Scalable Synthesis of 2-Methyl-3-Biphenylmethanol via Advanced Suzuki Coupling for Global Agrochemical Markets
Scalable Synthesis of 2-Methyl-3-Biphenylmethanol via Advanced Suzuki Coupling for Global Agrochemical Markets
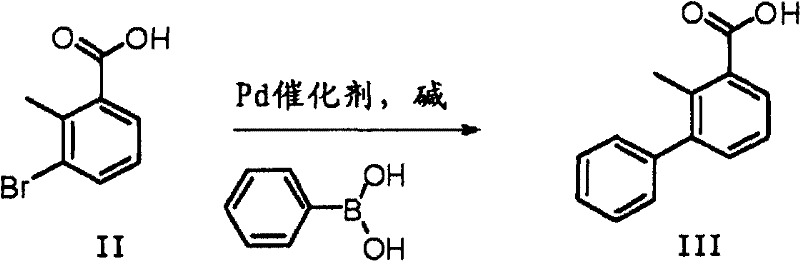
The global demand for high-efficiency pyrethroid insecticides continues to drive innovation in the synthesis of critical intermediates such as 2-methyl-3-biphenylmethanol. Patent CN102603485A introduces a robust and industrially viable preparation method that addresses longstanding challenges in the production of this key bifenthrin precursor. By leveraging a palladium-catalyzed Suzuki coupling reaction followed by a selective reduction step, this technology offers a pathway to high-purity intermediates with exceptional yield profiles. For R&D directors and procurement strategists in the agrochemical sector, understanding the nuances of this synthetic route is essential for optimizing supply chain resilience and reducing manufacturing costs. The method replaces hazardous and condition-sensitive traditional routes with a streamlined process that utilizes widely available starting materials like 3-bromo-2-methylbenzoic acid and phenylboronic acid derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biphenyl structures required for pyrethroid insecticides has relied heavily on classical coupling methodologies such as the Ullmann reaction, Wurtz coupling, or Grignard reagent formations. These traditional pathways are fraught with significant operational drawbacks that hinder efficient large-scale production. For instance, Grignard reactions necessitate strictly anhydrous conditions and the handling of highly reactive organomagnesium species, which pose substantial safety risks and require specialized equipment to prevent moisture ingress. Furthermore, Ullmann couplings often demand excessive temperatures and stoichiometric amounts of copper, leading to difficult purification processes and lower atom economy. The presence of severe reaction conditions in these legacy methods frequently results in the formation of complex impurity profiles, including homocoupling byproducts and dehalogenated species, which complicate downstream processing and reduce the overall purity of the final 2-methyl-3-biphenylmethanol product.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a Suzuki-Miyaura cross-coupling strategy that fundamentally shifts the paradigm towards milder and more controllable chemistry. This method employs 3-bromo-2-methylbenzoic acid and phenylboronic acid (or its esters/salts) as the primary building blocks, reacting them under the influence of a palladium catalyst in an alkaline environment. The reaction can be conducted at temperatures ranging from 10°C to 150°C, offering remarkable flexibility for process optimization. A distinct advantage of this route is the tolerance for various functional groups and the ability to operate in aqueous or mixed solvent systems, which drastically reduces the reliance on volatile organic compounds. Following the coupling, the resulting 3-phenyl-2-methylbenzoic acid is efficiently reduced to the target alcohol using agents like lithium aluminium hydride or borane. This two-step sequence not only simplifies the operational workflow but also ensures a cleaner reaction profile, making it ideally suited for the rigorous quality standards required in modern agrochemical manufacturing.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling and Reduction
The core of this synthetic breakthrough lies in the mechanistic efficiency of the palladium-catalyzed cycle. The reaction initiates with the oxidative addition of the aryl bromide (3-bromo-2-methylbenzoic acid) to the active palladium(0) species, forming an aryl-palladium(II) complex. This is followed by the transmetallation step, where the phenyl group from the boron species is transferred to the palladium center, a process facilitated by the presence of a base such as potassium carbonate or sodium phosphate which activates the boron atom. Finally, reductive elimination occurs to forge the new carbon-carbon bond, releasing the 3-phenyl-2-methylbenzoic acid product and regenerating the palladium catalyst for subsequent cycles. The patent highlights that heterogeneous catalysts like Palladium on Carbon (Pd/C) are particularly effective, likely due to their stability and ease of separation from the reaction mixture, which minimizes metal contamination in the final product.
Following the biaryl formation, the reduction mechanism involves the nucleophilic attack of hydride species on the carbonyl carbon of the carboxylic acid. When using lithium aluminium hydride (LiAlH4) or borane (BH3) in tetrahydrofuran (THF), the carboxyl group is converted directly to the primary alcohol functionality. The patent data suggests that controlling the temperature during this exothermic reduction—typically between -10°C and 50°C—is critical for maximizing yield and minimizing side reactions. The combination of a robust coupling step with a high-fidelity reduction ensures that the impurity spectrum is tightly controlled. By avoiding the harsh conditions of Grignard reagents, the formation of unwanted regioisomers or over-reduced byproducts is significantly suppressed, resulting in a crude product that requires less intensive purification to meet the stringent specifications demanded by formulators of bifenthrin insecticides.
How to Synthesize 2-Methyl-3-Biphenylmethanol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale execution, emphasizing the importance of catalyst selection and pH control. The process begins with the suspension of the aryl bromide and phenylboronic acid in a polar solvent system, where the pH is carefully adjusted to the 8-10 range using alkali metal carbonates or phosphates.
- Perform Suzuki coupling between 3-bromo-2-methylbenzoic acid and phenylboronic acid using a palladium catalyst (preferably Pd/C) in an alkaline aqueous or mixed solvent system at 25-100°C.
- Isolate the intermediate 3-phenyl-2-methylbenzoic acid through acidification and extraction.
- Reduce the carboxylic acid intermediate to 2-methyl-3-biphenylmethanol using lithium aluminium hydride or borane in THF under controlled temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this Suzuki-based methodology represents a strategic opportunity to optimize cost structures and enhance supply reliability. The shift away from moisture-sensitive reagents and cryogenic conditions inherently lowers the barrier to entry for manufacturing partners, allowing for a broader base of qualified suppliers. This diversification is crucial for mitigating supply chain risks associated with single-source dependencies on specialized organometallic reagents. Furthermore, the use of water-compatible solvent systems aligns with increasingly strict environmental regulations regarding VOC emissions, potentially reducing waste disposal costs and simplifying regulatory compliance audits. The robustness of the reaction conditions means that production schedules are less susceptible to delays caused by equipment failures related to extreme temperature or pressure requirements.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the utilization of cost-effective catalysts and solvents. The preference for Palladium on Carbon (Pd/C) over expensive homogeneous phosphine-ligand complexes significantly lowers the catalyst cost per kilogram of product. Additionally, the ability to use water as a co-solvent or primary solvent reduces the volume of expensive organic solvents required, leading to substantial savings in raw material procurement. The high yields reported in the patent examples, reaching up to 94% for the coupling step and 95% for the reduction, directly translate to improved material throughput and reduced waste generation, further enhancing the overall cost efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically 3-bromo-2-methylbenzoic acid and phenylboronic acid, are commodity chemicals with well-established global supply chains. Unlike specialized Grignard reagents which may have limited shelf-life and require cold chain logistics, these solid reagents are stable and easy to transport. This stability ensures consistent availability and reduces the risk of production stoppages due to raw material degradation. The simplified workup procedures, involving standard acidification and extraction, allow for faster batch turnover times, enabling manufacturers to respond more agilely to fluctuations in market demand for bifenthrin intermediates.
- Scalability and Environmental Compliance: From a scale-up perspective, the exothermic nature of the reactions is manageable within standard stainless steel reactors, eliminating the need for exotic metallurgy or specialized high-pressure vessels. The aqueous nature of the coupling step significantly reduces the fire hazard potential compared to processes relying on large volumes of flammable ethers or hydrocarbons. Moreover, the reduced generation of heavy metal waste (due to the efficiency of Pd/C) and the avoidance of stoichiometric magnesium or copper salts simplify effluent treatment processes. This environmental compatibility facilitates smoother permitting processes for capacity expansion and ensures long-term operational sustainability in regions with rigorous environmental oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making.
Q: What are the primary advantages of this Suzuki coupling route over traditional Grignard methods?
A: The Suzuki coupling route described in patent CN102603485A offers significantly milder reaction conditions compared to traditional Grignard or Ullmann couplings. It avoids the strict anhydrous requirements and hazardous handling associated with organometallic reagents, resulting in higher safety profiles and easier industrial scale-up.
Q: Which catalyst system provides the optimal yield for this transformation?
A: Experimental data indicates that Palladium on Carbon (Pd/C) serves as a highly effective catalyst, often outperforming homogeneous palladium complexes. Yields for the coupling step can reach up to 94% when using Pd/C with appropriate bases like potassium carbonate in mixed solvent systems.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is specifically designed for industrialization. It utilizes inexpensive and readily available raw materials, operates in aqueous or mixed solvent systems which reduces VOC emissions, and achieves high conversion rates, making it economically viable for multi-ton production of bifenthrin intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-3-Biphenylmethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy and safety of final agrochemical formulations. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 2-methyl-3-biphenylmethanol meets the exacting standards required for bifenthrin synthesis. Our commitment to process excellence allows us to deliver materials with consistent impurity profiles, supporting our partners in maintaining their own regulatory filings and product registrations globally.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive value and security for your agrochemical projects.
