical stability, addressing critical bottlenecks in high-resolution display applications. By leveraging advanced synthetic strategies, this invention provides a robust pathway to producing high-performance electronic chemicals that meet the rigorous standards of the optoelectronics industry.
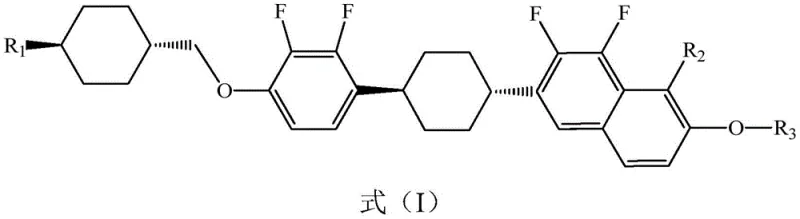
As a reliable electronic chemical supplier, understanding the structural nuances of such compounds is vital for developing next-generation mixtures. The compound features variable alkyl chains (R1, R3) and fluorine substitutions (R2) that can be tuned to optimize viscosity and clearing points. This flexibility makes it an invaluable building block for formulators seeking to balance response speed and thermal stability in IPS or VA mode liquid crystal displays. The strategic placement of fluorine atoms on the naphthalene ring not only enhances chemical stability but also contributes to a larger negative dielectric anisotropy, which is essential for reducing driving voltages in energy-efficient screens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of cyclohexyl-containing fluorobenzene liquid crystal compounds has relied heavily on sulfonylation reactions involving alkylcyclohexyl alcohol and sulfonyl chloride compounds. This conventional route presents significant challenges for industrial scale-up, primarily due to the hazardous nature of sulfonyl chlorides, which pose serious risks to both operator safety and environmental compliance. Furthermore, these traditional methods often suffer from lower product yields and generate complex impurity profiles that require extensive and costly purification processes. The reliance on high-risk solvents like petroleum ether further exacerbates safety concerns, making the manufacturing process less sustainable and more expensive. For procurement managers and supply chain heads, these factors translate into higher production costs, longer lead times, and increased regulatory burdens associated with hazardous waste disposal.
The Novel Approach
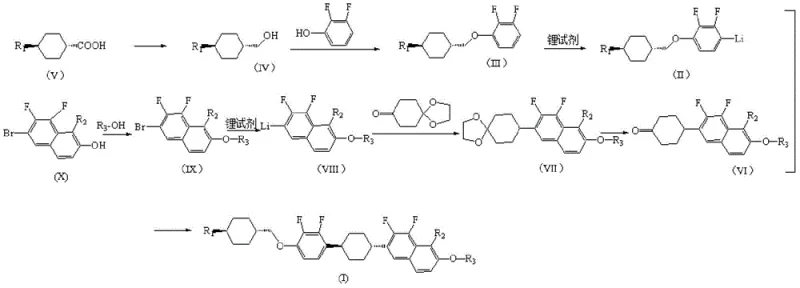
In stark contrast, the method disclosed in CN113881442B utilizes a sophisticated Mitsunobu reaction strategy to construct the critical ether linkages, completely bypassing the need for dangerous sulfonyl chloride intermediates. This novel approach involves reacting alkyl cyclohexyl methanol with 2,3-difluorophenol, and separately reacting fluorine-containing bromonaphthol with alkyl alcohol, to form the key precursors. The subsequent coupling of these fragments via nucleophilic addition and hydrogenation results in a streamlined synthesis that is both safer and more efficient. The elimination of hazardous reagents and the use of milder reaction conditions significantly reduce the potential for side reactions, leading to a cleaner crude product. This technological leap ensures that the final liquid crystal monomer can be obtained with a purity exceeding 99.5% and a yield greater than 86%, representing a substantial improvement over prior art.
Mechanistic Insights into Mitsunobu Etherification and Lithiation
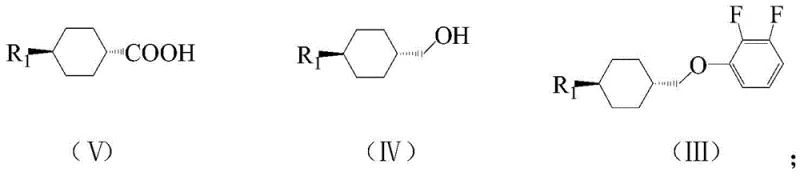
The core of this synthetic breakthrough lies in the precise execution of the Mitsunobu reaction, which facilitates the formation of the ether bond under mild conditions with inversion of configuration, although stereochemistry is managed via the trans-cyclohexyl starting materials. In the initial steps, alkyl cyclohexyl carboxylic acid is reduced to the corresponding alcohol using hydride reducing agents such as sodium bis(2-methoxyethoxy)aluminum hydride or lithium aluminum hydride at controlled temperatures between 0°C and 30°C. This alcohol is then coupled with 2,3-difluorophenol in the presence of triphenylphosphine and an azodicarboxylate, typically in toluene solvent. The mechanism proceeds through the formation of an alkoxyphosphonium salt, which is subsequently attacked by the phenol nucleophile. This step is critical for establishing the chiral integrity and connectivity of the left-hand side of the molecule, ensuring high optical purity which is paramount for liquid crystal performance.
On the right-hand side of the molecule, the synthesis employs organolithium chemistry to construct the naphthalene-cyclohexyl ketone framework. The fluorine-containing bromonaphthalene undergoes halogen-lithium exchange at cryogenic temperatures (-100°C to -60°C) to generate a reactive naphthalene lithium species. This nucleophile then attacks 1,4-cyclohexanedione monoethylene glycol ketal, forming a tertiary alcohol intermediate. Subsequent acidic hydrolysis, dehydration, and catalytic hydrogenation steps convert this intermediate into the desired ketone. The use of the ethylene glycol ketal protecting group is a strategic choice to prevent unwanted side reactions at the carbonyl position during the lithiation step. Finally, a second lithiation of the fluorobenzene fragment allows for nucleophilic addition to the naphthalene ketone, followed by dehydration and hydrogenation to yield the final saturated linkage. This multi-step sequence demonstrates exceptional control over regioselectivity and functional group tolerance.
How to Synthesize Polyfluoronaphthalene Liquid Crystal Efficiently
The synthesis of this high-value electronic chemical requires strict adherence to temperature controls and stoichiometry to maximize yield and purity. The process is divided into distinct stages: precursor preparation via reduction and Mitsunobu coupling, construction of the naphthalene core via lithiation, and final assembly through nucleophilic addition and hydrogenation. Each step has been optimized to minimize impurity formation, utilizing solvents like toluene, THF, and ethyl acetate which are amenable to recovery and recycling. For detailed operational parameters, including specific molar ratios, reaction times, and workup procedures, refer to the standardized guide below which outlines the critical process controls necessary for successful manufacturing.
- Reduce alkyl cyclohexyl carboxylic acid to alcohol, then perform Mitsunobu reaction with 2,3-difluorophenol to form alkyl cyclohexyl methoxy fluorobenzene.
- React fluorine-containing bromonaphthol with alkyl alcohol via Mitsunobu reaction, followed by lithiation and nucleophilic addition with cyclohexanedione ketal.
- Perform hydrolysis, dehydration, hydrogenation, and deprotection to obtain the final naphthalene cyclohexyl ketone, then couple with the fluorobenzene derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. By eliminating the use of sulfonyl chloride compounds, manufacturers can drastically reduce the costs associated with hazardous material handling, storage, and waste treatment. The simplified purification process, which relies on crystallization and standard column chromatography rather than complex distillation or specialized scavenging resins, further lowers the operational expenditure per kilogram of product. Additionally, the high purity achieved directly from the synthesis reduces the need for extensive reprocessing, thereby shortening the overall production cycle time and enhancing throughput capacity.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous sulfonyl chloride reagents with more accessible alcohols and phenols leads to significant raw material cost savings. Furthermore, the high reaction yields (>86%) and excellent purity (>99.5%) minimize material loss and reduce the burden on downstream purification units. This efficiency translates into a lower cost of goods sold (COGS), allowing for more competitive pricing in the global electronic chemical market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as alkyl cyclohexyl carboxylic acids and simple fluorophenols, are commercially available and stable, reducing the risk of supply disruptions. The robustness of the Mitsunobu reaction and the subsequent lithiation steps ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with display panel manufacturers. By mitigating the risks associated with hazardous reagents, facilities can maintain continuous operation with fewer safety-related shutdowns or regulatory delays.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing common organic solvents and standard reactor equipment capable of handling cryogenic and exothermic reactions safely. The absence of heavy metal catalysts in the coupling steps and the use of recyclable solvents align with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. This sustainability profile not only future-proofs the supply chain but also enhances the brand value of the final display products by associating them with responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this polyfluoronaphthalene liquid crystal compound. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance metrics and process feasibility. Understanding these details is essential for R&D teams evaluating this material for new formulations and for procurement specialists assessing its viability for long-term sourcing strategies.
Q: What are the purity levels achievable with this new synthesis method?
A: The novel preparation method described in patent CN113881442B achieves product purity exceeding 99.5%, significantly higher than traditional methods, ensuring superior performance in liquid crystal displays.
Q: How does this process improve safety compared to conventional routes?
A: This method eliminates the use of hazardous sulfonyl chloride compounds and high-risk petroleum ether solvents, replacing them with safer Mitsunobu reaction conditions and environmentally friendly solvents like toluene and THF.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the process features mild reaction conditions, high yields (>86%), and simplified purification steps, making it highly scalable and cost-effective for commercial manufacturing of electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyfluoronaphthalene Liquid Crystal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the advancement of display technologies. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped to handle the sensitive organolithium and hydrogenation steps required for this synthesis, adhering to stringent purity specifications and rigorous QC labs to guarantee product consistency. We are committed to delivering electronic chemicals that empower the next generation of high-performance displays.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a stable, high-quality supply of advanced liquid crystal materials.
