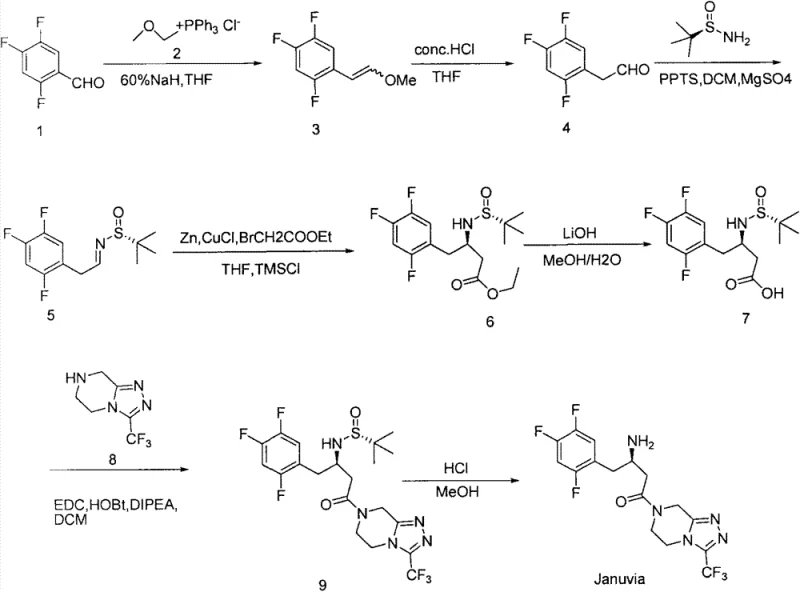
Advanced Synthetic Route for Sitagliptin: Enhancing Commercial Viability and Purity Standards
The global demand for antidiabetic medications continues to surge, placing immense pressure on supply chains to deliver high-quality Active Pharmaceutical Ingredients (APIs) like Sitagliptin efficiently. Patent CN103483340A discloses a robust and economically viable synthetic methodology that addresses the critical bottlenecks found in earlier generations of Sitagliptin manufacturing. By leveraging a chiral sulfinamide auxiliary strategy combined with a Reformatsky reaction, this process circumvents the reliance on costly precious metal catalysts and the inherent inefficiencies of racemic resolution. For pharmaceutical manufacturers and procurement strategists, this patent represents a pivotal shift towards a more sustainable and cost-effective production model that maintains rigorous purity standards while optimizing operational expenditures.
This technical insight report analyzes the mechanistic advantages and commercial implications of this novel route, specifically tailored for R&D directors seeking process robustness and supply chain leaders focused on continuity and cost reduction. The methodology transforms 2,4,5-trifluorobenzaldehyde into the final API through a sequence of high-yielding transformations that utilize commodity chemicals, thereby mitigating supply risks associated with specialized reagents. As we delve into the specific reaction parameters and scalability factors, it becomes evident that this approach offers a compelling alternative for the commercial scale-up of complex pharmaceutical intermediates, ensuring a reliable supply of this critical diabetes medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for Sitagliptin have historically relied heavily on asymmetric hydrogenation using rhodium or ruthenium complexes, or alternatively, classical chemical resolution techniques. While effective, asymmetric hydrogenation necessitates the use of extremely expensive chiral ligands and catalysts, which not only inflate raw material costs but also introduce significant challenges regarding residual metal removal to meet strict regulatory limits. Furthermore, chemical resolution methods are inherently inefficient, theoretically discarding fifty percent of the synthesized material as the unwanted enantiomer, which drastically reduces overall yield and increases waste disposal burdens. These legacy processes often require specialized high-pressure equipment and stringent safety protocols, creating substantial barriers to entry for contract development and manufacturing organizations (CDMOs) aiming to optimize their production lines for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in patent CN103483340A employs a chiral pool strategy using (R)-(+)-t-butyl sulfinamide, commonly known as Ellman's auxiliary, to induce stereochemistry during the carbon-carbon bond-forming step. This approach completely eliminates the need for transition metal hydrogenation catalysts, replacing them with inexpensive and readily available zinc and copper salts in a Reformatsky-type reaction. The process demonstrates exceptional diastereoselectivity, achieving a diastereomeric ratio greater than 98:2, which simplifies downstream purification and ensures high optical purity of the final product. By utilizing conventional reagents and avoiding high-pressure operations, this route significantly lowers the capital expenditure required for plant setup and reduces the operational complexity, making it an ideal candidate for a reliable sitagliptin intermediate supplier looking to maximize throughput.
Mechanistic Insights into Stereoselective Reformatsky Reaction
The cornerstone of this synthetic success lies in the stereoselective Reformatsky reaction where the N-tert-butanesulfinyl imine intermediate reacts with the zinc enolate derived from ethyl bromoacetate. The mechanism involves the coordination of the zinc atom to both the oxygen of the sulfinyl group and the nitrogen of the imine, forming a rigid, six-membered cyclic transition state. This chelation control effectively shields one face of the imine, forcing the nucleophilic attack of the enolate to occur exclusively from the less hindered face, thereby establishing the desired chiral center with high fidelity. The use of trimethylchlorosilane (TMSCl) and cuprous chloride further activates the zinc surface and facilitates the formation of the organozinc species, ensuring rapid reaction kinetics even at moderate temperatures ranging from 0 to 20 degrees Celsius.
Following the establishment of the chiral backbone, the synthesis proceeds through a series of robust functional group transformations including ester hydrolysis and amide coupling. The hydrolysis of the ethyl ester to the carboxylic acid is performed under mild basic conditions using lithium hydroxide, which prevents epimerization of the sensitive alpha-chiral center. Subsequent coupling with the triazolopyrazine fragment utilizes standard peptide coupling reagents such as EDC and HOBt, ensuring high conversion rates and minimal formation of deletion impurities. The final deprotection step employs methanolic hydrochloric acid to cleave the sulfinamide group, releasing the free amine and yielding the target molecule with excellent purity profiles suitable for direct formulation or salt formation.
How to Synthesize Sitagliptin Efficiently
The synthesis of Sitagliptin via this route is characterized by its operational simplicity and the use of standard laboratory and plant equipment, making it highly accessible for process chemists. The pathway begins with a Wittig olefination to install the vinyl ether moiety, followed by acidic hydrolysis to reveal the critical aldehyde functionality required for chiral induction. Detailed standardized operating procedures for each transformation, including precise stoichiometric ratios, temperature controls, and workup protocols, are essential for reproducing the high yields reported in the patent literature. For a comprehensive breakdown of the reaction conditions and purification methods required to achieve pharmaceutical-grade quality, please refer to the technical guide below.
- Perform Wittig reaction on 2,4,5-trifluorobenzaldehyde followed by acid hydrolysis to generate the key aldehyde intermediate.
- Condense the aldehyde with (R)-(+)-t-butyl sulfinamide and execute a stereoselective Reformatsky reaction with ethyl bromoacetate.
- Hydrolyze the ester to the corresponding acid, couple with the triazolopyrazine fragment, and perform final acid deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers transformative advantages by decoupling production from the volatility of the precious metals market. The substitution of expensive rhodium catalysts with commodity zinc and copper salts results in a drastic reduction in bill of materials (BOM) costs, allowing for more competitive pricing strategies in the generic API market. Additionally, the avoidance of high-pressure hydrogenation steps reduces the safety risks and insurance costs associated with manufacturing, while the use of common solvents like THF and DCM ensures that solvent recovery and recycling systems can be easily integrated into existing infrastructure. These factors collectively contribute to a more resilient supply chain capable of withstanding raw material fluctuations and meeting tight delivery schedules for high-purity sitagliptin.
- Cost Reduction in Manufacturing: The elimination of chiral transition metal catalysts removes a significant cost driver from the production budget, as these metals often account for a disproportionate share of raw material expenses. Furthermore, the high diastereoselectivity of the Reformatsky step negates the need for costly chiral chromatography or recrystallization steps typically required to upgrade optical purity, leading to substantial savings in both consumables and processing time. The overall yield improvement across the multi-step sequence ensures that less starting material is wasted, directly enhancing the cost-efficiency of the entire manufacturing campaign.
- Enhanced Supply Chain Reliability: By relying on widely available commodity chemicals such as ethyl bromoacetate, zinc powder, and 2,4,5-trifluorobenzaldehyde, manufacturers can mitigate the risk of supply disruptions caused by single-source dependencies on specialized reagents. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling production facilities to respond rapidly to changes in market demand without lengthy lead times for catalyst preparation or specialized equipment calibration. This flexibility is crucial for maintaining continuous supply to downstream formulation partners and ensuring reducing lead time for high-purity APIs.
- Scalability and Environmental Compliance: The process generates fewer hazardous waste streams compared to resolution-based methods, as there is no need to dispose of the unwanted enantiomer or spent chiral catalysts. The reaction conditions are mild and do not require extreme temperatures or pressures, simplifying the engineering controls needed for scale-up from pilot plant to commercial tonnage production. This alignment with green chemistry principles not only reduces environmental compliance costs but also enhances the corporate sustainability profile of the manufacturing entity, a key consideration for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route, derived directly from the patent specifications and process data. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of this supply source. The answers provided reflect the specific advantages of the Ellman auxiliary strategy over competing technologies in terms of purity, cost, and operational safety.
Q: How does this synthetic route improve upon traditional asymmetric hydrogenation methods?
A: This route utilizes an Ellman auxiliary strategy which avoids the need for expensive rhodium or ruthenium chiral catalysts required in asymmetric hydrogenation. Furthermore, it eliminates the 50% material loss inherent in classical chemical resolution methods, thereby significantly improving overall atom economy and cost efficiency.
Q: What represents the critical stereocontrolling step in this process?
A: The critical stereocontrolling step is the Reformatsky reaction involving the N-tert-butanesulfinyl imine intermediate. The bulky tert-butyl group on the sulfur atom creates a rigid chelated transition state with the zinc enolate, ensuring high diastereoselectivity (dr > 98:2) without requiring chiral chromatography.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly amenable to scale-up as it relies on conventional reagents like zinc powder, copper chloride, and standard organic solvents such as THF and DCM. It avoids high-pressure hydrogenation equipment and hazardous reagents, simplifying the engineering requirements for multi-ton production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Sitagliptin intermediate meets the highest global regulatory standards. We understand the critical nature of diabetes medication supply chains and are committed to delivering consistent quality and reliability to our international partners.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of adopting this methodology for your portfolio. Please contact us today to obtain specific COA data and route feasibility assessments, and let us demonstrate our capability as your trusted partner in pharmaceutical innovation.
