Revolutionizing Alpha-Chloro Ketone Production: A Silica-Catalyzed Green Approach for Commercial Scale
Revolutionizing Alpha-Chloro Ketone Production: A Silica-Catalyzed Green Approach for Commercial Scale
The synthesis of alpha-monochloroketone compounds represents a critical transformation in the preparation of high-value fine chemicals, serving as indispensable building blocks for a vast array of pharmaceutical and agrochemical active ingredients. Patent CN101429082A introduces a groundbreaking methodology that addresses long-standing inefficiencies in halogenation chemistry by utilizing a silica gel-catalyzed system with 1,3-dichloro-5,5-dimethylhydantoin (DCDMH). This innovative approach not only streamlines the synthetic pathway but also aligns perfectly with modern green chemistry principles by eliminating the need for corrosive liquid acids and toxic chlorine gas. The versatility of this method is demonstrated by its applicability to a wide range of carbonyl substrates, including acetophenones and beta-keto esters, as illustrated by the general structural framework below.
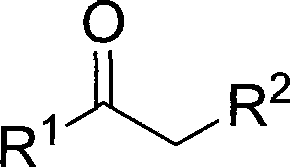
For R&D directors and process chemists seeking a reliable alpha-monochloroketone supplier, this technology offers a robust alternative to traditional routes that often suffer from poor selectivity and hazardous waste streams. By leveraging the mild Lewis acidity of silica gel, the reaction achieves exceptional yields ranging from 70% to 98% across diverse substrates, ensuring a consistent supply of high-purity alpha-chloro ketones essential for downstream coupling reactions. The operational simplicity, involving standard reflux conditions in methanol or ethanol, significantly lowers the barrier for technology transfer from laboratory bench to pilot plant, facilitating the commercial scale-up of complex fine chemical intermediates without compromising safety or quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alpha-chlorination of ketones has relied heavily on the use of elemental chlorine gas, sulfuryl chloride, or N-chlorosuccinimide (NCS), often in the presence of strong homogeneous acid catalysts like p-toluenesulfonic acid. These conventional methodologies present severe drawbacks for industrial application, primarily due to the high toxicity and handling hazards associated with chlorine gas, which requires specialized corrosion-resistant equipment and rigorous safety protocols. Furthermore, the use of strong liquid acids generates substantial volumes of acidic wastewater, imposing heavy burdens on environmental treatment facilities and escalating operational expenditures for waste neutralization and disposal. Selectivity is another persistent challenge, as aggressive chlorinating conditions frequently lead to over-chlorination, producing difficult-to-separate di-chloro impurities that compromise the purity profile required for pharmaceutical grade intermediates.
The Novel Approach
In stark contrast, the novel silica-catalyzed protocol described in the patent data utilizes 1,3-dichloro-5,5-dimethylhydantoin as a stable, solid chlorinating agent that releases chlorine equivalents in a controlled manner. This shift from hazardous gases and liquids to solid reagents fundamentally alters the safety profile of the manufacturing process, allowing for operation in standard glass-lined or stainless steel reactors without the need for exotic materials. The replacement of soluble strong acids with insoluble silica gel not only enhances reaction selectivity towards the mono-chlorinated product but also simplifies the post-reaction workup to a mere filtration step. This elimination of acidic effluents directly contributes to cost reduction in pharmaceutical intermediate manufacturing by removing the need for extensive neutralization processes and reducing the environmental footprint of the production facility.
Mechanistic Insights into Silica-Promoted Alpha-Chlorination
The efficacy of this transformation relies on the unique surface properties of silica gel, which acts as a solid acid catalyst to promote the enolization of the carbonyl substrate. Under reflux conditions in alcoholic solvents, the silanol groups on the silica surface interact with the carbonyl oxygen, increasing the acidity of the alpha-protons and facilitating the formation of the nucleophilic enol or enolate species. Simultaneously, the dichlorohydantoin reagent serves as an electrophilic chlorine source, reacting selectively with the activated enol intermediate to install the chlorine atom at the alpha position. This heterogeneous catalytic cycle ensures that the reaction proceeds under mild thermal conditions, typically between 10°C and 100°C, preventing thermal degradation of sensitive functional groups often present in complex drug intermediates.
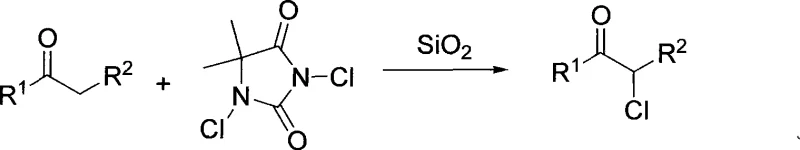
From an impurity control perspective, the mechanistic pathway inherently suppresses the formation of poly-chlorinated byproducts. Unlike free-radical chlorination with chlorine gas which can be difficult to arrest at the mono-stage, the stoichiometry and reactivity of the DCDMH-silica system favor single substitution. The solid catalyst surface likely provides a constrained environment that limits the accessibility of the already chlorinated product to further electrophilic attack, thereby ensuring high regioselectivity. For procurement teams, this high selectivity translates to simplified purification trains, as the crude product typically requires only basic extraction and crystallization to meet stringent purity specifications, reducing lead time for high-purity agrochemical intermediates and API precursors significantly compared to legacy processes.
How to Synthesize Alpha-Monochloroketones Efficiently
The practical implementation of this synthesis route is designed for operational ease, requiring minimal specialized equipment while delivering consistent results across various substrate classes. The process involves a straightforward charge of reagents followed by a thermal reaction period and a simple filtration workup, making it highly attractive for both kilogram-scale development and multi-ton commercial production. Detailed standardized operating procedures regarding specific molar ratios, solvent choices, and temperature profiles are critical for maximizing yield and maintaining safety standards during scale-up.
- Charge a reactor with commercially available silica gel (100-400 mesh), the carbonyl substrate, and 1,3-dichloro-5,5-dimethylhydantoin (0.5-1.0 equivalents) in methanol or ethanol.
- Heat the reaction mixture to reflux (10-100°C) and maintain stirring for 1 to 2 hours to ensure complete conversion to the monochlorinated product.
- Filter off the silica catalyst, remove the solvent via rotary evaporation, extract the residue with methyl butyl ether, wash with brine, dry over MgSO4, and concentrate to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this silica-catalyzed technology offers tangible strategic advantages beyond mere technical performance. The reliance on commodity chemicals such as silica gel and dichlorohydantoin, rather than specialized or regulated reagents like chlorine gas or NCS, ensures a stable and resilient supply chain that is less susceptible to market volatility or regulatory restrictions. The absence of corrosive liquid acids eliminates the logistical challenges and costs associated with the storage, handling, and neutralization of hazardous acidic waste, leading to substantial operational savings. Furthermore, the heterogeneous nature of the catalyst allows for potential recovery and reuse strategies, further driving down the cost of goods sold (COGS) and enhancing the overall sustainability profile of the manufacturing site.
- Cost Reduction in Manufacturing: The elimination of expensive chlorinating agents like NCS and the removal of costly acid waste treatment processes result in significant economic benefits. By utilizing cheap, bulk-available silica gel and DCDMH, manufacturers can drastically reduce raw material costs while avoiding the capital expenditure required for corrosion-resistant infrastructure. The simplified workup procedure also reduces labor hours and solvent consumption, contributing to a leaner and more cost-efficient production model that improves margin potential for high-volume intermediates.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key reagents are widely produced commodities with established global supply chains, unlike specialized catalysts or hazardous gases that may face shipping restrictions. The robustness of the reaction conditions means that production is less likely to be interrupted by minor variations in raw material quality or environmental constraints, ensuring consistent delivery schedules for downstream customers. This reliability is crucial for maintaining continuous API production lines and meeting the just-in-time delivery expectations of major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of solid catalysts and standard solvents, allowing for seamless transition from pilot batches to full commercial scale without complex engineering changes. From an environmental standpoint, the "green" nature of the process, characterized by zero acidic wastewater discharge and the use of recyclable catalysts, ensures full compliance with increasingly stringent global environmental regulations. This proactive approach to sustainability mitigates regulatory risk and enhances the corporate social responsibility profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced chlorination technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: What are the advantages of using silica gel over traditional acid catalysts?
A: Silica gel acts as a mild, heterogeneous solid acid catalyst, eliminating the need for corrosive liquid acids like p-toluenesulfonic acid. This simplifies workup by simple filtration, prevents acidic wastewater generation, and significantly reduces environmental compliance costs.
Q: How does this method control selectivity between mono- and di-chlorination?
A: The use of dichlorohydantoin (DCDMH) combined with the specific surface properties of silica gel allows for precise control over the chlorination potential. The reaction conditions favor the formation of the alpha-monochloro product with high selectivity, minimizing the formation of over-chlorinated di-chloro impurities often seen with harsher reagents.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. It utilizes cheap, commercially available reagents (silica, DCDMH) and common alcohol solvents. The heterogeneous nature of the catalyst allows for easy separation and potential reuse, making it ideal for cost reduction in pharmaceutical intermediate manufacturing on a multi-ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Monochloroketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality alpha-chloro ketones play in the synthesis of next-generation therapeutics and crop protection agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from gram-scale optimization to industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for GMP manufacturing environments.
We invite you to collaborate with our technical team to evaluate the feasibility of integrating this green chlorination route into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic and environmental benefits specific to your target molecule. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and value for your organization.
