Revolutionizing Rivaroxaban Production: A Safe, One-Pot Commercial Strategy
Revolutionizing Rivaroxaban Production: A Safe, One-Pot Commercial Strategy
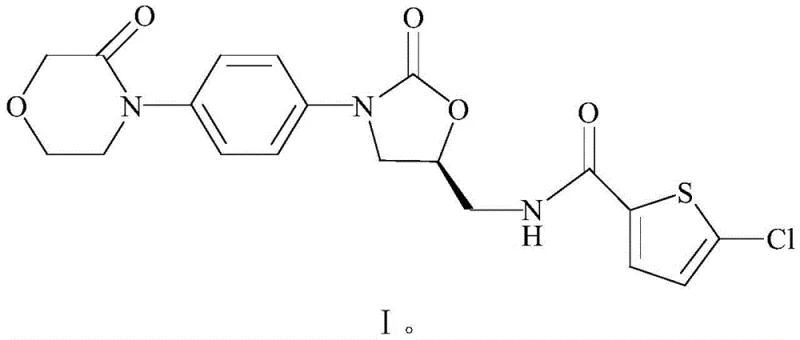
The global demand for direct Factor Xa inhibitors continues to surge, placing immense pressure on supply chains to deliver high-quality anticoagulants efficiently. Patent CN108250193B introduces a transformative approach to synthesizing Rivaroxaban, shifting away from hazardous multi-step protocols toward a streamlined one-pot methodology. This technical breakthrough utilizes 2-[[ (5S)-2-oxo-3-[4-(3-oxo-4-morpholinyl)phenyl]-5-oxazolidinyl]methyl]-1H-isoindole-1,3(2H)-diketone as a robust starting material, bypassing the instability issues inherent in previous generations of synthesis. By integrating ammonolysis and amidation into a continuous sequence, manufacturers can achieve superior control over impurity profiles while drastically simplifying downstream processing. For R&D directors and procurement leaders, this represents a critical opportunity to optimize the cost structure of high-purity Rivaroxaban production without compromising on the stringent quality standards required for cardiovascular therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Rivaroxaban has been plagued by significant safety and efficiency bottlenecks, particularly those relying on acid chloride intermediates. Prior art, such as US Patent 7585860B2, necessitates the use of 5-chlorothiophene-2-carbonyl chloride, a highly unstable and corrosive species that requires generation using toxic reagents like thionyl chloride or phosphorus oxychloride. These conventional routes often mandate cryogenic conditions, such as 0°C, to manage exothermic risks, followed by complex workups involving dichloromethane extraction and flash chromatography. Such reliance on column chromatography is commercially impractical for metric-ton scale operations, creating severe bottlenecks in throughput and generating excessive chemical waste. Furthermore, the handling of pyridine and volatile acid chlorides introduces substantial occupational health hazards, complicating regulatory compliance and increasing the overall cost of goods sold through expensive containment and scrubbing requirements.
The Novel Approach
In stark contrast, the methodology disclosed in CN108250193B leverages a direct coupling strategy that eliminates the need for pre-activating the carboxylic acid into an acid chloride. By employing 4-(4-aminomethyl-2-oxo-oxazolidin-3-yl)-phenyl-morpholin-3-one generated in situ, the process utilizes 5-chlorothiophene-2-carboxylic acid directly in the presence of the advanced condensing agent DMTMM (4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride). This innovation allows the reaction to proceed in environmentally benign alcohol or alcohol-water systems at ambient temperatures, typically between 20°C and 30°C. The elimination of hazardous activation steps not only enhances operator safety but also simplifies the reactor train requirements, enabling a true one-pot operation where the deprotection and coupling occur sequentially without intermediate isolation. This shift fundamentally alters the economic model of production, offering a pathway for cost reduction in API manufacturing by removing unit operations and minimizing solvent intensity.
Mechanistic Insights into DMTMM-Mediated Amidation
The core of this technological advancement lies in the efficient activation of the carboxylic acid moiety using DMTMM, a triazine-based condensing agent known for its high reactivity and water tolerance. Mechanistically, DMTMM activates the 5-chlorothiophene-2-carboxylic acid to form a highly reactive ester intermediate, which is subsequently attacked by the nucleophilic amine generated from the ammonolysis of the phthalimide protecting group. This reaction pathway is exceptionally clean, avoiding the formation of urea by-products common with carbodiimide reagents like EDCI or DCC, which are notoriously difficult to remove from the final API. The use of aqueous methylamine for the initial deprotection step ensures rapid cleavage of the phthalimide group under mild thermal conditions (60-70°C), generating the free amine intermediate (Formula III) which remains stable enough in the alcoholic medium to undergo immediate coupling. This tandem sequence minimizes the exposure of the chiral center to harsh basic or acidic conditions, thereby preserving the stereochemical integrity essential for the drug's biological activity.
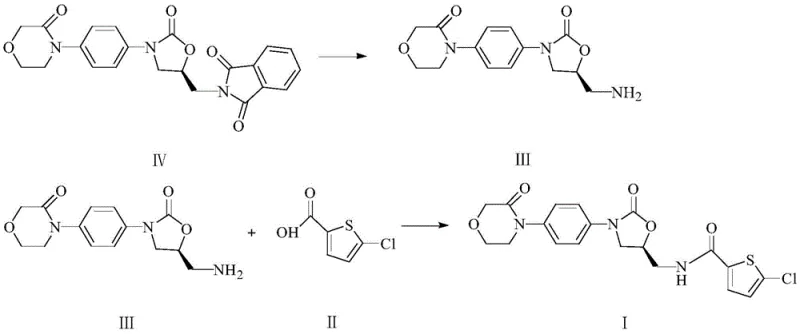
Impurity control is rigorously maintained through the selection of solvents and stoichiometry, specifically controlling the molar ratio of methylamine to the precursor between 1.1:1 and 1.3:1. Excess amine is carefully managed to prevent side reactions during the subsequent amidation phase, ensuring that the final crude product precipitates with high purity directly from the reaction mixture. The patent data indicates that by optimizing the solvent system to include ethanol or methanol mixed with water, the solubility of by-products is manipulated such that the target Rivaroxaban crystallizes out upon completion. This crystallization-driven purification replaces the need for chromatographic separation, a critical factor for scaling. The result is a product with chiral purity exceeding 99.9% and chemical purity that meets rigorous pharmacopeial standards without recrystallization from acetic acid or other aggressive solvents, demonstrating a robust mechanism for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Rivaroxaban Efficiently
Implementing this one-pot strategy requires precise control over reaction parameters to maximize yield and minimize cycle time. The process begins with the suspension of the phthalimide-protected precursor in absolute ethanol, followed by the addition of aqueous methylamine to initiate the deprotection phase at elevated temperatures. Once the ammonolysis is complete, indicated by the full conversion to the amine intermediate, the system is cooled to ambient temperature to prepare for the coupling event. The addition of DMTMM and the carboxylic acid solution must be timed to ensure the amine does not degrade before coupling occurs. For a detailed breakdown of the specific operational parameters, including stirring rates, addition times, and filtration protocols, please refer to the standardized synthesis guide below.
- Perform ammonolysis on the phthalimide-protected precursor using aqueous methylamine in alcohol at 60-70°C to generate the free amine intermediate.
- Cool the reaction mixture to 20-30°C and add the condensing agent DMTMM along with 5-chlorothiophene-2-carboxylic acid dissolved in alcohol.
- Stir the mixture for 10-16 hours to complete amidation, then filter, wash with aqueous alcohol, and dry under reduced pressure to obtain pure Rivaroxaban.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain executives and procurement managers, the transition to this DMTMM-mediated one-pot process offers tangible strategic benefits that extend beyond simple yield improvements. The most significant advantage is the complete removal of acid chloride chemistry from the supply chain, which eliminates the need to source, store, and handle hazardous reagents like thionyl chloride and phosphorus oxychloride. This reduction in hazardous material inventory lowers insurance costs, simplifies site permitting, and reduces the risk of supply disruptions caused by regulatory crackdowns on toxic precursors. Furthermore, the ability to perform the reaction in alcohol-water mixtures allows for the use of commodity solvents that are easily recovered and recycled, significantly driving down raw material expenses and waste disposal fees associated with chlorinated solvents like dichloromethane.
- Cost Reduction in Manufacturing: The elimination of column chromatography is a primary driver for cost optimization, as chromatographic purification is capital-intensive and difficult to scale continuously. By relying on precipitation and filtration, the process reduces equipment footprint and labor hours, leading to substantial operational savings. Additionally, the high yield reported in the patent data, consistently exceeding 90%, ensures that raw material utilization is maximized, directly improving the margin profile for the final API. The simplified workup also means less solvent is consumed per kilogram of product, further enhancing the economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: Utilizing stable carboxylic acids instead of reactive acid chlorides improves the shelf-life of starting materials, allowing for bulk purchasing and strategic stockpiling without degradation concerns. The robustness of the one-pot method reduces the number of discrete processing steps, which inherently lowers the probability of batch failures and deviations. This reliability translates to more predictable lead times for customers, ensuring a steady flow of reliable Rivaroxaban supplier inventory to meet global market demand without the volatility associated with complex multi-step syntheses.
- Scalability and Environmental Compliance: The process aligns perfectly with green chemistry principles by reducing the E-factor (mass of waste per mass of product) through the avoidance of toxic reagents and excessive solvent use. The mild reaction conditions (20-30°C) reduce energy consumption for heating and cooling compared to cryogenic processes, contributing to a lower carbon footprint. This environmental stewardship is increasingly critical for maintaining partnerships with major pharmaceutical companies that have strict sustainability mandates, positioning this manufacturing route as a future-proof solution for long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or capacity expansion. The answers provided are derived directly from the experimental data and comparative analysis found within the patent literature, ensuring accuracy and relevance for decision-makers.
Q: Why is the DMTMM condensing agent preferred over acid chlorides for Rivaroxaban?
A: Traditional methods rely on unstable and corrosive acid chlorides requiring toxic reagents like thionyl chloride. The DMTMM method uses stable carboxylic acids directly, significantly improving operational safety and eliminating hazardous waste streams associated with acid chloride formation.
Q: What represents the key advantage of the one-pot technique described in CN108250193B?
A: The one-pot technique combines deprotection and coupling in a single vessel without isolating the unstable amine intermediate. This eliminates solvent swaps and purification steps like column chromatography, drastically reducing production time and solvent consumption while maintaining yields above 90%.
Q: How does this process ensure high chiral purity for the final API?
A: By starting with a chirally pure phthalimide-protected precursor and utilizing mild reaction conditions (20-30°C for coupling), the process prevents racemization. The patent data confirms chiral purity levels reaching 99.9% via HPLC analysis without the need for additional chiral resolution steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rivaroxaban Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies like the one-pot DMTMM coupling is essential for maintaining competitiveness in the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the highest standards for chiral and chemical purity. Our infrastructure is designed to handle complex heterocyclic chemistry safely, providing a secure environment for the manufacture of potent anticoagulants.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your development timelines are met with precision and reliability.
