Advanced Synthesis of Ferrocenyl Thiadiazole Mannich Bases for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high efficiency with environmental sustainability. Patent CN110878106B introduces a significant advancement in the preparation of ferrocenyl thiadiazole or oxadiazolyl Mannich bases, compounds that hold immense potential in medicinal chemistry due to their unique redox properties and biological activity. The integration of the ferrocene moiety into heterocyclic scaffolds like thiadiazoles creates molecules with enhanced stability and diverse pharmacological profiles, ranging from antibacterial to antiproliferative applications. This patent specifically addresses the longstanding challenges associated with traditional Mannich reactions, which often rely on harsh acidic conditions that complicate downstream processing and generate substantial hazardous waste. By leveraging a novel catalytic system based on benzimidazole ionic liquids, this technology offers a streamlined pathway to access these valuable intermediates with exceptional purity and yield.
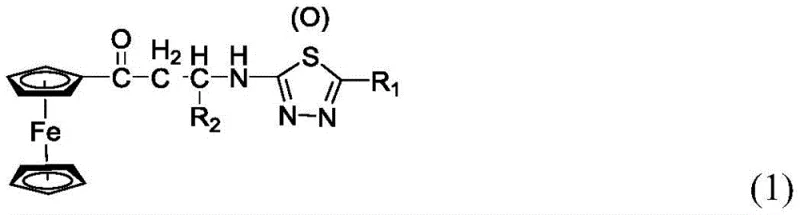
The core innovation lies in the structural versatility of the resulting Mannich bases, as depicted in the general formula where R1 and R2 can encompass a wide array of substituents including halogens, nitro groups, and alkoxy chains. This flexibility allows chemists to fine-tune the electronic and steric properties of the final molecule to optimize binding affinity for specific biological targets. Furthermore, the presence of the ferrocenyl group imparts distinct electrochemical characteristics that are increasingly valued in the development of biosensors and advanced material science applications alongside traditional drug discovery. For procurement managers and supply chain directors, understanding the underlying chemistry is crucial, as the choice of synthetic route directly impacts the cost of goods sold and the reliability of supply for these critical pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Mannich bases containing ferrocenyl and heterocyclic units has relied heavily on the use of strong mineral acids such as concentrated hydrochloric acid, Lewis acids like aluminum trichloride, or organic acids like acetic acid. While these catalysts can drive the reaction forward, they introduce severe operational drawbacks that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. The primary issue is the corrosive nature of these reagents, which necessitates the use of specialized, corrosion-resistant reactor equipment, thereby increasing capital expenditure. Moreover, the post-reaction workup is notoriously difficult; neutralizing large volumes of strong acid generates significant amounts of saline waste that requires expensive treatment before disposal. The catalysts themselves are typically consumed in the process or lost during aqueous workup, preventing recovery and leading to higher raw material costs per batch. Additionally, the harsh acidic environment can sometimes lead to side reactions or decomposition of sensitive functional groups, compromising the overall yield and purity of the desired product.
The Novel Approach
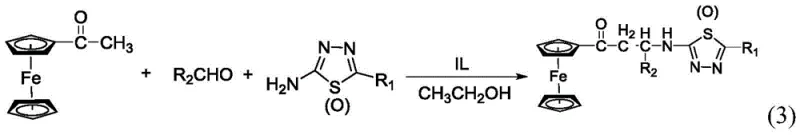
In stark contrast, the methodology described in patent CN110878106B utilizes a benzimidazole ionic liquid as a reusable, green catalyst, fundamentally altering the economic and environmental landscape of this synthesis. The reaction proceeds smoothly in anhydrous ethanol, a benign and widely available solvent, under mild reflux conditions at approximately 80°C. As illustrated in the reaction scheme below, the process involves a simple one-pot condensation of 2-amino-5-substituted-1,3,4-thiadiazole, an aldehyde, and acetylferrocene. The ionic liquid not only accelerates the formation of the C-N and C-C bonds characteristic of the Mannich reaction but also remains stable throughout the process. Crucially, upon completion of the reaction, the ionic liquid precipitates out or can be easily separated via filtration, allowing it to be recovered and reused for subsequent batches without significant loss of activity. This eliminates the need for acid neutralization steps and drastically simplifies the isolation of the crude product, which is then purified via straightforward recrystallization.
Mechanistic Insights into Benzimidazole Ionic Liquid Catalysis
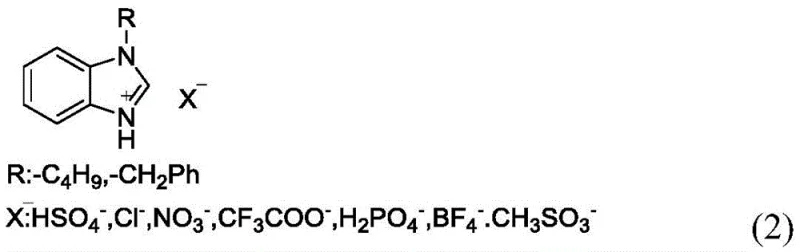
The efficacy of this synthetic route is rooted in the unique physicochemical properties of the benzimidazole ionic liquid catalyst. Unlike traditional protonic acids that rely solely on Brønsted acidity, ionic liquids can offer a dual activation mechanism involving both hydrogen bonding and electrostatic interactions. In the context of the Mannich reaction, the ionic liquid likely activates the carbonyl group of the aldehyde, making it more susceptible to nucleophilic attack by the amine nitrogen of the thiadiazole ring to form the imine intermediate. Simultaneously, the ionic environment stabilizes the transition states involved in the subsequent addition of the ferrocenyl enol or enolate species. The structure of the catalyst, featuring a benzimidazole cation paired with various anions such as nitrate or bisulfate as shown in the patent data, provides a tunable platform where the acidity and solubility can be optimized for specific substrates. This precise control over the reaction microenvironment minimizes the formation of by-products and ensures high regioselectivity, which is paramount when synthesizing complex molecules for drug development.
From a quality control perspective, the mechanism also contributes to a cleaner impurity profile. Traditional acid-catalyzed reactions often promote polymerization of the aldehyde or degradation of the acid-sensitive ferrocene unit, leading to complex mixtures that are difficult to separate. The mild, non-corrosive nature of the ionic liquid preserves the integrity of the ferrocene sandwich structure and the heterocyclic ring throughout the synthesis. Experimental data from the patent confirms this, showing consistent melting points and high purity across a series of derivatives with different electronic substituents on the phenyl ring (e.g., chloro, nitro, methoxy). The ability to tolerate such a wide range of functional groups without protection-deprotection sequences significantly shortens the synthetic timeline. For R&D directors, this implies a faster route to generating libraries of analogs for structure-activity relationship (SAR) studies, accelerating the lead optimization phase of drug discovery projects.
How to Synthesize Ferrocenyl Thiadiazole Mannich Base Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory-scale optimization and pilot plant production. The process begins by charging a dry three-necked flask with the amine and aldehyde components in anhydrous ethanol, followed by the addition of the ionic liquid catalyst. The acetylferrocene solution is then added dropwise to control the exotherm and ensure uniform mixing. Reaction progress is conveniently monitored using thin-layer chromatography (TLC) with a standard ethyl acetate and petroleum ether system, allowing operators to determine the exact endpoint and prevent over-reaction. Once the starting material is consumed, the solvent is removed under reduced pressure, and the residue is treated with ethyl acetate. A simple suction filtration step separates the solid ionic liquid catalyst from the product-containing solution. The filtrate is evaporated to yield the crude solid, which is finally recrystallized from absolute ethanol to afford the pure target compound. Detailed standardized synthesis steps are provided in the guide below.
- Mix 2-amino-5-substituted-1,3,4-thiadiazole and aldehyde in anhydrous ethanol within a dry three-necked flask.
- Add benzimidazole ionic liquid catalyst and slowly drip in the acetylferrocene ethanol solution while stirring.
- Heat to reflux until TLC indicates completion, then filter to recover the catalyst and recrystallize the filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the shift from traditional acid catalysis to this ionic liquid-mediated process offers compelling economic benefits. The most immediate impact is seen in the reduction of operational expenditures related to waste management and raw material consumption. By eliminating the need for stoichiometric amounts of corrosive acids and the subsequent neutralization bases, the facility avoids the costs associated with handling hazardous chemicals and treating saline wastewater. Furthermore, the recyclability of the catalyst means that the effective cost of the catalytic system is amortized over many batches, leading to substantial long-term savings. The use of ethanol as the sole solvent simplifies solvent recovery systems, as there is no need for complex multi-solvent distillation trains often required when using mixed organic-aqueous systems in traditional workups.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous mineral acids directly lowers the bill of materials for each production run. More importantly, the ability to recover and reuse the benzimidazole ionic liquid multiple times without significant degradation transforms the catalyst from a consumable expense into a durable asset. This circular usage model significantly reduces the cost per kilogram of the final API intermediate. Additionally, the high yields reported (often exceeding 90%) mean that less raw material is wasted on side products, maximizing the atom economy of the process and ensuring that the expensive ferrocene starting material is utilized with maximum efficiency.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method enhances supply security by reducing the risk of batch failures due to sensitive reaction conditions. Traditional methods requiring strict anhydrous conditions or low temperatures are prone to variability, whereas this reflux protocol in ethanol is forgiving and easy to control. The simplified workup, involving merely filtration and evaporation, reduces the processing time per batch, allowing for higher throughput in existing manufacturing facilities. This agility enables suppliers to respond more quickly to fluctuations in demand from downstream pharmaceutical clients, ensuring consistent availability of these critical building blocks.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this green chemistry approach mitigates those concerns. The absence of corrosive acids reduces the risk of equipment failure and operator injury, lowering insurance and maintenance costs. From a regulatory standpoint, the process aligns with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and hazardous waste generation. The "green" label associated with ionic liquid catalysis can also be a valuable marketing asset for pharmaceutical companies aiming to improve the sustainability profile of their supply chain, potentially facilitating faster regulatory approvals in markets that prioritize eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the advantages of using benzimidazole ionic liquid over traditional acid catalysts?
A: Unlike corrosive mineral acids like hydrochloric acid or aluminum trichloride, benzimidazole ionic liquids are environmentally benign, easily recoverable via simple filtration, and can be reused multiple times without significant loss of catalytic activity, drastically reducing waste treatment costs.
Q: What is the typical yield and purity profile for this synthesis route?
A: Experimental data from the patent indicates high yields ranging from 90.7% to 93.6% across various substituted derivatives. The products are obtained as high-purity solids after simple recrystallization from anhydrous ethanol, minimizing the need for complex chromatographic purification.
Q: Is this process scalable for industrial manufacturing of pharmaceutical intermediates?
A: Yes, the process utilizes standard solvents like anhydrous ethanol and operates at atmospheric pressure under reflux conditions (approx. 80°C). The simple workup procedure involving filtration and evaporation makes it highly suitable for scale-up from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ferrocenyl Thiadiazole Mannich Base Supplier
The synthesis of ferrocenyl thiadiazole Mannich bases represents a sophisticated intersection of organometallic chemistry and heterocyclic synthesis, requiring a partner with deep technical expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of ionic liquid chemistry, ensuring that the benefits of catalyst recovery and high purity are fully realized at an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity pharmaceutical intermediates meets the exacting standards required for drug substance manufacturing.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for these valuable compounds. By leveraging our expertise in green catalysis and process intensification, we can help you achieve significant efficiencies in your production workflows. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of patent CN110878106B can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
