Scalable Asymmetric Synthesis of (S)-ar-Turmerone via Cobalt Catalysis for Industrial Applications
Scalable Asymmetric Synthesis of (S)-ar-Turmerone via Cobalt Catalysis for Industrial Applications
The global demand for high-purity natural product derivatives continues to surge across the pharmaceutical and flavor industries, driving the need for robust synthetic methodologies. A pivotal advancement in this domain is detailed in patent CN103787855A, which discloses a novel method for the asymmetric synthesis of (S)-ar-Turmerone. This compound, a bioactive constituent of Curcuma species, has garnered significant attention for its potential cytotoxic and anti-inflammatory properties. The disclosed technology leverages a sophisticated cobalt-catalyzed asymmetric Kumada cross-coupling reaction to establish the critical chiral center with high fidelity. By utilizing racemic 2-bromopropionate as a cost-effective starting material, this route circumvents the traditional reliance on expensive chiral pool precursors. The process delivers the target molecule with an impressive overall yield of 31% and an optical purity of 92% ee, representing a substantial leap forward in process efficiency.
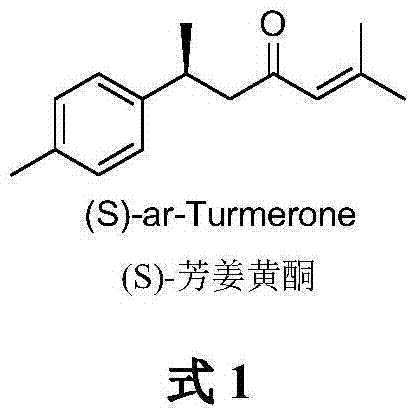
For procurement managers and supply chain directors, the implications of this technology are profound. The ability to synthesize complex chiral molecules from simple, achiral, or racemic feedstocks fundamentally alters the cost structure of production. Traditional extraction from plant sources is often plagued by seasonal variability, low concentration in biomass, and arduous purification processes. In contrast, this chemical synthesis offers a reliable, year-round supply chain that is decoupled from agricultural constraints. Furthermore, the streamlined nature of the synthetic route, comprising fewer operational units than previous methods, suggests a significant reduction in manufacturing overheads. This positions the technology as a cornerstone for any organization seeking a reliable flavor & fragrance intermediate supplier capable of delivering consistent quality at competitive price points.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (S)-ar-Turmerone has been hindered by significant technical and economic bottlenecks inherent to conventional strategies. Early approaches relied heavily on the chiral source method, necessitating the use of enantiomerically pure starting materials derived from the chiral pool, such as specific amino acids or sugars. These materials are not only expensive but also subject to market volatility and supply shortages. Additionally, methods involving chiral induction often require stoichiometric amounts of chiral auxiliaries, which must be attached and subsequently removed, adding at least two extra synthetic steps and generating substantial chemical waste. Enzymatic methods, while green in principle, often suffer from narrow substrate specificity and slow reaction rates, making them difficult to implement in high-throughput industrial settings. Consequently, these legacy routes fail to meet the rigorous demands of modern cost reduction in fine chemical manufacturing, as they inflate both the cost of goods sold (COGS) and the environmental footprint of the process.
The Novel Approach
The methodology presented in the patent data introduces a paradigm shift by employing a transition metal-catalyzed asymmetric transformation. The core innovation lies in the use of a bisoxazoline/cobalt catalyst system to mediate the cross-coupling between a racemic electrophile and a Grignard reagent. This dynamic kinetic resolution-like process effectively converts a 50:50 mixture of enantiomers into a single, highly enriched chiral product. Following the establishment of chirality, the synthesis proceeds through a logical sequence of functional group interconversions: reduction, sulfonylation, cyanation, and a second reduction to install the aldehyde moiety. The final carbon-carbon bond formation is achieved via Grignard addition followed by oxidation. This linear, convergent strategy minimizes the number of isolation steps and maximizes atom economy. For stakeholders focused on the commercial scale-up of complex pharmaceutical intermediates, this approach offers a clear pathway to multi-ton production without the complexity of biocatalytic fermentation or the expense of chiral chromatography.
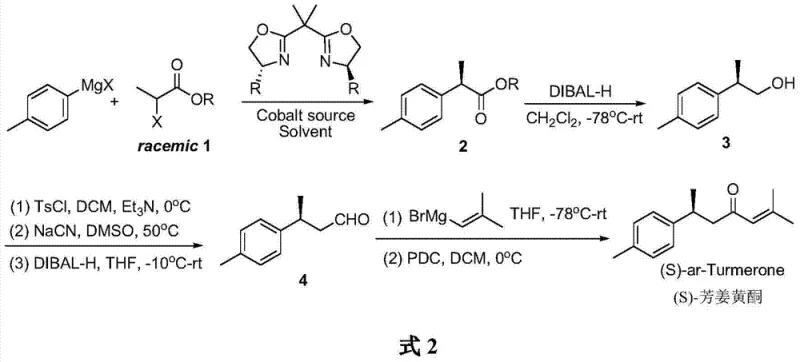
Mechanistic Insights into Cobalt-Catalyzed Asymmetric Kumada Coupling
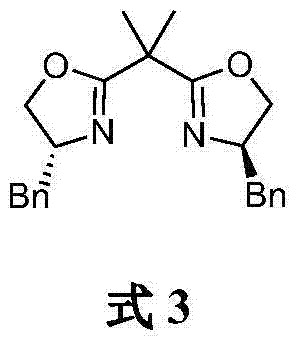
The heart of this synthetic success is the stereoselective control exerted by the chiral cobalt complex during the initial coupling step. The catalyst system utilizes a specific bisoxazoline ligand, the structure of which is critical for inducing asymmetry. In the catalytic cycle, the cobalt center coordinates with the chiral ligand to create a rigid, chiral environment around the metal. When the racemic 2-bromopropionate interacts with this complex, the catalyst differentiates between the two enantiomers of the substrate, preferentially facilitating the oxidative addition and subsequent transmetallation with the p-tolyl Grignard reagent for one specific enantiomeric pathway. This results in the formation of the (R)-configured ester intermediate with high enantiomeric excess. The steric bulk and electronic properties of the benzyl groups on the oxazoline rings play a pivotal role in shielding one face of the reactive intermediate, thereby enforcing the observed stereochemistry. Understanding this mechanism is vital for R&D directors aiming to optimize reaction parameters such as temperature and solvent polarity to further enhance the 92% ee reported in the examples.
Beyond the initial coupling, the subsequent transformations are designed to preserve the newly formed chiral center while extending the carbon skeleton. The reduction of the ester to the alcohol using DIBAL-H is performed at cryogenic temperatures (-78°C) to prevent racemization and ensure chemoselectivity. The conversion of the alcohol to the nitrile via the tosylate intermediate is a classic SN2 displacement, which proceeds with inversion of configuration; however, since the chirality is at the beta-position relative to the leaving group in the nitrile formation step (actually alpha to the original center, requiring careful tracking), the process is managed to maintain optical integrity. The final oxidation using Pyridinium Dichromate (PDC) is a mild method chosen to avoid over-oxidation or degradation of the sensitive alkene moiety introduced in the final step. This meticulous control over reaction conditions ensures that the impurity profile remains clean, a critical factor for meeting the stringent specifications required by regulatory bodies for high-purity API intermediates.
How to Synthesize (S)-ar-Turmerone Efficiently
The execution of this synthesis requires strict adherence to anhydrous conditions and precise temperature control, particularly during the organometallic steps. The process begins with the preparation of the active cobalt catalyst in situ, followed by the slow addition of the Grignard reagent to manage exotherms. Subsequent steps involve standard workup procedures including aqueous quenching, extraction, and silica gel chromatography for purification. While the laboratory examples demonstrate the feasibility on a gram scale, the transition to pilot plant operations necessitates careful engineering of the mixing and heat transfer systems to handle the reactivity of DIBAL-H and Grignard reagents safely. The detailed standardized operating procedures for each unit operation, including specific molar ratios and quenching protocols, are essential for reproducibility. For a comprehensive breakdown of the exact experimental conditions and stoichiometry required for successful replication, please refer to the technical guide below.
- Perform asymmetric Kumada cross-coupling of racemic 2-bromopropionate with p-tolyl Grignard reagent using a bisoxazoline/cobalt catalyst system to generate chiral ester 2.
- Reduce ester 2 to alcohol 3 using DIBAL-H, followed by sulfonylation and cyanation to extend the carbon chain.
- Reduce the nitrile to aldehyde 4, react with isobutenyl magnesium bromide, and oxidize the resulting alcohol with PDC to yield (S)-ar-Turmerone.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented route offers compelling advantages that directly address the pain points of modern supply chain management. The primary driver of value is the substitution of scarce, high-cost chiral starting materials with abundant, commodity-grade racemic esters. This shift drastically reduces the raw material bill of materials (BOM), allowing for more aggressive pricing strategies in a competitive market. Furthermore, the elimination of enzymatic steps or complex chiral resolution processes simplifies the manufacturing workflow, reducing the requirement for specialized equipment and highly skilled labor. This operational simplicity translates into enhanced supply chain reliability, as the risk of batch failure due to biological variability or resolution inefficiencies is virtually eliminated. Companies adopting this technology can secure a more stable supply of high-purity flavor & fragrance intermediates, insulating themselves from the volatility of agricultural harvests.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the fundamental chemistry rather than marginal efficiencies. By utilizing a catalytic asymmetric method, the need for stoichiometric chiral reagents is removed, which represents a direct saving in reagent costs. Additionally, the overall yield of 31% across the entire sequence is respectable for a multi-step asymmetric synthesis, minimizing the loss of valuable intermediates. The use of common solvents like THF, DCM, and hexane further ensures that solvent recovery and recycling can be implemented easily, reducing waste disposal costs. Qualitatively, the removal of expensive transition metals like palladium (often used in alternative cross-couplings) in favor of earth-abundant cobalt significantly lowers the catalyst cost burden, contributing to substantial cost savings in the final product pricing.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for downstream manufacturers who cannot afford production stoppages. This synthetic route relies on chemicals that are universally available in the global chemical market, such as p-tolyl magnesium bromide precursors and standard reducing agents. Unlike bio-based extraction which is subject to climate change and crop diseases, this chemical synthesis can be run continuously in a reactor, independent of external environmental factors. This predictability allows procurement managers to negotiate long-term contracts with greater confidence. Moreover, the robustness of the cobalt catalyst system implies that the process is less sensitive to minor fluctuations in feedstock quality, ensuring consistent output quality even when sourcing raw materials from different vendors, thereby reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: As production volumes increase from kilograms to tons, environmental compliance becomes a critical bottleneck. This route is designed with scalability in mind, avoiding the use of highly toxic heavy metals or persistent organic pollutants where possible. While chromium-based oxidants (PDC) are used in the final step, they are employed in a controlled manner that allows for effective waste treatment and chromium recovery, adhering to strict environmental regulations. The streamlined nature of the synthesis means fewer unit operations, which correlates to lower energy consumption for heating, cooling, and agitation. This reduced energy intensity aligns with corporate sustainability goals and reduces the carbon footprint of the manufacturing process, making it an attractive option for companies committed to green chemistry principles without sacrificing economic performance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding the nuances of the catalyst system and the purification requirements is essential for successful technology transfer. We have compiled these responses to assist technical teams in evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the optical purity achieved in this novel synthesis method?
A: The patented method achieves a high optical purity of 92% ee for the final (S)-ar-Turmerone product, ensuring suitability for high-value applications.
Q: Why is the cobalt-catalyzed Kumada coupling advantageous over chiral pool methods?
A: Unlike chiral pool methods which rely on expensive, naturally occurring starting materials with limited supply, this method uses readily available racemic esters and an efficient catalyst system, significantly lowering raw material costs.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the route utilizes standard reagents like DIBAL-H and PDC and avoids extremely harsh conditions, making it highly amenable to scale-up from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-ar-Turmerone Supplier
The development of efficient synthetic routes like the one described in CN103787855A underscores the importance of partnering with a contract development and manufacturing organization (CDMO) that possesses deep technical expertise. NINGBO INNO PHARMCHEM stands at the forefront of this industry, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling sensitive organometallic reactions under inert atmospheres, ensuring that the delicate cobalt-catalyzed steps are executed with precision. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify optical purity and impurity profiles at every stage of the synthesis. This commitment to quality ensures that the (S)-ar-Turmerone we supply meets the exacting standards required for both flavor applications and pharmaceutical development.
We invite potential partners to engage with our technical team to explore how this innovative synthesis can be tailored to your specific volume and purity requirements. By leveraging our process optimization capabilities, we can help you achieve a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this catalytic route. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can accelerate your product development timelines and secure a sustainable, cost-effective supply chain for this valuable chiral intermediate.
