Advanced Synthesis of Push-Pull Imidazole Ionic Salts for Next-Generation Nonlinear Optical Applications
The landscape of nonlinear optical (NLO) materials is undergoing a significant transformation driven by the demand for faster information processing and advanced laser technologies. Patent CN101143848A introduces a groundbreaking class of imidazole ionic salts characterized by a unique push-pull electronic structure, addressing the longstanding limitations of traditional organic NLO crystals. These novel compounds combine the high nonlinear optical coefficients typical of organic molecules with the superior thermal stability usually associated with inorganic salts. For R&D directors and procurement specialists in the electronic chemicals sector, this technology represents a pivotal shift towards more robust and processable materials for frequency conversion and optical switching applications. The synthesis strategy outlined in the patent leverages straightforward quaternization reactions followed by tunable anion exchange, offering a scalable pathway to high-performance optoelectronic components.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic nonlinear optical materials has been hindered by a critical trade-off between optical performance and thermal stability. Traditional organic crystals, while possessing excellent nonlinear coefficients often one to two orders of magnitude higher than inorganic counterparts, frequently degrade at relatively low temperatures, limiting their utility in high-power laser systems or harsh processing environments. Furthermore, the crystallization and processing of these conventional organic materials can be notoriously difficult, often requiring complex purification techniques that drive up manufacturing costs and reduce overall yield. The inherent instability of the molecular arrangement in many standard organic NLO materials also leads to issues with long-term reliability and consistency in device performance, creating significant supply chain risks for manufacturers of advanced optical devices.
The Novel Approach
The methodology described in CN101143848A circumvents these challenges by constructing an ionic lattice architecture that inherently stabilizes the push-pull chromophore. By reacting an electron-donating aryl N-substituted imidazole with an electron-withdrawing halogenated aromatic hydrocarbon, the process generates a cationic imidazolium core paired with a stable anion. This ionic nature significantly elevates the thermal decomposition temperature, with specific embodiments demonstrating stability well above 300°C, thereby expanding the operational window for these materials. The approach not only enhances thermal resilience but also simplifies the isolation process, as the ionic products often precipitate directly from the reaction mixture, allowing for purification via simple filtration rather than energy-intensive distillation or chromatography.
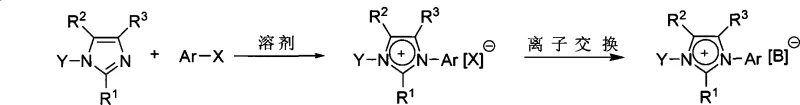
Mechanistic Insights into Quaternization and Anion Exchange
The core chemical transformation relies on a nucleophilic substitution mechanism where the nitrogen atom of the N-substituted imidazole acts as a nucleophile attacking the electron-deficient aromatic ring of the halogenated substrate. This quaternization step creates a permanent positive charge on the imidazole ring, establishing the necessary electrostatic environment for the push-pull electron distribution. The electron-donating group on one nitrogen and the electron-withdrawing group on the other create a strong dipole moment across the conjugated system, which is the fundamental driver of the second-order nonlinear optical response. The efficiency of this reaction is highly dependent on the electronic nature of the substituents, with strong donors like methoxy or dimethylamino groups and strong acceptors like nitro groups maximizing the hyperpolarizability of the resulting ionic salt.
Beyond the initial salt formation, the patent highlights a critical secondary mechanism: anion exchange. This step allows for the fine-tuning of the material's physical properties without altering the optically active cation. By dissolving the initial chloride salt in a mixed solvent system of water and acetone, and introducing a salt containing a larger, more diffuse anion such as hexafluorophosphate (PF6-) or bis(trifluoromethanesulfonyl)imide (NTf2-), a metathesis reaction occurs. The low solubility of the new ionic pair in the aqueous medium drives the equilibrium towards the product, facilitating high-yield isolation. This modularity is essential for tailoring the solubility, melting point, and crystallization behavior of the final NLO material to specific device fabrication requirements.
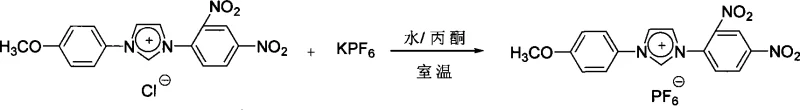
How to Synthesize Imidazole Ionic Salts Efficiently
The synthesis protocol detailed in the patent offers a robust framework for producing these high-value electronic chemicals with minimal operational complexity. The process begins with the selection of appropriate starting materials, specifically an N-aryl imidazole bearing an electron-donating substituent and a halogenated aromatic compound featuring strong electron-withdrawing groups. The reaction is typically conducted in polar aprotic solvents such as acetonitrile or N,N-dimethylformamide at elevated temperatures ranging from 100°C to 150°C. Following the quaternization, the crude product is isolated through filtration and washed with non-polar solvents to remove unreacted starting materials. For applications requiring specific anionic counterparts, the subsequent ion exchange step is performed at ambient temperature in water-acetone mixtures, ensuring energy efficiency and ease of scale-up. Detailed standardized synthesis steps are provided in the guide below.
- React an electron-donating aryl N-substituted imidazole with an electron-withdrawing halogenated aromatic hydrocarbon in an organic solvent like acetonitrile at 40-180°C.
- Isolate the resulting imidazolium chloride salt via filtration and washing with organic solvents such as diethyl ether.
- Optionally perform an anion exchange reaction by dissolving the chloride salt in a water/acetone mixture and adding a salt containing the desired anion (e.g., KPF6 or LiNTf2).
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this ionic salt technology offers substantial strategic benefits over legacy NLO material sourcing. The simplified workup procedure, which relies primarily on filtration and washing rather than complex chromatographic separation, drastically reduces the consumption of silica gel and elution solvents. This reduction in consumable usage translates directly into lower variable costs per kilogram of production. Additionally, the high thermal stability of the final products minimizes the risk of degradation during storage and transportation, reducing waste and ensuring consistent quality upon delivery to downstream device manufacturers. The ability to utilize common, commodity-grade solvents like acetonitrile and acetone further insulates the production process from volatility in specialty solvent markets.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reliance on simple thermal quaternization removes the need for expensive metal scavenging steps and rigorous heavy metal testing, which are often cost-prohibitive bottlenecks in fine chemical manufacturing. The high yields reported in the patent examples, often exceeding 80-90%, ensure that raw material utilization is maximized, significantly lowering the cost of goods sold. Furthermore, the direct precipitation of the product simplifies the isolation phase, reducing labor hours and equipment occupancy time compared to multi-step extraction processes.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, such as substituted imidazoles and nitro-halobenzenes, are widely available commodity chemicals with established global supply chains. This abundance mitigates the risk of raw material shortages that often plague specialized synthetic routes relying on exotic reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent qualities, ensures consistent production output even when minor variations in feedstock quality occur, thereby guaranteeing reliable delivery schedules for long-term contracts.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton commercial production without requiring fundamental changes to the reaction engineering. The use of recyclable solvents and the generation of benign inorganic salt byproducts during the anion exchange step align with increasingly stringent environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and avoids potential fines or shutdowns associated with hazardous waste disposal, ensuring uninterrupted supply continuity for our partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this imidazole ionic salt technology in industrial settings. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating the material for specific optical applications and for procurement officers assessing the total cost of ownership.
Q: What are the primary advantages of these imidazole ionic salts over traditional organic NLO crystals?
A: Unlike traditional organic crystals which often suffer from low thermal stability and poor processability, these push-pull imidazole ionic salts exhibit significantly enhanced thermal decomposition temperatures (often exceeding 300°C) while maintaining large nonlinear optical coefficients.
Q: Can the anion of the ionic salt be modified for specific application requirements?
A: Yes, the patent describes a versatile anion exchange protocol where the initial chloride anion can be swapped for various counterions such as PF6-, NTf2-, or BF4- using simple metathesis reactions in mixed solvent systems.
Q: What is the typical purity profile achievable with this synthesis method?
A: The synthesis method allows for high purity isolation through simple filtration and washing steps, avoiding complex chromatographic purification, which is critical for electronic grade materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazole Ionic Salts Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercially viable electronic chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory curiosity to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the structural integrity and optical performance of every batch. Our commitment to quality assurance means that every shipment of imidazole ionic salts meets the exacting standards required for high-performance nonlinear optical devices.
We invite you to collaborate with us to optimize your supply chain for next-generation optical materials. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the display and optoelectronic sectors.
