Scalable Synthesis of Belinostat Key Impurity via Efficient Base Hydrolysis
The pharmaceutical landscape for oncology treatments continues to evolve, with Histone Deacetylase (HDAC) inhibitors like Belinostat representing a critical class of therapeutic agents. As detailed in patent CN102850244A, the synthesis and control of specific impurities are paramount for ensuring the safety and efficacy of the final Active Pharmaceutical Ingredient (API). This patent discloses a highly efficient preparation method for 3-(3-phenylsulfamoyl-phenyl)-acrylic acid, designated as Compound II, which serves as both a crucial synthetic intermediate and a key process-related impurity for Belinostat. The structural complexity of Belinostat, featuring a sulfonamide linkage and a hydroxamic acid moiety, demands precise control over every synthetic step to prevent the carryover of genotoxic or pharmacologically active impurities. 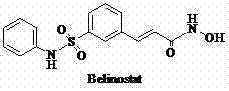 Understanding the genesis of Compound II is essential for R&D teams aiming to optimize the Belinostat supply chain. The disclosed method addresses the need for a reliable source of this specific acid derivative, utilizing a straightforward hydrolysis strategy that converts the corresponding ester precursor into the target carboxylic acid. By leveraging inorganic bases in a mixed solvent system, this process offers a robust alternative to more cumbersome synthetic routes, ensuring that manufacturers can maintain strict quality control standards while meeting the rigorous demands of global regulatory bodies for anticancer drug production.
Understanding the genesis of Compound II is essential for R&D teams aiming to optimize the Belinostat supply chain. The disclosed method addresses the need for a reliable source of this specific acid derivative, utilizing a straightforward hydrolysis strategy that converts the corresponding ester precursor into the target carboxylic acid. By leveraging inorganic bases in a mixed solvent system, this process offers a robust alternative to more cumbersome synthetic routes, ensuring that manufacturers can maintain strict quality control standards while meeting the rigorous demands of global regulatory bodies for anticancer drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing complex aromatic acrylic acids often suffer from significant drawbacks that hinder commercial viability and process efficiency. Many conventional ester hydrolysis protocols require harsh acidic conditions or expensive enzymatic catalysts that can lead to incomplete conversion or the formation of difficult-to-remove byproducts. In the context of sulfonamide-containing molecules, there is a heightened risk of cleaving the sensitive sulfonamide bond if the reaction conditions are too aggressive, leading to degraded material and reduced overall yields. Furthermore, older methods frequently rely on organic solvents that are environmentally hazardous and difficult to recover, increasing the operational expenditure for waste treatment. The lack of precise temperature control in bulk reactions can also result in thermal runaways or inconsistent batch-to-batch reproducibility, which is unacceptable for GMP manufacturing of oncology intermediates. These limitations create bottlenecks in the supply chain, forcing procurement teams to deal with volatile pricing and extended lead times due to low throughput.
The Novel Approach
The methodology presented in patent CN102850244A introduces a streamlined and chemically elegant solution to these challenges by employing a mild alkaline hydrolysis technique. By utilizing readily available inorganic bases such as sodium hydroxide in a binary solvent system of alcohol and water, the process achieves high conversion rates under relatively gentle thermal conditions. This approach minimizes the risk of degrading the sensitive sulfonamide functionality while effectively cleaving the ester group to generate the free acid. The flexibility of the solvent system, allowing for the use of methanol or ethanol, provides manufacturers with the ability to select the most cost-effective and supply-secure solvent options based on regional availability. Moreover, the reaction can be driven to completion simply by monitoring via Thin Layer Chromatography (TLC), eliminating the need for complex in-process analytical technologies. This simplicity translates directly into operational efficiency, allowing for faster batch cycles and a more predictable manufacturing schedule for high-purity pharmaceutical intermediates.
Mechanistic Insights into Base-Catalyzed Ester Hydrolysis
The core chemical transformation described in this patent is a classic nucleophilic acyl substitution, specifically the saponification of an acrylate ester. In this mechanism, the hydroxide ion acts as a potent nucleophile, attacking the electrophilic carbonyl carbon of the ester group on the 3-(3-phenylsulfamoyl-phenyl)-acrylic acid R ester. This attack forms a tetrahedral intermediate, which subsequently collapses to expel the alkoxide leaving group (methoxide or ethoxide), resulting in the formation of the carboxylate salt. 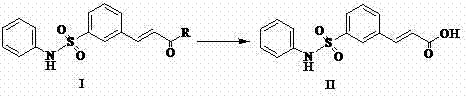 The use of a mixed solvent system is mechanistically significant; the alcohol component ensures the solubility of the organic ester substrate, while the water component facilitates the dissociation of the inorganic base and stabilizes the transition states through hydrogen bonding. Once the hydrolysis is complete, the reaction mixture contains the water-soluble sodium salt of the target acid. The subsequent acidification step with hydrochloric acid protonates the carboxylate anion, drastically reducing its solubility in the aqueous-alcoholic medium and causing the product to precipitate as a white solid. This precipitation mechanism serves as an intrinsic purification step, as many neutral organic impurities remain dissolved in the mother liquor, thereby enhancing the purity of the isolated cake without the need for chromatographic separation.
The use of a mixed solvent system is mechanistically significant; the alcohol component ensures the solubility of the organic ester substrate, while the water component facilitates the dissociation of the inorganic base and stabilizes the transition states through hydrogen bonding. Once the hydrolysis is complete, the reaction mixture contains the water-soluble sodium salt of the target acid. The subsequent acidification step with hydrochloric acid protonates the carboxylate anion, drastically reducing its solubility in the aqueous-alcoholic medium and causing the product to precipitate as a white solid. This precipitation mechanism serves as an intrinsic purification step, as many neutral organic impurities remain dissolved in the mother liquor, thereby enhancing the purity of the isolated cake without the need for chromatographic separation.
From an impurity control perspective, the patent emphasizes the importance of monitoring the reaction progress to prevent over-reaction or side reactions. While the sulfonamide bond is generally stable under basic conditions, prolonged exposure to high temperatures could theoretically lead to desulfonylation or other degradation pathways. By strictly controlling the temperature between 0°C and the reflux temperature of the solvent system, and preferably maintaining it around 60-70°C, the process maximizes the rate of hydrolysis while minimizing thermal stress on the molecule. The final isolation at pH 2-3 is also critical; this specific acidity range ensures complete protonation of the carboxylic acid without risking the hydrolysis of the amide bond in the final Belinostat structure if this intermediate were to be carried forward directly. This precise control over reaction parameters ensures that the resulting 3-(3-phenylsulfamoyl-phenyl)-acrylic acid meets the stringent purity profiles required for downstream coupling reactions in the synthesis of the final API.
How to Synthesize 3-(3-phenylsulfamoyl-phenyl)-acrylic acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this critical intermediate with high fidelity and yield. The process begins by charging a reactor with the ester starting material and a mixture of alcohol and water, creating a homogeneous solution ready for base addition. The careful addition of the alkaline solution allows for exotherm management, ensuring the reaction proceeds safely and controllably. Following the reaction period, the workup involves a simple concentration step to remove excess solvent, followed by pH adjustment to induce crystallization. This sequence of operations is designed to be easily transferable from laboratory glassware to large-scale stainless steel reactors, facilitating rapid technology transfer and commercialization.
- Dissolve 3-(3-phenylsulfamoyl-phenyl)-acrylic acid R ester in a mixed solvent system comprising an alcohol (methanol or ethanol) and water.
- Add an aqueous inorganic base solution, such as sodium hydroxide, dropwise while maintaining the temperature between 0°C and reflux conditions.
- Monitor reaction progress via TLC, then acidify the mixture to pH 2-3 to precipitate the pure product, followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hydrolysis method offers tangible benefits that extend beyond mere chemical yield. The reliance on commodity chemicals such as sodium hydroxide, hydrochloric acid, methanol, and ethanol significantly reduces the raw material cost profile compared to routes requiring specialized reagents or precious metal catalysts. This simplification of the bill of materials mitigates supply risk, as these inputs are globally available and less susceptible to market volatility. Furthermore, the elimination of complex purification steps like column chromatography reduces the consumption of silica gel and organic solvents, leading to substantial cost reduction in pharmaceutical intermediate manufacturing. The high yields reported in the examples, consistently exceeding 95%, mean that less starting material is wasted, directly improving the atom economy and lowering the cost of goods sold (COGS) for the final intermediate.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts or exotic reagents, relying instead on inexpensive inorganic bases and common solvents. This fundamental shift in reagent selection drastically lowers the direct material costs associated with production. Additionally, the simplified workup procedure, which relies on precipitation and filtration rather than energy-intensive distillation or chromatography, reduces utility consumption and labor hours per batch. These factors combine to create a highly cost-competitive manufacturing route that can withstand pricing pressure in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: By utilizing a robust chemical transformation that tolerates a range of temperatures and solvent ratios, the process offers greater operational flexibility. Manufacturers are not locked into a single, fragile set of conditions; if one solvent grade is unavailable, alternatives like switching between methanol and ethanol are feasible without re-validating the entire process. This flexibility ensures continuity of supply even during raw material shortages. The high purity of the precipitated product also reduces the likelihood of batch failures due to out-of-specification impurities, further stabilizing the supply chain for downstream API producers.
- Scalability and Environmental Compliance: The aqueous-alcoholic solvent system is inherently safer and more environmentally friendly than processes relying on chlorinated solvents or toxic organics. The ability to recover and recycle the alcohol solvent through distillation post-filtration aligns with green chemistry principles and reduces the environmental footprint of the manufacturing site. The scalability of the hydrolysis reaction is proven by its linear nature; heat transfer and mixing requirements do not become exponentially more difficult as the batch size increases from kilograms to tons. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates, ensuring that supply can meet global demand without requiring massive capital investment in new equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. Understanding these details is crucial for process engineers and quality assurance teams evaluating the feasibility of adopting this method for their own production lines. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable foundation for technical decision-making.
Q: What are the optimal reaction conditions for hydrolyzing the Belinostat ester intermediate?
A: The patent specifies using a mixed solvent of alcohol and water with an inorganic base like NaOH. The temperature can range from 0°C to reflux, with a preferred range of 60-70°C for optimal kinetics and yield.
Q: How is the purity of 3-(3-phenylsulfamoyl-phenyl)-acrylic acid ensured during isolation?
A: Purity is controlled by monitoring the reaction completion via Thin Layer Chromatography (TLC) to ensure full conversion of the starting ester. The product is isolated by adjusting the pH to 2-3 using hydrochloric acid, which precipitates the solid acid while leaving impurities in the supernatant.
Q: Can this synthesis method be scaled for commercial production?
A: Yes, the method utilizes commodity chemicals (NaOH, ethanol/methanol) and standard unit operations (hydrolysis, acidification, filtration), making it highly suitable for scale-up from laboratory to multi-ton commercial manufacturing without requiring exotic catalysts or complex purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(3-phenylsulfamoyl-phenyl)-acrylic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the consistent supply of high-quality intermediates is the backbone of successful drug development and commercialization. Our technical team has extensively analyzed the pathway described in CN102850244A and possesses the expertise to execute this hydrolysis with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive material that meets stringent purity specifications regardless of order volume. Our rigorous QC labs are equipped to detect and quantify trace impurities, guaranteeing that every batch of 3-(3-phenylsulfamoyl-phenyl)-acrylic acid we deliver supports the integrity of your final Belinostat API.
We invite global partners to leverage our manufacturing capabilities to secure their supply chains against disruption. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized processes can drive value and efficiency in your oncology drug portfolio.
