Advanced Synthesis of 9-Chloroquetiapine: Technical Insights for Commercial Scale-Up
Advanced Synthesis of 9-Chloroquetiapine: Technical Insights for Commercial Scale-Up
The pharmaceutical industry faces increasing regulatory pressure to fully characterize impurity profiles for active pharmaceutical ingredients (APIs), particularly for CNS drugs like Quetiapine. Patent CN114380769A introduces a pivotal breakthrough in the preparation of 9-chloroquetiapine, a critical impurity that poses significant safety concerns due to its enhanced lipophilicity and ability to cross the blood-brain barrier. This document details a robust, one-step synthetic route that transforms readily available Quetiapine into the target chloro-derivative using mild chlorinating agents. For R&D directors and procurement specialists, understanding this methodology is essential for establishing rigorous quality control standards and securing a reliable supply of reference materials. The innovation lies not just in the chemical transformation itself, but in the strategic simplification of the supply chain for high-purity analytical standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the disclosure of patent CN114380769A, the synthesis of specific chlorinated impurities of Quetiapine was notably absent from scientific literature, creating a significant bottleneck for quality assurance teams. Traditional approaches to generating such impurities often involved complex multi-step syntheses starting from basic raw materials, which introduced numerous opportunities for side reactions and difficult-to-remove by-products. Furthermore, attempting to isolate these impurities directly from crude API batches is inefficient and yields inconsistent results, making it nearly impossible to obtain sufficient quantities for validation studies. The lack of a defined synthetic pathway meant that pharmaceutical manufacturers struggled to accurately quantify this specific safety risk, potentially compromising patient safety assessments and regulatory filings.
The Novel Approach
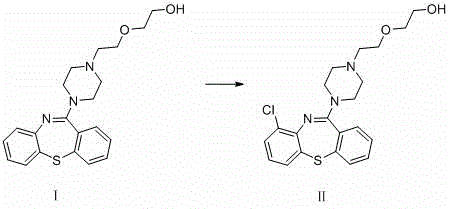
The patented method revolutionizes this landscape by utilizing Quetiapine itself as the starting substrate, effectively bypassing the need for de novo synthesis of the complex dibenzothiazepine core. By employing electrophilic chlorinating reagents such as N-chlorosuccinimide (NCS) or trichloroisocyanuric acid, the process achieves direct functionalization at the 9-position of the aromatic ring in a single operational step. This direct approach drastically reduces the number of unit operations required, minimizing solvent consumption and waste generation while maximizing overall throughput. The simplicity of the reaction conditions, which operate at moderate temperatures between 40-80°C, ensures that the process is both energy-efficient and easily adaptable to various reactor configurations.
Mechanistic Insights into Electrophilic Aromatic Substitution
The core chemical transformation relies on a classic electrophilic aromatic substitution mechanism, where the electron-rich dibenzothiazepine system of Quetiapine acts as the nucleophile. The chlorinating agent, activated by the polar organic solvent, generates a positive chlorine species that selectively attacks the most electron-dense position on the aromatic ring. In the case of Quetiapine, the position ortho to the sulfur and nitrogen bridgeheads (position 9) exhibits heightened reactivity due to resonance stabilization effects provided by the heteroatoms. This inherent electronic bias ensures high regioselectivity, preventing the formation of unwanted isomers that would complicate downstream purification efforts and compromise the utility of the reference standard.
Controlling the stoichiometry and reaction temperature is paramount to preventing over-chlorination, which could lead to di- or tri-chlorinated by-products. The patent specifies a molar ratio range of 1:1 to 1:3 between the substrate and the chlorinating agent, allowing fine-tuning of the reaction kinetics to favor mono-substitution. Additionally, the choice of solvent plays a critical role in stabilizing the transition state and solubilizing the intermediates; acetonitrile and chloroform are highlighted for their ability to support the reaction without participating in side reactions. This precise control over the reaction environment is what enables the production of high-purity 9-chloroquetiapine suitable for sensitive analytical applications.
How to Synthesize 9-Chloroquetiapine Efficiently
Implementing this synthesis requires careful attention to reaction parameters to ensure reproducibility and safety. The process begins with the dissolution of the Quetiapine substrate in a dry, aprotic solvent under an inert atmosphere to prevent moisture interference. Following the addition of the chlorinating agent, the mixture is heated to the specified range and monitored until conversion is complete. While the general concept is straightforward, the specific details regarding workup and purification are critical for achieving the necessary purity levels for analytical use.
- Dissolve Quetiapine (Compound I) in a suitable organic solvent such as acetonitrile or chloroform under an inert nitrogen atmosphere.
- Slowly add the chlorinating reagent (NCS or TCCA) dropwise to the reaction mixture while maintaining controlled temperature conditions between 40-80°C.
- Stir the reaction for 12-36 hours, followed by aqueous workup, extraction with DCM, drying, and purification via column chromatography to isolate Compound II.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits beyond mere technical feasibility. The reliance on commercially available starting materials and reagents eliminates the need for custom synthesis of exotic precursors, thereby reducing lead times and mitigating supply risk. The simplified workflow translates directly into lower operational expenditures, as fewer processing steps mean reduced labor costs, lower energy consumption, and decreased solvent usage. These factors collectively contribute to a more resilient and cost-effective supply chain for critical pharmaceutical reference standards.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences significantly lowers the cost of goods sold by reducing material loss and processing time. By avoiding the need for specialized catalysts or extreme reaction conditions, the process utilizes standard equipment found in most fine chemical facilities, further driving down capital expenditure requirements. This efficiency allows suppliers to offer competitive pricing for high-purity impurities without compromising on quality margins.
- Enhanced Supply Chain Reliability: Sourcing strategies are strengthened by the use of commodity chemicals like acetonitrile and NCS, which are widely available from multiple global vendors. This diversification of the supply base prevents bottlenecks that often occur when relying on single-source specialty reagents. Consequently, manufacturers can maintain consistent inventory levels and meet tight delivery schedules even during periods of market volatility.
- Scalability and Environmental Compliance: The mild reaction conditions and straightforward workup procedures make this process highly scalable from gram-scale laboratory synthesis to multi-kilogram production runs. Furthermore, the reduced solvent intensity and absence of heavy metal catalysts simplify waste treatment protocols, ensuring compliance with increasingly stringent environmental regulations. This alignment with green chemistry principles enhances the long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 9-chloroquetiapine. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this material into their quality control workflows.
Q: Why is the synthesis of 9-chloroquetiapine critical for pharmaceutical quality control?
A: During the synthesis of Quetiapine, chlorinated by-products can form and persist through to the final API. Since chlorination increases lipid solubility, this impurity can cross the blood-brain barrier more easily than the parent drug, posing potential safety risks. Therefore, having a pure reference standard is essential for accurate quantification and safety assessment.
Q: What are the advantages of using N-chlorosuccinimide (NCS) over other chlorinating agents?
A: NCS offers a milder and more controllable source of electrophilic chlorine compared to harsher gaseous reagents. It allows for better regioselectivity at the 9-position of the dibenzothiazepine ring and simplifies the workup procedure, as the succinimide by-product is generally easier to separate than inorganic salts generated by other methods.
Q: Can this synthesis method be scaled for industrial production of reference standards?
A: Yes, the process described in patent CN114380769A utilizes common organic solvents like acetonitrile and chloroform and operates at moderate temperatures (40-80°C). These conditions are highly amenable to scale-up in standard stainless steel reactors without requiring specialized high-pressure or cryogenic equipment, ensuring robust supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Chloroquetiapine Supplier
As the demand for comprehensive impurity profiling grows, partnering with a CDMO that possesses deep technical expertise is crucial for success. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver high-quality intermediates and reference standards. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of 9-chloroquetiapine meets the exacting standards required for regulatory submissions and stability studies.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize their supply chains for critical impurities. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your drug development goals efficiently and reliably.
