Advanced Synthesis of Fluoroalkylsulfonyl Fluorides for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking robust methodologies to access complex fluorinated scaffolds, particularly for kinase inhibitors where metabolic stability is paramount. Patent CN109134316B introduces a groundbreaking preparation method for fluoroalkylsulfonyl fluoride compounds that addresses critical limitations in current organofluorine chemistry. This technology enables the simultaneous introduction of fluoroalkyl and fluorosulfonyl groups onto alkene and alkyne substrates under remarkably mild conditions. The significance of this innovation lies in its application towards Janus kinase (JAK) inhibitors, a vital class of therapeutics for autoimmune diseases and cancers. By leveraging a radical addition mechanism mediated by transition metals and sulfur dioxide donors, this process bypasses the need for hazardous gaseous reagents while maintaining high functional group tolerance. For procurement and R&D teams, this represents a pivotal shift towards safer, more versatile synthetic routes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
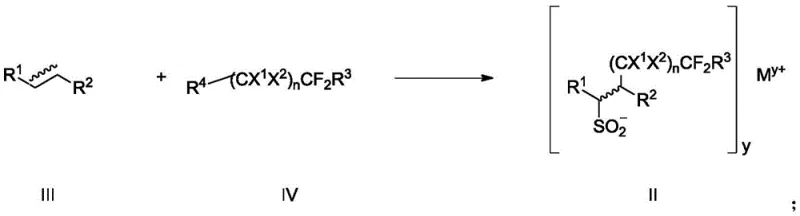
Historically, the introduction of fluorosulfonyl groups onto carbon skeletons has relied heavily on chloro-fluorine exchange reactions of sulfonyl chlorides or the 1,4-addition of vinylsulfonyl fluoride. These conventional pathways are fraught with significant operational hazards and chemical limitations. Vinylsulfonyl fluoride, a common reagent in these transformations, is highly volatile and exhibits substantial toxicity, posing severe risks during large-scale manufacturing and requiring specialized containment infrastructure. Furthermore, existing methods utilizing silver fluorosulfonyldifluoroacetate are often restricted to introducing only trifluoromethyl groups, severely limiting the structural diversity available to medicinal chemists. The sensitivity of functional groups to harsh reaction conditions in traditional protocols often necessitates extensive protection and deprotection sequences, driving up both material costs and production lead times. Additionally, the poor applicability of these older methods to aryl alkene and alkyne substrates restricts their utility in synthesizing complex drug-like molecules.
The Novel Approach
The methodology disclosed in CN109134316B offers a transformative solution by utilizing a radical cascade reaction that operates under inert gas protection with readily available metal sources such as zinc or silver fluoride. This novel approach eliminates the reliance on toxic vinylsulfonyl fluoride, replacing it with stable fluoroalkyl halides and solid sulfur dioxide donors like DABSO (DABCO·2SO2). The reaction conditions are exceptionally mild, typically proceeding at temperatures between -5°C and 60°C in water-miscible organic solvents like DMF or acetone. Crucially, this method is not limited to trifluoromethyl incorporation; it successfully accommodates a wide array of fluoroalkyl chains, including difluoroacetates and longer perfluoroalkyl segments. The compatibility with diverse substrates, including terminal alkenes, internal alkenes, and alkynes, ensures that this chemistry can be applied to a broad spectrum of pharmaceutical intermediates. This flexibility allows for the rapid generation of compound libraries for structure-activity relationship (SAR) studies without the bottleneck of specialized reagent synthesis.
Mechanistic Insights into Metal-Mediated Radical Fluorosulfonylation
The core of this synthetic breakthrough lies in a carefully orchestrated radical mechanism initiated by the single-electron transfer from the metal source to the fluoroalkyl halide. In the presence of a sulfur dioxide donor, the generated fluoroalkyl radical adds across the unsaturated bond of the alkene or alkyne substrate to form a carbon-centered radical intermediate. This intermediate subsequently traps the sulfur dioxide species, forming a sulfonyl radical that is stabilized by the metal cation to yield a fluoroalkyl sulfinate salt intermediate. The use of DABSO is particularly advantageous as it serves as a safe, solid surrogate for gaseous SO2, ensuring precise stoichiometric control and enhancing workplace safety. The reaction environment, often a mixture of polar aprotic solvents and water, facilitates the solubility of inorganic salts while maintaining the reactivity of the organic radicals. This mechanistic pathway avoids the high-energy barriers associated with nucleophilic substitutions on saturated carbons, allowing for the functionalization of unactivated olefins which are typically inert in other contexts.
Following the formation of the sulfinate intermediate, the process employs an electrophilic fluorination step to install the final fluorine atom on the sulfur center. Reagents such as N-fluorobisbenzenesulfonamide (NFSI) or Selectfluor act as the fluorine source, converting the sulfinate anion into the stable sulfonyl fluoride moiety. This two-step one-pot strategy minimizes purification losses and reduces solvent consumption, which is critical for green chemistry metrics. The mechanism ensures high regioselectivity, with the fluoroalkyl group adding to the less hindered position of the double bond in most cases, dictated by the stability of the radical intermediate. Impurity profiles are significantly cleaner compared to traditional methods because the radical pathway avoids side reactions like polymerization or elimination that plague ionic mechanisms. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the molar ratio of the SO2 donor to the substrate, to maximize yield and minimize waste generation.
How to Synthesize Fluoroalkylsulfonyl Fluoride Efficiently
Implementing this synthesis requires strict adherence to inert atmosphere techniques to prevent radical quenching by oxygen. The process begins with the activation of the metal source, typically zinc powder, in a solvent system comprising DMF and a small amount of water. The fluoroalkyl halide and alkene substrate are introduced sequentially or together, followed by the addition of the sulfur dioxide donor. Reaction progress is monitored via LC-MS or TLC, looking for the disappearance of the starting alkene. Once the sulfinate intermediate is formed, the electrophilic fluorinating agent is added directly to the reaction mixture without intermediate isolation. This telescoped procedure streamlines the workflow and reduces the exposure of potentially unstable intermediates to air. Detailed standardized operating procedures for scaling this reaction from gram to kilogram scale are essential for maintaining consistency and safety.
- React alkene or alkyne substrates with fluoroalkyl halides in the presence of a metal source (e.g., Zn) and SO2 donor (e.g., DABSO) under inert gas.
- Monitor the formation of the fluoroalkyl sulfinate intermediate using LC-MS or TLC until completion.
- Add an electrophilic fluorinating reagent such as NFSI to convert the intermediate into the final fluoroalkylsulfonyl fluoride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical efficiency. The elimination of toxic and volatile reagents like vinylsulfonyl fluoride drastically reduces the regulatory burden and safety compliance costs associated with manufacturing. By utilizing solid SO2 donors and common transition metals like zinc, the supply chain becomes more resilient against fluctuations in the availability of specialized gases or hazardous liquids. The mild reaction temperatures mean that standard glass-lined reactors can be used without the need for expensive cryogenic cooling systems or high-pressure vessels, leading to significant capital expenditure savings. Furthermore, the broad substrate scope implies that a single manufacturing line can be adapted to produce various analogues simply by changing the starting alkene or fluoroalkyl halide, enhancing asset utilization rates.
- Cost Reduction in Manufacturing: The process utilizes inexpensive and commercially available reagents such as zinc powder and DABSO, avoiding the need for costly noble metal catalysts or custom-synthesized organometallic reagents. The ability to run reactions in aqueous-compatible solvents reduces the volume of organic waste generated, lowering disposal costs and environmental fees. Since the reaction proceeds efficiently at near-ambient temperatures, energy consumption for heating or cooling is minimized compared to cryogenic processes. The high functional group tolerance reduces the number of synthetic steps required to reach the target molecule, directly correlating to lower raw material consumption and labor costs per kilogram of product.
- Enhanced Supply Chain Reliability: Sourcing stable solid reagents like DABSO and NFSI is far more reliable than managing the logistics of toxic gases or unstable liquids that require special transport permits. The robustness of the reaction against moisture and air (after initial setup) simplifies the operational requirements for contract manufacturing organizations (CMOs). This stability ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or out-of-specification results. The versatility of the method allows for rapid switching between different fluoroalkyl variants, enabling agile responses to changing market demands for specific JAK inhibitor intermediates without retooling entire production lines.
- Scalability and Environmental Compliance: The absence of heavy metal catalysts simplifies the downstream purification process, as there is no need for rigorous metal scavenging steps to meet strict pharmaceutical limits. The use of water as a co-solvent aligns with green chemistry principles, reducing the overall organic solvent load and facilitating easier wastewater treatment. The reaction generates minimal hazardous byproducts, making it easier to obtain environmental permits for new manufacturing facilities. Scalability is further supported by the exothermic nature of the radical addition being manageable under the described conditions, allowing for safe scale-up from pilot plant to commercial production volumes without significant engineering redesigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluoroalkylsulfonyl fluoride synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to ensure accuracy and reliability for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this chemistry into existing production pipelines.
Q: What are the key advantages of this synthesis method over traditional chloro-fluorine exchange?
A: This method avoids harsh conditions and toxic vinylsulfonyl fluoride reagents, offering better functional group compatibility and milder reaction temperatures ranging from -5°C to 60°C.
Q: Is this process scalable for commercial production of JAK inhibitor intermediates?
A: Yes, the use of commercially available reagents like DABSO and zinc powder, along with aqueous-compatible solvents, facilitates safe scale-up without requiring specialized high-pressure equipment.
Q: What types of fluoroalkyl groups can be introduced using this protocol?
A: Unlike prior art limited to trifluoromethyl groups, this protocol supports diverse fluoroalkyl chains including difluoroacetates and perfluoroalkyl groups, expanding the chemical space for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkylsulfonyl Fluoride Supplier
The technological advancements detailed in CN109134316B underscore the immense potential of fluoroalkylsulfonyl fluorides in modern drug discovery, particularly for oncology and immunology applications. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative academic and patent research into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to the manufacturing floor. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications required for clinical-grade intermediates. We understand the critical nature of supply continuity for life-saving medications and have established robust protocols to mitigate risks associated with raw material sourcing and process variability.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next-generation JAK inhibitor programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecular targets, demonstrating how this method can optimize your overall budget. Please contact our technical procurement team to request specific COA data for similar fluorinated intermediates and to discuss route feasibility assessments for your proprietary compounds. Together, we can accelerate the development of novel therapeutics while maintaining the highest standards of quality and efficiency.
