Advanced Enzymatic Synthesis of Safraninib Intermediates for Commercial Scale-up
The pharmaceutical landscape for oncology treatments continues to evolve with the demand for potent c-Met kinase inhibitors like Safraninib (also known as HMPL-504 or Voltinib). Patent CN112930348A presents a significant technological leap in the manufacturing of this critical therapeutic agent, specifically addressing the bottlenecks associated with its complex heterocyclic structure. This specification details an improved process for the manufacture of 3-[(1S)-1-imidazo[1,2-a]pyridin-6-ylethyl]-5-(1-methylpyrazol-4-yl)triazolo[4,5-b]pyrazine, focusing on efficiency, stereochemical control, and scalability. By shifting from a resolution-based approach to an enzymatic asymmetric synthesis, the patent outlines a pathway that is inherently more robust for industrial application. The core structure of the target molecule, which features a fused triazolo-pyrazine system linked to an imidazopyridine and a methylpyrazole moiety, presents significant synthetic challenges that this new methodology aims to overcome.
![Chemical structure of Safraninib (Formula I) showing the triazolo[4,5-b]pyrazine core linked to imidazopyridine and methylpyrazole groups](/insights/img/safraninib-intermediate-synthesis-supplier-20260306001841-01.webp)
For R&D directors evaluating process viability, the structural integrity and purity of such complex small molecules are paramount. The patent explicitly defines the compound and its pharmaceutically acceptable salts, ensuring that the synthetic route delivers a product suitable for therapeutic use in treating non-small cell lung cancer and papillary renal cell carcinoma. The introduction of this improved process signals a move towards greener chemistry and higher atom economy, which are critical metrics for modern API development. Understanding the molecular architecture is the first step in appreciating the synthetic innovations proposed, as the placement of the chiral center and the fusion of the nitrogen-rich rings dictate the reactivity profiles required for successful scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically referenced as WO 2011079804, describes a 7-step synthesis starting from 1-imidazo[1,2-a]pyridin-6-yl ethanone. While functional, this conventional route suffers from severe inefficiencies that render it suboptimal for large-scale commercial production. The most critical drawback lies in the final step, which involves the chiral resolution of the racemic triazolo[4,5-b]pyrazine into its R and S enantiomers. This resolution step inherently results in a maximum theoretical yield loss of approximately 50% of the material produced throughout the entire preceding synthesis. Such material attrition is economically unsustainable and generates a substantial volume of chemical waste, posing environmental and disposal challenges.
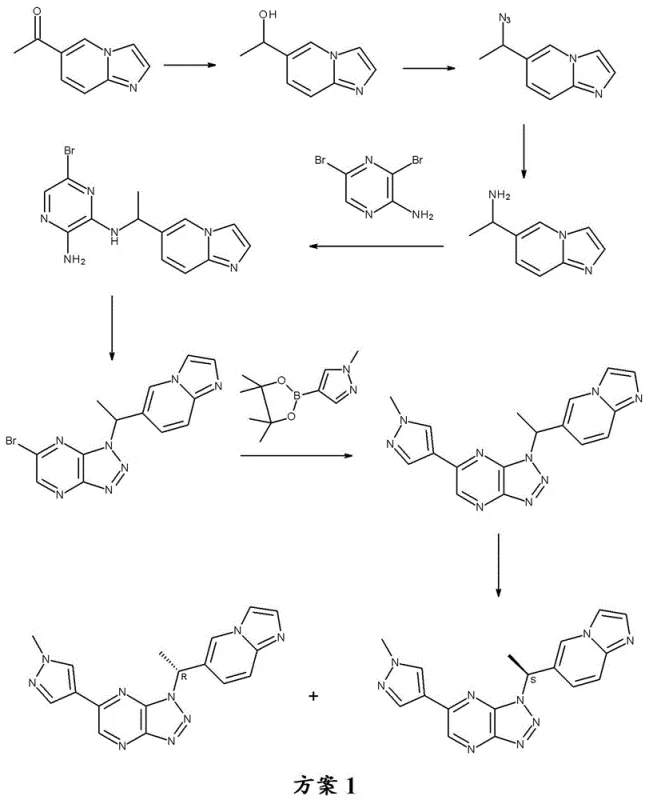
Furthermore, the conventional route lacks controlled separation points for purification, necessitating chromatographic purification of key intermediates like 5-bromo-3-(1-imidazo[1,2-a]pyridin-6-ylethyl)triazolo[4,5-b]pyrazine. Chromatography is notoriously difficult to scale industrially due to solvent consumption, column packing limitations, and throughput constraints. The reliance on this technique indicates a process that was likely designed for laboratory-scale discovery rather than multi-kilogram or ton-scale manufacturing. For procurement managers, these factors translate into higher costs of goods sold (COGS) and potential supply chain vulnerabilities due to the complexity of waste management and low overall yield.
The Novel Approach
In stark contrast, the improved process detailed in CN112930348A streamlines the synthesis into just 5 steps from the same starting ketone, fundamentally altering the economic and operational profile of the manufacture. The pivotal innovation is the introduction of a chiral center at the very beginning of the sequence via enzymatic asymmetric transamination. This produces a chiral intermediate compound (III), (1S)-1-imidazo[1,2-a]pyridin-6-ylethylamine, which carries the desired stereochemistry through the entire synthesis. By establishing chirality early, the process completely avoids the need for the uneconomical chiral resolution of the final product, theoretically doubling the yield relative to the resolution-based route.
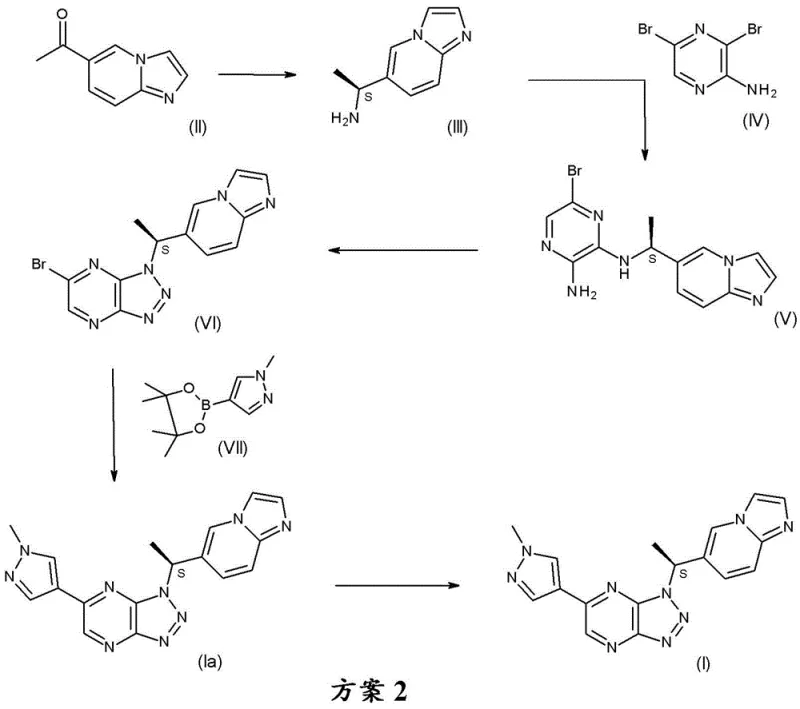
Additionally, this novel route is designed with industrial practicality in mind, enabling intermediates (III) and (V) to be easily isolated and purified without the need for chromatography. The ability to purify via crystallization or simple extraction significantly enhances the scalability of the process. For supply chain heads, this means a more robust manufacturing protocol that is less susceptible to the bottlenecks associated with complex purification techniques. The reduction in step count from 7 to 5 not only saves time but also reduces the cumulative loss of material at each stage, leading to a substantially higher overall yield and a more reliable supply of high-purity pharmaceutical intermediates.
Mechanistic Insights into Enzymatic Asymmetric Transamination and Suzuki Coupling
The cornerstone of this improved synthesis is the enzymatic asymmetric transamination of 1-imidazo[1,2-a]pyridin-6-yl-ethanone (II). This biocatalytic step utilizes an amine transaminase enzyme, specifically identified in the examples as ATA-436, in the presence of a pyridoxal phosphate cofactor. The reaction proceeds in a buffered aqueous system, typically at pH 10 using sodium tetraborate, with isopropylamine hydrochloride serving as the amine donor. The mechanism involves the transfer of an amino group to the prochiral ketone, selectively generating the (S)-enantiomer of the amine. This biocatalytic approach offers exquisite stereoselectivity that is difficult to achieve with traditional chemical catalysts, ensuring high enantiomeric excess (ee) right from the start. The reaction is conducted at elevated temperatures, ranging from 44°C to 54°C, which facilitates reasonable reaction rates while maintaining enzyme stability.
Following the formation of the chiral amine, the synthesis proceeds through a nucleophilic aromatic substitution with 3,5-dibromopyrazin-2-amine (IV) to form the diamine intermediate (V). This is followed by a diazotization and cyclization step using sodium nitrite under acidic conditions to construct the triazolo[4,5-b]pyrazine ring system. The final key transformation is a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction. Here, the bromo-triazolo-pyrazine intermediate (VI) reacts with 1-methyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyrazole (VII). The patent specifies the use of specialized palladium catalysts such as Pd(AmPhos)2Cl2 (Pd-132) to facilitate this bond formation efficiently. Crucially, the process includes a dedicated step for treating the crude product with a palladium scavenger, such as L-cysteine or activated carbon, to reduce residual metal content to acceptable levels, a critical quality attribute for oncology drugs.
How to Synthesize Safraninib Efficiently
The synthesis of Safraninib via this improved route requires precise control over reaction parameters to maximize yield and purity. The process begins with the biocatalytic generation of the chiral amine, followed by sequential coupling and cyclization steps that build the complex heterocyclic core. The final stages involve the Suzuki coupling and rigorous purification protocols to ensure the removal of catalysts and impurities. The detailed operational parameters, including specific solvent systems like sec-butanol for the coupling reaction and temperature cycles for crystallization, are critical for reproducibility. For technical teams looking to implement this chemistry, understanding the nuances of each isolation step is essential for achieving the desired polymorphic form and particle size distribution.
- Perform enzymatic asymmetric transamination of 1-imidazo[1,2-a]pyridin-6-yl-ethanone using ATA-436 enzyme and isopropylamine to establish chirality early.
- React the chiral amine intermediate with 3,5-dibromopyrazin-2-amine followed by cyclization with sodium nitrite to form the triazolo[4,5-b]pyrazine core.
- Execute a palladium-catalyzed Suzuki coupling with a methylpyrazole boronic ester, followed by scavenging and crystallization to isolate high-purity Safraninib.
Commercial Advantages for Procurement and Supply Chain Teams
The transition from the conventional resolution-based route to this enzymatic asymmetric synthesis offers profound commercial advantages that extend beyond simple yield improvements. For procurement managers, the elimination of the late-stage chiral resolution represents a direct and significant reduction in raw material costs. By avoiding the inherent 50% loss associated with resolving racemates, the effective cost of the starting materials is halved relative to the output of active ingredient. Furthermore, the removal of chromatographic purification steps drastically reduces the consumption of expensive solvents and silica gel, lowering both material costs and waste disposal fees. This streamlined approach translates into a more competitive pricing structure for the final API, allowing for better margin management in a cost-sensitive pharmaceutical market.
- Cost Reduction in Manufacturing: The primary driver for cost reduction is the strategic placement of the chirality introduction at the beginning of the synthesis. This eliminates the need for resolving agents and the associated loss of half the product mass. Additionally, the replacement of chromatography with crystallization and filtration operations significantly lowers operational expenditures (OPEX). Crystallization is a unit operation that scales linearly and predictably, unlike chromatography which faces diminishing returns at larger scales. The use of robust enzymes like ATA-436 also suggests a potential for catalyst recycling or immobilization, further driving down the cost per kilogram of the chiral intermediate.
- Enhanced Supply Chain Reliability: The improved process enhances supply chain reliability by simplifying the manufacturing workflow. With fewer steps (5 vs 7) and the removal of bottleneck operations like preparative HPLC or flash chromatography, the lead time for production batches is significantly reduced. The intermediates generated in this route, such as the chiral amine (III) and the bromo-triazolo intermediate (VI), are stable and can be stockpiled, providing flexibility in production scheduling. This modularity allows manufacturers to decouple different stages of synthesis, mitigating the risk of a single point of failure disrupting the entire supply of the final drug substance.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the new route is superior. The enzymatic step operates in aqueous buffers, reducing the reliance on volatile organic compounds (VOCs) in the early stages. The subsequent steps utilize solvents like NMP and sec-butanol which, while requiring careful handling, are manageable in standard stainless steel reactors. The reduction in chemical waste, particularly the avoidance of the unwanted enantiomer from resolution, aligns with green chemistry principles and simplifies regulatory filings regarding environmental impact. This makes the process more attractive for manufacturing in regions with strict environmental regulations, ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of Safraninib. These insights are derived directly from the experimental data and claims within CN112930348A, providing clarity on the feasibility and advantages of this manufacturing route. Understanding these details is crucial for stakeholders evaluating the technology for licensing or contract manufacturing opportunities.
Q: How does the new process improve upon the conventional synthesis of Safraninib?
A: The improved process reduces the synthesis from 7 steps to 5 steps and eliminates the inefficient late-stage chiral resolution, thereby avoiding a theoretical 50% material loss and removing the need for chromatographic purification.
Q: What specific enzyme is utilized for the asymmetric synthesis in this patent?
A: The process utilizes the ATA-436 amine transaminase enzyme in the presence of a pyridoxal phosphate cofactor and an amine source such as isopropylamine hydrochloride to generate the chiral amine intermediate.
Q: How is residual palladium managed in the final coupling step?
A: Residual palladium is effectively removed using specific scavengers such as L-cysteine or activated carbon, ensuring the final API meets stringent heavy metal specifications without requiring complex filtration setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Safraninib Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent methodologies into commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the enzymatic and chemical steps described in CN112930348A to ensure consistent quality and yield. We understand that the production of oncology intermediates like Safraninib demands stringent purity specifications and rigorous QC labs to detect and control genotoxic impurities and residual metals. Our state-of-the-art facilities are equipped to handle the specific requirements of biocatalytic reactions and palladium-mediated couplings, ensuring a seamless transition from lab scale to full commercial manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this improved process can benefit your supply chain. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this enzymatic route. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our manufactured intermediates against your internal standards. Let us collaborate to secure a reliable, cost-effective, and high-quality supply of Safraninib for your global pharmaceutical needs.
