Revolutionizing Sulfenamide Production: A Metal-Free, Scalable Route for High-Purity Pharmaceutical Intermediates
The landscape of organic synthesis is constantly evolving, driven by the need for greener, more efficient, and scalable processes. A significant breakthrough in this domain is documented in Chinese Patent CN116143722A, which discloses a novel synthesis technique for sulfenamide compounds. Sulfenamides, characterized by their unique sulfur-nitrogen bonds, are pivotal intermediates in medicinal chemistry, agrochemicals, and industrial applications such as rubber vulcanization accelerators. Traditional methods for constructing the S-N bond have often been plagued by harsh conditions, limited substrate scope, or the reliance on expensive and toxic heavy metal catalysts. This new technology addresses these critical pain points by introducing a mild, metal-free oxidative coupling strategy using dimethylsulfur bromide. For R&D directors and procurement managers alike, this represents a paradigm shift towards more sustainable and cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of sulfenamides relied on several distinct methodologies, each carrying significant drawbacks that hindered large-scale industrial adoption. One common approach utilized TEMPO as a catalyst with oxygen as the oxidant; while green in principle, this method was severely restricted to unsubstituted 2-mercaptobenzothiazole substrates, failing to accommodate the diverse structural requirements of modern drug discovery. Another prevalent method employed iodine catalysis in polyethylene glycol with hydrogen peroxide; however, this route generated corrosive hydroiodic acid as a byproduct, posing serious safety hazards and equipment corrosion risks during scale-up. Furthermore, electrochemical synthesis methods, though avoiding chemical oxidants, exhibited extreme sensitivity to the steric hindrance of amine substrates, resulting in prohibitively low yields for bulky amines. Finally, copper-catalyzed systems required prolonged reaction times of up to 36 hours and necessitated rigorous post-reaction heavy metal removal steps to meet stringent pharmaceutical purity standards.
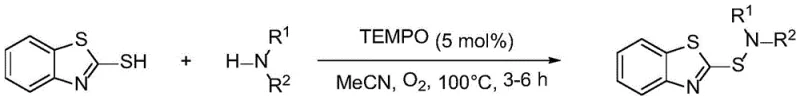
The Novel Approach
In stark contrast to these legacy techniques, the method described in CN116143722A leverages dimethylsulfur bromide as a highly efficient organic sulfur reagent to drive the oxidative coupling of thiols and amines. This approach operates under remarkably mild conditions, typically between -20°C and 20°C, eliminating the thermal stress associated with high-temperature protocols. By utilizing commercially available and air-insensitive reagents, the process ensures robust supply chain continuity and reduces raw material costs significantly. The reaction proceeds smoothly in a single organic solvent system, preferably 1,2-dichloroethane (DCE), without the need for specialized electrolytic cells or toxic transition metals. This simplicity not only streamlines the operational workflow but also drastically reduces the environmental footprint by avoiding the generation of heavy metal waste streams, making it an ideal candidate for reliable pharmaceutical intermediates supplier operations aiming for green chemistry compliance.
Mechanistic Insights into Dimethylsulfur Bromide Mediated Oxidative Coupling
The core of this technological advancement lies in the unique reactivity of dimethylsulfur bromide (DMSBr) as an electrophilic sulfur transfer agent. In the proposed mechanism, the mercapto compound (thiol) initially interacts with the DMSBr, where the sulfur atom of the thiol acts as a nucleophile attacking the electrophilic sulfur center of the reagent. This interaction facilitates the formation of a highly reactive sulfenyl bromide intermediate in situ, releasing dimethyl sulfide as a volatile byproduct that can be easily removed. The presence of a base, such as cesium carbonate, is critical in this stage to scavenge the generated hydrogen bromide, thereby driving the equilibrium forward and preventing the protonation of the amine nucleophile. This activation step is far more efficient than radical-based oxidations, as it avoids the formation of uncontrolled radical species that often lead to disulfide byproducts.
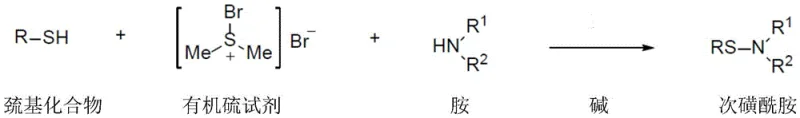
Following the activation of the thiol, the amine component performs a nucleophilic attack on the activated sulfur species to form the desired S-N bond. The beauty of this mechanism is its tolerance to steric bulk; unlike electrochemical methods where electron transfer is hindered by spatial crowding, this chemical pathway relies on direct nucleophilic substitution which remains effective even with hindered amines like 2,2,6,6-tetramethylpiperidine. Furthermore, the choice of base plays a pivotal role in impurity control. The patent data indicates that inorganic bases like cesium carbonate provide superior yields compared to organic bases like pyridine, likely due to better solubility profiles and more effective neutralization of acidic byproducts. This mechanistic clarity allows process chemists to fine-tune reaction parameters, ensuring that the final high-purity sulfenamides meet the rigorous specifications required for API intermediate manufacturing without the need for complex chromatographic separations.
How to Synthesize Sulfenamides Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize efficiency and yield. The process begins with the preparation of the reaction vessel under an inert nitrogen atmosphere to prevent moisture interference. The thiol and dimethylsulfur bromide are dissolved in the solvent at low temperature to control the exotherm upon mixing. Subsequently, the base is added to activate the system before the controlled addition of the amine. Detailed standardized synthetic steps, including precise molar ratios, addition rates, and workup procedures, are outlined in the technical guide below to ensure reproducibility and safety during operation.
- Prepare the reaction mixture by dissolving the mercapto compound and dimethylsulfur bromide in dichloroethane (DCE) at 0°C under nitrogen protection.
- Add the base, preferably cesium carbonate, to the mixture and stir to activate the sulfur species.
- Slowly add the amine compound dropwise over 30 minutes, allow the reaction to warm to room temperature, and stir for 14 hours before extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis protocol offers tangible strategic benefits beyond mere technical feasibility. The elimination of transition metal catalysts such as copper or iodine removes the necessity for expensive and time-consuming metal scavenging resins or complex aqueous washes, directly translating to substantial cost savings in manufacturing. Additionally, the reliance on commercially available, shelf-stable reagents like dimethylsulfur bromide mitigates the risk of supply chain disruptions often associated with specialized catalysts or gases. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a reduced overall carbon footprint and aligning with corporate sustainability goals. These factors collectively enhance the reliability of the supply chain for complex organic intermediates.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily driven by the simplification of the downstream processing workflow. By avoiding heavy metal catalysts, manufacturers eliminate the capital expenditure and operational costs associated with metal removal technologies, which are mandatory for pharmaceutical grade products. Furthermore, the high atom economy and the use of inexpensive inorganic bases like cesium carbonate reduce the raw material cost per kilogram of the final product. The ability to run the reaction in a single solvent system without the need for phase transfer catalysts or exotic ligands further streamlines the bill of materials, ensuring that cost reduction in fine chemical manufacturing is achieved through fundamental process design rather than temporary market fluctuations.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, commodity-grade chemicals that are widely sourced globally. Unlike methods dependent on electrochemical cells which require specialized equipment maintenance and electrode replacement, or TEMPO which can be cost-prohibitive at scale, the reagents here are standard organic building blocks. The insensitivity of the reaction to air during the reagent handling phase (prior to nitrogen protection for the reaction itself) simplifies logistics and storage requirements. This stability ensures that production schedules are not derailed by reagent degradation or availability issues, providing a consistent flow of high-purity sulfenamides to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden safety and environmental liabilities, but this method is inherently designed for industrial promotion. The absence of corrosive byproducts like hydroiodic acid protects reactor integrity and reduces waste treatment costs. Moreover, the mild temperature profile (-20°C to 20°C) reduces the load on plant utilities compared to high-temperature reflux conditions. The process generates minimal hazardous waste, facilitating easier compliance with increasingly strict environmental regulations. This ease of commercial scale-up of complex organic intermediates allows manufacturers to respond rapidly to market demand without the lengthy validation periods typically required for more hazardous chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this sulfenamide synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying the operational boundaries and advantages for potential adopters. Understanding these nuances is crucial for process development teams evaluating this route against existing legacy methods.
Q: Does this synthesis method require heavy metal catalysts?
A: No, unlike conventional methods using Copper(I) Iodide or electrochemical cells, this process utilizes dimethylsulfur bromide as a reagent, completely eliminating the need for transition metal catalysts and simplifying downstream purification.
Q: What is the substrate scope for the amine component?
A: The method demonstrates excellent compatibility with a wide range of amines, including aliphatic primary amines, secondary amines, heterocyclic amines, and sterically hindered amines, overcoming the limitations of previous electrolytic methods.
Q: What are the optimal reaction conditions for maximum yield?
A: Optimal results are achieved using a molar ratio of thiol to DMSBr to amine to base of 1:1:3:1.5, with cesium carbonate as the base in DCE solvent at 0°C warming to room temperature over 14 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfenamides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to maintain competitiveness in the global fine chemicals market. Our team of expert process chemists has thoroughly evaluated the methodology described in CN116143722A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering sulfenamide intermediates with stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets the highest international standards. Our infrastructure is uniquely positioned to leverage this metal-free technology, offering clients a greener and more economical alternative to traditional manufacturing routes.
We invite you to collaborate with us to optimize your supply chain for sulfenamide derivatives. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments for your target molecules, ensuring that your project moves forward with the most efficient and reliable chemistry available.
