Scalable Production of 5-Bromo-7-Azaindole via Sulfonate Protection Strategy
The pharmaceutical industry's relentless pursuit of novel kinase inhibitors has placed 5-bromo-7-azaindole in the spotlight as a critical building block for next-generation oncology therapeutics. As detailed in patent CN102584820A, a groundbreaking preparation method has emerged that fundamentally alters the economic and technical landscape for producing this valuable heterocyclic scaffold. Unlike traditional approaches that rely on costly transition metals or harsh halogenation conditions, this innovative protocol leverages a clever sulfonate protection strategy to achieve high regioselectivity and yield. For R&D directors and procurement specialists alike, understanding this shift is vital, as it promises not only superior purity profiles but also a drastic simplification of the supply chain for pharmaceutical intermediates. The method transforms 7-azaindole, a commercially accessible feedstock, into the target bromide through a sequence of aqueous-compatible reactions that minimize waste and maximize throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-bromo-7-azaindole has been plagued by significant technical hurdles that inflate costs and complicate scale-up. Conventional routes, such as those depicted in prior art references, often involve multi-step sequences requiring precious metal catalysts like palladium or copper for cross-coupling reactions. These methods, while chemically valid, introduce severe bottlenecks: the necessity for rigorous heavy metal scavenging to meet ppm-level specifications for API intermediates, the use of expensive ligands, and sensitivity to moisture and oxygen. Furthermore, direct bromination of 7-azaindole without protection often leads to poly-brominated byproducts or polymerization due to the high electron density of the pyrrole ring, resulting in difficult purification processes and suboptimal yields. The reliance on volatile organic solvents and cryogenic conditions in some legacy pathways further exacerbates safety concerns and energy consumption, making them less attractive for modern green chemistry mandates.
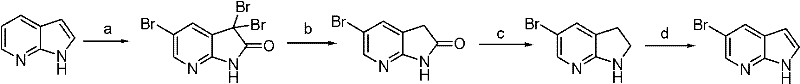
The Novel Approach
In stark contrast, the method disclosed in CN102584820A introduces a streamlined, three-step cascade that bypasses these pitfalls through intelligent molecular design. The core innovation lies in the initial reaction of 7-azaindole with sodium bisulfite to form a dihydro-sulfonate intermediate. This temporary modification effectively 'masks' the reactive pyrrole double bond, preventing unwanted side reactions during the subsequent bromination step. By controlling the electronic properties of the ring system, the process ensures that bromine attacks exclusively at the desired C5 position. Following bromination, a simple alkaline treatment restores the aromaticity of the pyrrole ring, releasing the final product with exceptional purity. This approach not only utilizes inexpensive, bulk chemicals like sodium bisulfite and bromine but also operates in mixed aqueous-organic systems, significantly reducing solvent costs and environmental impact compared to anhydrous organometallic routes.

Mechanistic Insights into Sulfonate-Mediated Electrophilic Substitution
The success of this synthesis hinges on the unique reactivity of the dihydro-7-azaindole-2-sodium sulfonate intermediate. Mechanistically, the addition of sodium bisulfite across the C2-C3 double bond of the 7-azaindole pyrrole ring converts the electron-rich five-membered ring into a saturated, electron-deficient system. This saturation is crucial because it dramatically lowers the nucleophilicity of the C2 and C3 positions, which are typically the most reactive sites for electrophilic attack in indoles and azaindoles. Consequently, when elemental bromine is introduced in the second step, the electrophile is forced to seek the next most electron-rich site, which is the C5 position on the pyridine ring. This electronic steering effect eliminates the formation of 2-bromo or 3-bromo isomers and prevents the formation of di-bromo impurities that commonly plague direct halogenation attempts. The use of hydrogen peroxide in this step serves a dual purpose: it helps manage the oxidation state and ensures the complete consumption of excess bisulfite, preventing the reduction of the added bromine.
Furthermore, the final desulfonation step under alkaline conditions is a thermodynamically driven restoration of aromaticity. The base facilitates the elimination of the sulfonate group, regenerating the C2-C3 double bond and restoring the fully conjugated 7-azaindole system. This step is remarkably clean, as the byproduct is simply sodium sulfite or sulfate, which remains in the aqueous phase during workup. From an impurity control perspective, this mechanism is superior because the intermediate sulfonate salts are often crystalline and water-soluble, allowing for easy purification via aqueous washes before the final cyclization. This inherent 'self-purifying' nature of the intermediate stages ensures that the final crude product already possesses high purity, reducing the burden on downstream recrystallization and chromatography, which are major cost drivers in fine chemical manufacturing.
How to Synthesize 5-Bromo-7-Azaindole Efficiently
The operational simplicity of this route makes it highly attractive for process chemists aiming to transfer technology from the lab to the pilot plant. The procedure begins with the suspension of 7-azaindole in a mixture of water and a water-miscible organic solvent such as ethanol or isopropanol. Sodium bisulfite is added, and the mixture is stirred at moderate temperatures (20-40°C) until the starting material is consumed, forming the soluble sulfonate salt. Without isolation, the solution is cooled, and bromine is added dropwise, often with a catalytic amount of hydrogen peroxide to maintain reaction efficiency. After the bromination is complete, verified by HPLC to ensure less than 0.2% of the intermediate remains, the reaction mixture is treated directly with a base like sodium hydroxide. The final product precipitates or can be extracted, followed by a standard recrystallization from toluene to achieve pharmaceutical-grade purity. Detailed standardized synthesis steps are provided in the guide below.
- React 7-azaindole with sodium bisulfite in a water/organic solvent system to generate dihydro-7-azaindole-2-sodium sulfonate.
- Perform a controlled bromination reaction on the sulfonate intermediate using bromine and hydrogen peroxide to form dihydro-5-bromo-7-azaindole-2-sodium sulfonate.
- Treat the brominated intermediate with an alkali base under mild heating to remove the sulfonate group and yield high-purity 5-bromo-7-azaindole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this sulfonate-based methodology represents a strategic opportunity to optimize the cost structure of oncology drug pipelines. By replacing noble metal catalysts with commodity chemicals, the direct material cost is substantially reduced, insulating the supply chain from the volatility of precious metal markets. Moreover, the elimination of complex metal scavenging steps shortens the overall cycle time, allowing for faster batch turnover and improved responsiveness to demand fluctuations. The use of aqueous systems also simplifies waste management, as the effluent contains primarily inorganic salts rather than toxic heavy metals or persistent organic pollutants, aligning with increasingly stringent environmental regulations and reducing disposal costs.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the complete avoidance of palladium, copper, or other transition metal catalysts, which are not only expensive to purchase but also costly to remove to trace levels required for GMP production. Additionally, the high atom economy of the bromination step and the ability to recycle solvents like ethanol or toluene contribute to a leaner cost profile. The process operates at near-ambient temperatures, eliminating the need for energy-intensive cryogenic cooling or high-pressure reactors, further driving down utility expenses and capital expenditure requirements for specialized equipment.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis, specifically 7-azaindole, sodium bisulfite, and bromine, are globally sourced commodities with robust supply chains, minimizing the risk of shortages that often plague specialty reagents. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, ensuring consistent batch-to-batch performance. This reliability is critical for maintaining continuous production schedules and meeting the tight deadlines associated with clinical trial material supply, thereby reducing the risk of project delays due to manufacturing bottlenecks.
- Scalability and Environmental Compliance: The chemistry is inherently scalable, having been demonstrated in multi-kilogram batches with consistent yields and purity. The aqueous workup procedures are easily adapted to large-scale reactor trains, and the absence of hazardous reagents like strong Lewis acids or pyrophoric organometallics enhances operational safety. From an environmental standpoint, the generation of benign inorganic byproducts simplifies wastewater treatment, facilitating compliance with green chemistry principles and reducing the regulatory burden associated with hazardous waste disposal, which is a key consideration for long-term sustainable manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 5-bromo-7-azaindole using this advanced sulfonate protection strategy. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, offering clarity on why this method is becoming the preferred choice for forward-thinking chemical manufacturers. Understanding these nuances helps stakeholders make informed decisions about supplier selection and process validation.
Q: What are the advantages of the sulfonate protection method over traditional palladium-catalyzed routes?
A: The sulfonate protection method eliminates the need for expensive transition metal catalysts like palladium or copper, significantly reducing raw material costs and simplifying the purification process by avoiding heavy metal residue removal.
Q: How does this process ensure high regioselectivity for the 5-position?
A: The formation of the dihydro-sulfonate intermediate temporarily saturates the C2-C3 double bond of the pyrrole ring, which deactivates these positions and directs the electrophilic bromine attack specifically to the C5 position of the pyridine ring.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process utilizes readily available raw materials like sodium bisulfite and operates under mild temperature conditions (20-40°C), making it highly scalable and safer for industrial manufacturing compared to methods requiring cryogenic temperatures or hazardous oxidants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromo-7-Azaindole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of your final therapeutic agent depends heavily on the integrity of its starting materials. As a premier CDMO partner, we have successfully integrated this efficient sulfonate-mediated synthesis into our production capabilities, ensuring a steady supply of high-purity 5-bromo-7-azaindole for global pharmaceutical clients. Our facilities boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We are committed to delivering intermediates that not only meet but exceed the demanding standards of the oncology sector.
We invite you to collaborate with us to leverage these technological advancements for your specific drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized process can lower your overall COGS. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with confidence and efficiency.
