Revolutionizing Trazodone Production: Continuous Flow Technology for Scalable API Intermediates
Revolutionizing Trazodone Production: Continuous Flow Technology for Scalable API Intermediates
The pharmaceutical industry is currently witnessing a paradigm shift from traditional batch processing to advanced continuous manufacturing technologies, particularly for high-volume antidepressant active pharmaceutical ingredients. Patent CN111886235A introduces a groundbreaking continuous process for the preparation of Trazodone and its hydrochloride salt, addressing critical limitations inherent in legacy synthetic routes. This innovation leverages flow reactor technology to execute key alkylation and coupling steps with unprecedented precision, transforming the production landscape for reliable pharmaceutical intermediates supplier networks globally. By transitioning to a continuous mode, manufacturers can achieve superior control over reaction parameters such as temperature and residence time, which are pivotal for managing the stability of sensitive alkyl halides. The methodology outlined in this patent not only enhances the safety profile by minimizing operator exposure to toxic reagents but also drastically improves the impurity profile of the final drug substance. As we delve deeper into the technical specifics, it becomes evident that this approach represents a significant leap forward in process intensification for complex organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional batch synthesis of Trazodone has long been plagued by inefficiencies that pose substantial challenges for large-scale commercial production and cost reduction in API manufacturing. Conventional methods typically involve prolonged reaction times extending over several hours, during which reactive intermediates are subjected to elevated temperatures in bulk quantities. This extended thermal stress often leads to the decomposition of reagents, particularly alkyl halides like 1-bromo-3-chloropropane, resulting in the formation of undesirable by-products that complicate downstream purification. Furthermore, the handling of large volumes of toxic alkylating agents in open or semi-open batch vessels presents significant occupational health and safety risks for plant personnel. The inability to rapidly dissipate heat in batch reactors can also lead to thermal runaways or inconsistent reaction kinetics, causing batch-to-batch variability in yield and purity. These factors collectively contribute to higher production costs, increased waste generation, and longer lead times, making the conventional approach less sustainable for modern pharmaceutical supply chains.
The Novel Approach
In stark contrast, the novel continuous process described in the patent utilizes flow chemistry to overcome these historical bottlenecks with remarkable efficacy. By confining the reaction to a micro-scale or meso-scale flow reactor, the system achieves exceptional heat and mass transfer rates that are unattainable in batch vessels. This allows for the precise maintenance of reaction temperatures between 90°C and 160°C for very short residence times, typically ranging from 70 to 180 seconds. Such rapid processing prevents the thermal degradation of sensitive intermediates, thereby preserving the integrity of the molecular structure and maximizing the conversion yield. The continuous nature of the process ensures that only minute quantities of hazardous reagents are present within the reactor at any given moment, fundamentally altering the safety dynamics of the operation. Moreover, the ability to seamlessly integrate multiple reaction steps, such as the initial alkylation and subsequent coupling, enables a telescoped synthesis that reduces solvent usage and intermediate isolation steps. This streamlined workflow not only accelerates production throughput but also aligns perfectly with the principles of green chemistry and sustainable manufacturing.
Mechanistic Insights into Flow Reactor Alkylation and Coupling
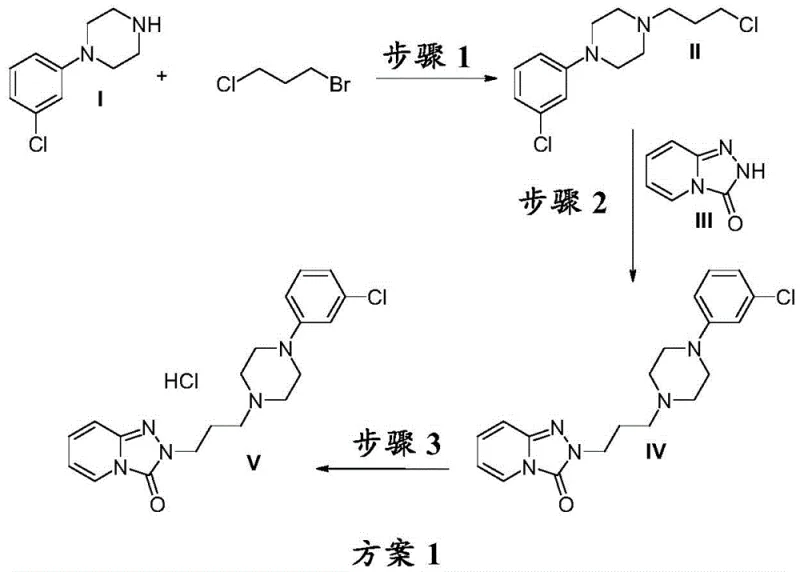
The core of this technological advancement lies in the sophisticated manipulation of biphasic reaction systems within a continuous flow environment. The synthesis begins with the alkylation of m-chlorophenylpiperazine using 1-bromo-3-chloropropane in the presence of an inorganic base such as sodium hydroxide. In a flow reactor, the aqueous alkaline phase and the organic phase containing the alkylating agent are mixed with high shear forces, creating a large interfacial area that facilitates rapid nucleophilic substitution. This efficient mixing is crucial because alkyl halides are notoriously unstable in alkaline aqueous solutions at elevated temperatures; however, the short residence time in the flow channel mitigates hydrolysis side reactions. Following the formation of the piperazine intermediate, the stream is directed into a second reaction zone where it encounters s-triazolo-[4,3-a]-pyridin-3-one. Here, the precise control of stoichiometry and temperature ensures that the coupling reaction proceeds to near-quantitative conversion without the accumulation of unreacted starting materials. The flow system's ability to maintain a steady state means that the concentration of reactants remains optimal throughout the reactor length, driving the equilibrium towards the desired Trazodone base product.
Impurity control is another critical aspect where the continuous mechanism excels, particularly regarding the suppression of genotoxic alkylating impurities. In batch processes, local hot spots or incomplete mixing can lead to over-alkylation or the persistence of unreacted alkyl halides, which are difficult to remove to trace levels. The plug-flow characteristic of the reactor ensures that every molecule experiences the same thermal history and reaction time, effectively eliminating the tailing effects seen in batch tanks. Consequently, the resulting Trazodone base exhibits an exceptionally clean impurity profile, with alkylated species reduced to levels below 2.5 ppm. This high level of purity is achieved directly from the reactor output, often negating the need for rigorous chromatographic purification steps that are both costly and time-consuming. The mechanistic efficiency of this flow process thus translates directly into a robust and reliable manufacturing protocol that meets stringent pharmacopoeial standards for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Trazodone Efficiently
The implementation of this continuous synthesis route requires a systematic approach to reactor configuration and parameter optimization to ensure consistent product quality. The process is generally divided into distinct stages where reagents are pumped through heated zones with specific residence times to facilitate the transformation from starting materials to the final hydrochloride salt. Operators must carefully balance the flow rates of the aqueous and organic streams to maintain the desired phase ratio, typically between 2:1 and 1:2, which is critical for effective mass transfer. Detailed standard operating procedures regarding pressure settings, back-pressure regulation, and quenching protocols are essential to maintain the stability of the flow system. For a comprehensive understanding of the specific operational parameters and equipment setup required to replicate this high-efficiency synthesis, please refer to the standardized guide below.
- Synthesize N-(3-chlorophenyl)-N'-(3-chloropropyl)-piperazine (II) by reacting m-chlorophenylpiperazine (I) with 1-bromo-3-chloropropane in a flow reactor at temperatures above 70°C.
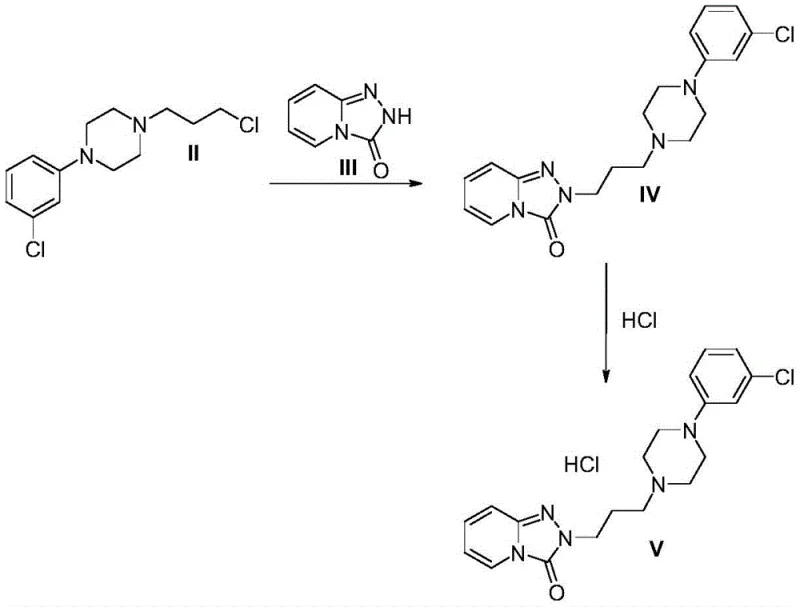
- React intermediate (II) with s-triazolo-[4,3-a]-pyridin-3-one (III) in a biphasic alkaline aqueous-organic system within a flow reactor at 90°C to 160°C to form Trazodone base (IV).
- Convert Trazodone base (IV) to Trazodone Hydrochloride (V) by treatment with hydrochloric acid in an organic solvent, followed by crystallization and filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this continuous flow technology offers transformative benefits that extend far beyond simple yield improvements. The shift from batch to flow fundamentally alters the cost structure of Trazodone production by enhancing process safety and reducing the burden of regulatory compliance associated with hazardous material handling. By minimizing the inventory of toxic alkylating agents on-site, facilities can lower their insurance premiums and reduce the capital expenditure required for specialized containment infrastructure. Furthermore, the drastic reduction in reaction time from hours to minutes significantly increases the asset utilization rate of the manufacturing plant, allowing for higher production volumes without the need for additional reactor capacity. This intensification of production capability ensures a more resilient supply chain that can respond rapidly to fluctuations in market demand for antidepressant medications. The elimination of extensive purification steps also reduces solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable manufacturing model.
- Cost Reduction in Manufacturing: The continuous process eliminates the need for expensive transition metal catalysts and reduces the reliance on energy-intensive long-duration heating cycles typical of batch reactions. By achieving high conversion rates in seconds rather than hours, the facility saves substantially on utility costs and labor hours per kilogram of product. The high purity of the crude output minimizes the loss of material during purification, leading to better overall mass balance and reduced raw material costs. Additionally, the simplified workflow reduces the number of unit operations, which lowers maintenance costs and decreases the likelihood of equipment downtime. These cumulative efficiencies result in a significantly lower cost of goods sold, providing a competitive edge in the global marketplace for generic pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: Continuous manufacturing systems are inherently more scalable and flexible than batch processes, allowing for seamless capacity expansion by running the reactor for longer durations or numbering up parallel units. This flexibility ensures a consistent and uninterrupted supply of high-purity pharmaceutical intermediates, mitigating the risk of stockouts that can disrupt downstream drug formulation schedules. The robust nature of the flow chemistry platform also reduces the variability between production runs, ensuring that every batch meets strict quality specifications without the need for extensive re-testing. This reliability fosters stronger partnerships between chemical suppliers and pharmaceutical companies, as it guarantees the timely delivery of critical materials needed for life-saving medications. Ultimately, the stability of the supply chain is reinforced by the technological superiority of the continuous process.
- Scalability and Environmental Compliance: The compact footprint of flow reactors allows for significant production capacity to be installed in existing facilities without major structural modifications, facilitating rapid scale-up from pilot to commercial production. The process generates significantly less chemical waste due to higher selectivity and reduced solvent usage, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The reduced exposure to hazardous chemicals also simplifies compliance with occupational safety standards, creating a safer working environment for employees. By adopting this green chemistry approach, manufacturers can future-proof their operations against evolving regulatory landscapes while demonstrating a commitment to environmental stewardship. This alignment with global sustainability trends enhances the brand reputation of the supplier and opens up new market opportunities with eco-conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous synthesis of Trazodone, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational advantages and quality benchmarks associated with this advanced manufacturing technique. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating flow chemistry into their existing supply chains. The answers provided reflect the proven capabilities of the technology as demonstrated in the patent examples.
Q: How does continuous flow chemistry improve the safety of Trazodone synthesis?
A: Continuous flow processes significantly reduce operator exposure to toxic alkylating agents like 1-bromo-3-chloropropane by minimizing the inventory of hazardous reagents held in the reactor at any given time, unlike traditional batch methods which require large volumes.
Q: What purity levels can be achieved with this continuous method?
A: The patented continuous method yields Trazodone Hydrochloride with extremely low levels of alkylated impurities, specifically below 2.5 ppm, and overall HPLC purity exceeding 99.5%, often eliminating the need for extensive downstream purification.
Q: Does this process require expensive transition metal catalysts?
A: No, the synthesis relies on efficient thermal activation and precise mixing in a flow reactor using inorganic bases like sodium hydroxide or potassium hydroxide, avoiding the cost and contamination risks associated with heavy metal catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trazodone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous flow technology in delivering high-quality pharmaceutical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN111886235A can be successfully translated into industrial reality. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every shipment of Trazodone meets the highest international standards. Our state-of-the-art facilities are equipped to handle complex continuous synthesis routes, providing our clients with a secure and efficient source of critical API intermediates. By leveraging our technical expertise, we help pharmaceutical companies accelerate their development timelines and bring essential medications to patients faster.
We invite you to contact our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project requirements. Request a Customized Cost-Saving Analysis today to understand the economic benefits of switching to our continuous flow-produced intermediates. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your needs. Let us partner with you to optimize your supply chain and drive innovation in antidepressant therapy manufacturing.
