Advanced Synthesis of Piperazine Derivatives for CNS Drug Manufacturing and Commercial Scale-Up
Introduction to Novel Piperazine Synthesis Technologies
The pharmaceutical landscape for Central Nervous System (CNS) disorders continues to evolve, driven by the urgent need for effective treatments for drug addiction, schizophrenia, and Parkinson's disease. A pivotal development in this field is documented in Chinese patent CN102786497A, which discloses a robust preparation method for piperazine compounds and their critical intermediates. These molecules, exemplified by the dopamine D3 receptor partial agonist BP897, represent a cornerstone in modern neuropharmacology. The patent outlines a comprehensive strategy that moves beyond traditional limitations, offering a pathway to high-purity intermediates essential for drug discovery and development. By leveraging accessible starting materials and optimized reaction conditions, this technology addresses the growing demand for reliable pharmaceutical intermediate supplier capabilities in the CNS sector.
The structural versatility of these compounds is immense, covering a broad spectrum of substituted phenylpiperazines and spirocyclic derivatives. As illustrated in the foundational structures below, the core scaffold allows for significant modification to tune pharmacokinetic properties. This flexibility is crucial for medicinal chemists aiming to optimize potency and selectivity. The patent details the synthesis of numerous analogues, such as NGB2904, demonstrating the platform's adaptability. For procurement and supply chain leaders, understanding these synthetic routes is vital for securing long-term access to high-quality active pharmaceutical ingredients (APIs) and their precursors.
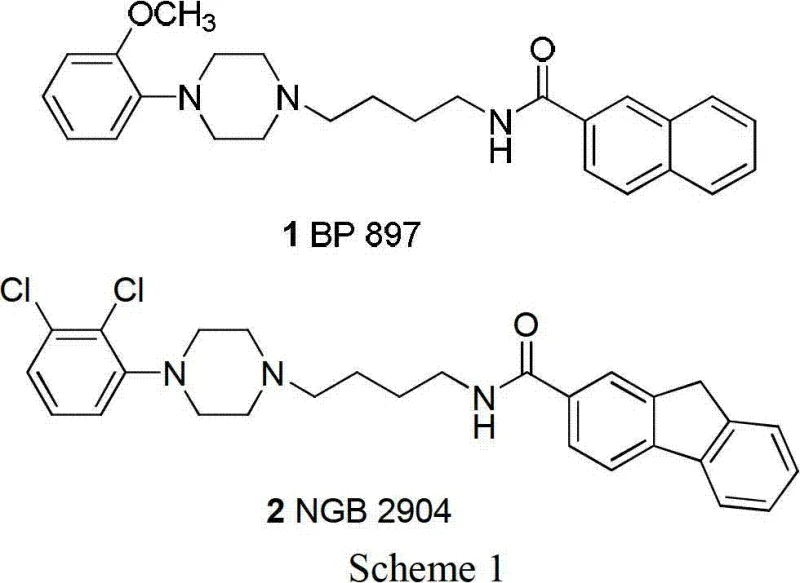
Furthermore, the economic implications of adopting this novel methodology cannot be overstated. Traditional methods often rely on expensive reagents and complex purification steps that inflate the cost of goods sold (COGS). In contrast, the approach detailed in CN102786497A emphasizes efficiency, utilizing common industrial solvents and catalysts. This shift not only reduces the environmental footprint through minimized waste generation but also enhances the overall sustainability of the manufacturing process. As the industry moves towards greener chemistry, such innovations provide a competitive edge for manufacturers seeking to align with global regulatory standards while maintaining profitability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key piperazine intermediates like those required for BP897 has relied on routes involving nitrile reduction. Prominent literature, such as work by Wenhua Chu et al., describes reacting o-methoxyphenylpiperazine with 4-bromobutyronitrile followed by reduction using lithium aluminum hydride (LiAlH4). While chemically feasible, this approach presents significant hurdles for large-scale production. LiAlH4 is a pyrophoric reagent that requires stringent safety protocols, inert atmospheres, and specialized handling equipment, thereby increasing operational risks and capital expenditure. Additionally, the workup for aluminum salts is notoriously tedious, generating substantial solid waste that complicates downstream processing and disposal.
Alternative conventional methods utilize catalytic hydrogenation with Raney Nickel to reduce the nitrile group. Although this avoids pyrophoric reagents, it introduces different challenges, primarily related to high-pressure hydrogen gas usage and catalyst sensitivity. These reactions often require elevated temperatures and pressures, necessitating heavy-duty reactor infrastructure. Moreover, the removal of trace nickel from the final product to meet pharmaceutical purity specifications can be difficult and costly. The combination of safety hazards, complex waste streams, and rigorous purification requirements makes these traditional pathways less attractive for cost reduction in API manufacturing, especially when scaling to commercial volumes.
The Novel Approach
The methodology presented in patent CN102786497A offers a transformative alternative by shifting the synthetic logic to a diethanolamine-based route. Instead of building the side chain onto an existing piperazine, this novel approach constructs the piperazine ring itself from readily available diethanolamine. The process begins with chlorination to form bis(2-chloroethyl)amine, which then undergoes cyclization with various substituted anilines. This strategy bypasses the need for hazardous nitrile reductions entirely. The reaction conditions are milder, typically operating at atmospheric pressure with standard heating, which significantly lowers the barrier for implementation in existing multipurpose facilities.
Moreover, this new route demonstrates exceptional tolerance for diverse functional groups, enabling the synthesis of a wide array of derivatives including halogenated, methoxylated, and nitro-substituted phenylpiperazines. The use of phase transfer catalysts, such as polyethylene glycols (PEG) or quaternary ammonium salts, further enhances reaction efficiency and yield. By simplifying the synthetic sequence and eliminating dangerous reagents, this approach facilitates the commercial scale-up of complex pharmaceutical intermediates. It represents a paradigm shift towards safer, more economical, and environmentally friendly production methods that align with modern green chemistry principles.
Mechanistic Insights into Diethanolamine Cyclization
The core of this innovative synthesis lies in the nucleophilic substitution and cyclization mechanism involving bis(2-chloroethyl)amine hydrochloride. Initially, diethanolamine is treated with thionyl chloride (SOCl2) to convert the hydroxyl groups into chlorides, yielding the reactive dichloro intermediate. This step is highly exothermic and requires careful temperature control, typically maintained below 30°C during addition, followed by heating to ensure complete conversion. The resulting bis(2-chloroethyl)amine acts as a potent electrophile, ready to engage with nucleophilic amines. When reacted with substituted anilines in the presence of a base, the nitrogen of the aniline attacks one of the chloroethyl arms, initiating the ring closure.
The cyclization step is critically dependent on the choice of solvent and acid-binding agent. The patent highlights the effectiveness of solvents like diethylene glycol monomethyl ether or acetonitrile, which provide the necessary polarity to dissolve reactants while stabilizing the transition state. Bases such as potassium carbonate or sodium hydroxide serve to neutralize the hydrochloric acid generated during the substitution, driving the equilibrium towards the product. The addition of phase transfer catalysts plays a pivotal role in facilitating the interaction between the organic substrates and the inorganic base, particularly in biphasic systems. This mechanistic optimization ensures high conversion rates and minimizes the formation of oligomeric by-products, which are common pitfalls in piperazine synthesis.
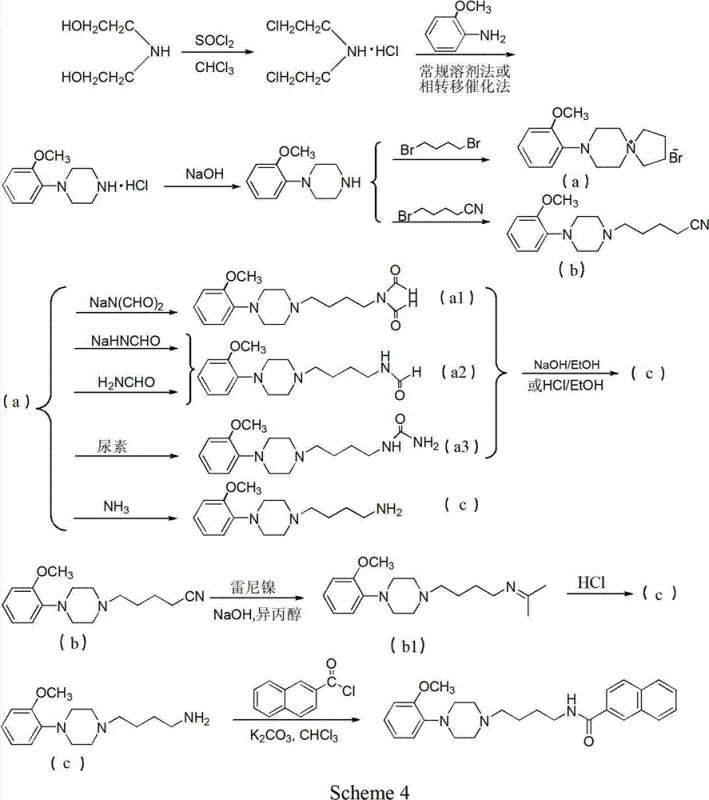
Following the formation of the piperazine ring, the resulting salt can be easily isolated and purified. The patent describes recrystallization techniques using solvents like isopropanol and n-hexane to achieve high purity levels. Subsequent neutralization with strong bases liberates the free piperazine base, which is then available for further functionalization, such as amidation to form the final drug candidates like BP897. This modular approach allows for the late-stage introduction of diverse acyl groups, providing medicinal chemists with the flexibility to explore structure-activity relationships (SAR) efficiently. The robustness of this mechanism underpins the reliability of the supply chain for these critical CNS drug intermediates.
How to Synthesize Substituted Phenylpiperazines Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process generally involves three main stages: chlorination of diethanolamine, cyclization with the chosen aniline, and final purification. Operators must adhere to strict temperature profiles, particularly during the thionyl chloride addition, to prevent side reactions. The selection of the appropriate phase transfer catalyst is also crucial, with PEG-400 or TBAB often providing optimal results depending on the specific aniline substrate. Detailed standard operating procedures (SOPs) should be established to manage the exothermic nature of the initial steps and ensure consistent batch-to-batch quality.
- Chlorinate diethanolamine using thionyl chloride to form bis(2-chloroethyl)amine hydrochloride.
- React the chlorinated intermediate with substituted anilines under basic conditions with phase transfer catalysts.
- Purify the resulting piperazine salt via recrystallization and neutralize to obtain the free base for further amidation.
For the final amidation step, converting the piperazine intermediate to the target amide, the patent recommends activating the carboxylic acid (e.g., 2-naphthoic acid) with thionyl chloride to form the acid chloride in situ. This reactive species is then coupled with the piperazine amine in the presence of a base like potassium carbonate. This acylation is typically high-yielding and straightforward, completing the synthesis of the target molecule. By following these optimized conditions, manufacturers can achieve yields exceeding 90% in the final coupling step, demonstrating the efficiency of the overall process.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift to this diethanolamine-based route offers substantial strategic benefits. The primary advantage lies in the availability and cost of raw materials. Diethanolamine and substituted anilines are commodity chemicals produced on a massive global scale, ensuring a stable supply chain不受 market fluctuations affecting specialized reagents. Unlike lithium aluminum hydride or Raney Nickel, which require specific sourcing and handling protocols, these inputs are readily accessible from multiple vendors. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages, enhancing overall supply chain resilience for critical pharmaceutical intermediates.
Operational safety and regulatory compliance are further enhanced by eliminating hazardous reagents. The removal of pyrophoric reducing agents and high-pressure hydrogenation steps significantly lowers the insurance and safety infrastructure costs associated with manufacturing. Facilities can operate with standard glass-lined or stainless steel reactors without the need for specialized high-pressure vessels. This reduction in capital intensity allows for more flexible production scheduling and faster turnaround times. Consequently, this leads to reduced lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands and clinical trial requirements without compromising on safety or quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reducing agents like LiAlH4 directly lowers reagent costs. Furthermore, the simplified workup procedures reduce solvent consumption and waste disposal fees. The high yields reported in the patent examples mean less raw material is wasted per kilogram of product, driving down the effective cost per unit. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: By relying on commodity starting materials, manufacturers mitigate the risk of supply disruptions. The robustness of the reaction conditions allows for production in a wider range of facilities, increasing the available capacity. This flexibility ensures that even during periods of high demand or logistical challenges, the flow of critical intermediates remains uninterrupted. Reliable sourcing is paramount for maintaining continuous clinical supply and meeting commercial launch timelines for new CNS therapies.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional metal-mediated reductions. The aqueous workups and standard organic solvents used are easier to treat and recycle. This alignment with green chemistry principles simplifies environmental permitting and reduces the carbon footprint of the manufacturing process. Scalability is inherent in the design, as the reaction kinetics and heat transfer characteristics are well-suited for large-scale batch reactors, facilitating a smooth transition from pilot plant to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of piperazine derivatives described in patent CN102786497A. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical teams evaluating this technology for adoption. Understanding these nuances is essential for making informed decisions about process integration and vendor qualification.
Q: What are the advantages of the diethanolamine route over traditional nitrile reduction?
A: The diethanolamine route avoids hazardous reducing agents like lithium aluminum hydride and high-pressure hydrogenation, offering safer operation and easier purification.
Q: Can this method be scaled for commercial production of BP897?
A: Yes, the process utilizes commodity chemicals and standard unit operations like reflux and filtration, making it highly suitable for multi-kilogram to ton-scale manufacturing.
Q: What types of substituents are compatible with this piperazine synthesis?
A: The method tolerates a wide range of substituents including methoxy, halogen, nitro, and trifluoromethoxy groups on the phenyl ring, allowing for diverse library generation.
For further technical details regarding specific impurity profiles or custom scale-up requirements, direct engagement with the technical team is recommended. Access to specific Certificate of Analysis (COA) data and route feasibility assessments can help tailor the manufacturing process to your specific project needs, ensuring alignment with your quality targets and timeline constraints.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BP897 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving CNS medications. Our expertise extends beyond simple supply; we act as a strategic partner in process optimization and scale-up. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the infrastructure and technical know-how to bring complex molecules like BP897 and its analogues to market efficiently. Our commitment to stringent purity specifications ensures that every batch meets the rigorous demands of global regulatory agencies.
We invite you to collaborate with our technical procurement team to explore how this advanced synthesis technology can benefit your pipeline. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages specific to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to validate the suitability of our processes for your projects. Together, we can accelerate the delivery of innovative therapies to patients while optimizing your supply chain performance.
