Scalable Synthesis of 1-Aminobenzo Imidazo Pyrazine Carboxamide for Immunotherapy
The pharmaceutical landscape is continuously evolving with the discovery of novel small molecule inhibitors that target specific immune checkpoints. Patent CN114276354B introduces a significant advancement in this field by disclosing a series of 1-aminobenzo[4,5]imidazo[1,2-a]pyrazine-3-carboxamide compounds. These molecules exhibit potent inhibitory activity against the adenosine A2A receptor, a critical target in tumor immunotherapy. By blocking the immunosuppressive signals mediated by extracellular adenosine in the tumor microenvironment, these compounds can effectively restore the function of immune cells such as T cells and NK cells. The structural novelty and high subtype selectivity of these inhibitors make them promising candidates for treating various cancers, including melanoma and lung carcinoma. This technical insight report analyzes the synthetic feasibility and commercial potential of this patented technology for global supply chains. ![General chemical structure of 1-aminobenzo[4,5]imidazo[1,2-a]pyrazine-3-carboxamide compounds showing variable R groups](/insights/img/1-aminobenzo-imidazo-pyrazine-supplier-pharma-20260315031548-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for complex heterocyclic compounds often suffer from significant drawbacks that hinder their commercial viability. Conventional methods frequently rely on harsh reaction conditions, such as extreme temperatures or pressures, which can compromise the stability of sensitive functional groups and lead to the formation of numerous by-products. Furthermore, many existing pathways require the use of expensive transition metal catalysts, which not only increase the raw material costs but also introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient. The removal of these metal residues often necessitates additional purification steps, such as chromatography or specialized scavenging treatments, which drastically reduce the overall yield and extend the production lead time. These inefficiencies create bottlenecks in the supply chain, making it difficult to secure a reliable supply of high-purity intermediates for clinical and commercial needs.
The Novel Approach
In contrast, the methodology described in patent CN114276354B offers a streamlined and efficient alternative that addresses these critical pain points. The new route utilizes cheap and easily obtainable starting materials, such as 1,2-phenylenediamine, and proceeds through a series of mild reaction steps that are highly conducive to scale-up. The process avoids the use of precious metal catalysts, thereby eliminating the need for complex metal removal procedures and significantly reducing the environmental footprint of the manufacturing process. The reaction conditions are carefully optimized to ensure high conversion rates and selectivity, resulting in a total yield and purity that meet stringent pharmaceutical standards. This approach not only simplifies the operational complexity but also enhances the robustness of the supply chain, ensuring consistent quality and availability of the target compounds for downstream drug development applications.
Mechanistic Insights into the Cyclization and Functionalization Strategy
The core of this synthetic strategy lies in the efficient construction of the benzo[4,5]imidazo[1,2-a]pyrazine scaffold. The initial step involves the cyclization of 1,2-phenylenediamine with methyl 2,2,2-trichloroimidoacetate in an acidic solvent, typically acetic acid, to form the benzimidazole intermediate. This reaction is conducted at controlled low temperatures to manage exothermicity and ensure high selectivity. Subsequent transformation of the trichloromethyl group into a cyano group is achieved using ammonia in dioxane under sealed conditions, a critical step that sets the stage for the formation of the pyrazine ring. The mildness of these conditions preserves the integrity of the molecular framework while introducing the necessary functionality for further elaboration. 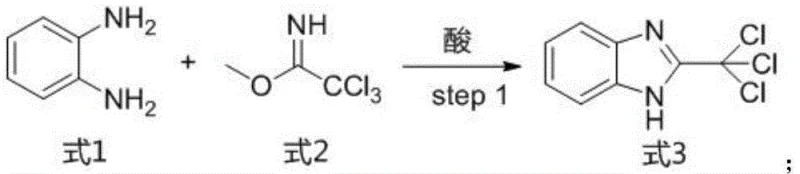
Further functionalization is achieved through a carefully orchestrated sequence of alkylation and cyclization reactions. The cyano intermediate is alkylated with 3-bromo-2-oxopropyl acetate in the presence of a base, followed by cyclization with ammonium acetate at elevated temperatures to close the pyrazine ring. This step is pivotal in establishing the core heterocyclic system with high fidelity. The final stages involve the introduction of diverse amide side chains through coupling reactions, allowing for the generation of a broad library of analogs with varying biological activities. The use of standard coupling reagents and mild deprotection conditions ensures that the final compounds are obtained with high purity and minimal impurity profiles, which is essential for their development as clinical candidates. 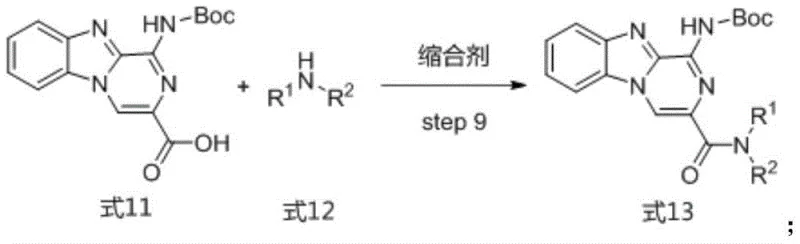
How to Synthesize 1-Aminobenzo[4,5]imidazo[1,2-a]pyrazine-3-carboxamide Efficiently
The synthesis of these high-value pharmaceutical intermediates requires precise control over reaction parameters to ensure optimal yield and purity. The patented process outlines a ten-step sequence that transforms simple starting materials into complex, biologically active molecules. Each step has been optimized to minimize waste and maximize efficiency, making it suitable for both laboratory-scale optimization and large-scale commercial production. The detailed standardized synthesis steps provided below offer a comprehensive guide for technical teams looking to implement this route, ensuring reproducibility and compliance with quality standards throughout the manufacturing process.
- Cyclization of 1,2-phenylenediamine with methyl 2,2,2-trichloroimidoacetate in acidic solvent to form the benzimidazole core.
- Conversion of the trichloromethyl group to a cyano group using ammonia in dioxane under sealed conditions.
- Alkylation with 3-bromo-2-oxopropyl acetate followed by cyclization with ammonium acetate to form the pyrazine ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement and supply chain management. The use of readily available and cost-effective raw materials significantly reduces the overall cost of goods sold, making the final product more competitive in the global market. The elimination of expensive catalysts and complex purification steps further contributes to cost savings, while the mild reaction conditions enhance operational safety and reduce energy consumption. These factors collectively improve the economic viability of the project, allowing for more flexible pricing strategies and better margin protection in a volatile market environment.
- Cost Reduction in Manufacturing: The process design inherently lowers production costs by avoiding the use of precious metal catalysts and reducing the number of purification steps required. This qualitative improvement in process efficiency translates directly into lower manufacturing expenses, as the need for specialized reagents and extensive waste treatment is minimized. The high yield and purity achieved at each step also reduce material loss, ensuring that a greater proportion of the input raw materials are converted into valuable product. This efficiency is crucial for maintaining profitability in the highly competitive pharmaceutical intermediate sector.
- Enhanced Supply Chain Reliability: The reliance on common, commercially available starting materials mitigates the risk of supply disruptions that often plague specialized chemical supply chains. By simplifying the sourcing strategy, manufacturers can secure a more stable and resilient supply of raw materials, ensuring continuous production even in the face of market fluctuations. The robustness of the synthetic route also means that production can be easily scaled up or down to meet changing demand without significant re-engineering of the process. This flexibility is a key asset for supply chain heads who need to manage inventory levels and lead times effectively.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metals make this process highly scalable and environmentally friendly. The reduction in hazardous waste generation simplifies compliance with increasingly stringent environmental regulations, reducing the burden on waste management systems. The ability to scale from tens of grams to multi-ton production levels without compromising quality ensures that the supply chain can support the transition from clinical trials to commercial launch seamlessly. This scalability is essential for meeting the long-term demand of global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these compounds. The answers are derived directly from the patent data and provide clarity on the feasibility and advantages of this technology. Understanding these details is crucial for stakeholders involved in the decision-making process for sourcing and development.
Q: What is the primary therapeutic application of these compounds?
A: These compounds act as potent adenosine A2A receptor inhibitors, designed for tumor immunotherapy by blocking immunosuppressive signals in the tumor microenvironment.
Q: How does this synthesis route improve manufacturing efficiency?
A:
Q: Is the process scalable for commercial production?
A: Yes, the patent explicitly supports scale-up from tens of grams to industrial levels, with high total yield and purity suitable for clinical candidate development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Aminobenzo[4,5]imidazo[1,2-a]pyrazine-3-carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a reliable partner for the production of complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in heterocyclic synthesis allows us to navigate the complexities of this patented route efficiently, providing you with a secure and high-quality supply chain solution.
We invite you to collaborate with us to optimize your supply chain and reduce costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities can support your drug development goals. Together, we can accelerate the delivery of life-saving therapies to patients worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
