Advanced Resolution Technology for High-Purity S-3-Cyclohexene Carboxylic Acid Manufacturing
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has led to significant advancements in resolution technologies, particularly for critical anticoagulant precursors. Patent CN111099989B introduces a robust refining method for S-3-cyclohexene carboxylic acid, a pivotal building block in the synthesis of Edoxaban. This technical breakthrough addresses the longstanding challenge of separating enantiomers with similar physical-chemical properties, offering a pathway to achieve enantiomeric excess (ee) values exceeding 99%. For R&D directors and procurement specialists, understanding the nuances of this diastereomeric salt resolution process is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory standards. The methodology outlined in this patent not only ensures superior product quality but also integrates efficient solvent recovery systems that align with modern green chemistry principles.
In the competitive landscape of API manufacturing, the ability to consistently produce chiral acids with minimal impurities is a decisive factor for project success. The technology described herein leverages the specific interaction between S-3-cyclohexenecarboxylic acid and R-alpha-phenylethylamine to form a distinct diastereomeric salt. This salt exhibits differential solubility characteristics that can be exploited through controlled crystallization, effectively filtering out the unwanted R-enantiomer and other structural impurities. By adopting this refined approach, manufacturers can significantly mitigate the risks associated with batch-to-batch variability, ensuring that the final active pharmaceutical ingredient meets the rigorous purity profiles required by global health authorities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of single-enantiomer chiral compounds like S-3-cyclohexene carboxylic acid has relied on three primary strategies, each fraught with significant operational and economic drawbacks. The chiral pool synthesis method, while conceptually straightforward, is severely restricted by the limited availability and high cost of natural chiral starting materials, making it unsuitable for large-scale commercial production of diverse target molecules. Alternatively, asymmetric synthesis using catalysts or enzymes has gained traction; however, achieving high optical yields with an ee value greater than 90% often requires complex catalyst systems, stringent reaction conditions, and extensive downstream purification, which can drastically inflate manufacturing costs. Furthermore, biological asymmetric synthesis, despite its high selectivity, frequently suffers from slow reaction kinetics and difficult product separation protocols that hinder industrial scalability.
The third conventional avenue, racemate resolution via diastereomeric salts, has been the workhorse of the industry since Pasteur's era, yet traditional implementations often struggle with efficiency. Many existing resolution processes for S-3-cyclohexene carboxylic acid involve multiple influencing parameters that are difficult to control, resulting in suboptimal purification purity and poor recovery rates of the valuable resolving agent. When the resolving agent cannot be efficiently recycled, the cost of goods sold increases exponentially, rendering the process economically unviable for cost-sensitive generic drug markets. Additionally, conventional methods often fail to push the ee value beyond the 97% threshold without resorting to repetitive, yield-killing chromatographic separations, creating a bottleneck for high-purity pharmaceutical intermediates supply chains.
The Novel Approach
The innovative method disclosed in patent CN111099989B overcomes these historical limitations through a meticulously optimized three-step resolution protocol that balances purity, yield, and cost. By specifically selecting R-alpha-phenylethylamine as the resolving agent and utilizing ethyl acetate as the primary solvent for salt formation, the process creates a diastereomeric salt with ideal crystallization properties. The novelty lies in the precise thermal control during the reflux and cooling phases, where heating to 50°C–100°C followed by cooling to 20°C–40°C maximizes the formation of the target salt while minimizing the inclusion of impurities. This strategic manipulation of solubility curves allows for the direct isolation of high-quality intermediates from crude starting materials with an initial ee value of less than 97%.
Furthermore, this novel approach integrates a highly effective recrystallization loop using ethanol, which serves as a powerful polishing step to elevate the enantiomeric purity to over 99%. Unlike traditional methods that discard the mother liquor or the resolving agent, this process includes a dedicated dissociation and recovery phase. The use of dichloromethane and water for dissociation, followed by pH adjustment and extraction, ensures that the R-phenylethylamine is recovered and can be reused in subsequent batches. This closed-loop system not only reduces waste generation but also substantially lowers the raw material consumption per kilogram of product, offering a compelling value proposition for cost reduction in API manufacturing.
Mechanistic Insights into Diastereomeric Salt Resolution
The core of this purification technology relies on the fundamental principle of diastereomeric salt formation, where the chiral acid reacts with a chiral base to form two distinct salts with different physical properties. In this specific system, the S-enantiomer of the cyclohexene carboxylic acid reacts with the R-enantiomer of alpha-phenylethylamine to form the (S)-acid-(R)-base salt. This specific diastereomer possesses a lower solubility in ethyl acetate compared to the (R)-acid-(R)-base salt or other impurity complexes under the defined thermal conditions. As the solution is heated to reflux, all components dissolve, creating a homogeneous mixture; upon controlled cooling, the thermodynamically less soluble (S)-acid-(R)-base salt preferentially nucleates and grows into crystals, effectively excluding the unwanted R-enantiomer of the acid which remains in the mother liquor.
Impurity control is further enhanced through the iterative recrystallization process in ethanol. During this phase, the crude salt is dissolved at elevated temperatures (60°C–100°C) and then slowly cooled to induce crystallization. This step acts as a molecular sieve, where the crystal lattice of the desired diastereomer selectively incorporates the target molecules while rejecting structurally similar impurities and the minor enantiomer. By repeating this operation three times, as specified in the patent examples, the cumulative effect is a dramatic increase in optical purity. The final dissociation step involves breaking the ionic bond between the acid and the amine using hydrochloric acid in a biphasic system, releasing the free S-3-cyclohexene carboxylic acid into the organic phase while the amine salt remains in the aqueous phase for recovery.
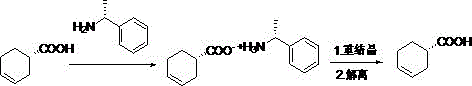
How to Synthesize S-3-Cyclohexene Carboxylic Acid Efficiently
Implementing this synthesis route requires precise adherence to the molar ratios and thermal profiles established in the patent data to ensure reproducibility at scale. The process begins with the careful mixing of the crude acid and the resolving agent in ethyl acetate, followed by a controlled reflux period to ensure complete salt formation before initiating the crystallization sequence. Operators must monitor the cooling rate closely during the filtration step to maximize the yield of the diastereomeric salt, as rapid cooling can trap impurities within the crystal matrix. The subsequent recrystallization in ethanol is the critical quality gate, where the number of cycles directly correlates with the final ee value, necessitating a balance between purity targets and overall process yield.
- Mix crude S-3-cyclohexenecarboxylic acid with R-alpha-phenylethylamine in ethyl acetate, reflux at 50-100°C, and cool to form the diastereomeric salt.
- Dissolve the obtained salt in ethanol at 60-100°C, cool to 30-50°C for crystallization, and repeat the recrystallization process three times to enhance purity.
- Treat the recrystallized salt with dichloromethane and water, adjust pH to 1-3 with hydrochloric acid to dissociate the acid, and recover the resolving agent from the aqueous layer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this refined resolution method translates into tangible strategic advantages regarding cost stability and supply continuity. The primary economic driver is the efficient recycling of the chiral resolving agent, R-phenylethylamine, which is typically a high-cost input in chiral synthesis. By recovering and reusing this amine through the alkaline extraction and distillation process described in the patent, manufacturers can drastically reduce the variable cost per unit of production. This reduction in raw material dependency insulates the supply chain from market volatility in chiral amine prices, ensuring more predictable budgeting and long-term cost reduction in pharmaceutical intermediate manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive chromatographic purification steps and the ability to recycle the resolving agent significantly lower the overall operational expenditure. Traditional methods often require multiple columns or complex enzymatic setups that drive up capital and operating costs; in contrast, this crystallization-based approach utilizes standard unit operations like filtration and distillation which are inherently cheaper to run. The high yield reported in the patent examples, ranging from 82% to 86%, further contributes to cost efficiency by maximizing the output from every kilogram of crude input, thereby reducing waste disposal costs and improving the overall atom economy of the process.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions, which utilize common industrial solvents like ethyl acetate, ethanol, and dichloromethane, ensures that the supply chain is not vulnerable to shortages of exotic reagents. Because the process does not rely on sensitive biological catalysts or rare transition metals, it can be easily scaled across multiple manufacturing sites without the need for specialized equipment or highly trained personnel. This robustness guarantees a consistent supply of high-purity S-3-cyclohexene carboxylic acid, reducing lead time for high-purity pharmaceutical intermediates and preventing production delays for downstream API manufacturers who depend on timely deliveries.
- Scalability and Environmental Compliance: The process is designed for commercial scale-up of complex chiral acids, utilizing equipment that is standard in any fine chemical facility, such as glass-lined reactors and centrifuges. The ability to recover solvents and the resolving agent minimizes the environmental footprint, aligning with increasingly strict global regulations on waste discharge and solvent emissions. This environmental compliance reduces the risk of regulatory shutdowns and facilitates smoother audits from international clients, making the manufacturer a preferred partner for sustainable pharmaceutical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production lines.
Q: What is the achievable enantiomeric excess (ee) using this resolution method?
A: According to patent CN111099989B, this specific refining method consistently achieves an ee value of greater than or equal to 99%, which is critical for the synthesis of the anticoagulant Edoxaban.
Q: Is the chiral resolving agent recyclable in this process?
A: Yes, the process includes a dedicated recovery step where the aqueous layer is treated with sodium hydroxide to adjust pH to 8-10, allowing the R-phenylethylamine to be extracted and recovered via distillation, significantly reducing raw material costs.
Q: What are the key solvents used in the purification cycle?
A: The process utilizes ethyl acetate for the initial salt formation, ethanol for the critical recrystallization steps to ensure high purity, and dichloromethane for the final dissociation and extraction phases.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-3-Cyclohexene Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation anticoagulants depends on the availability of ultra-high purity intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques such as chiral HPLC to verify that every batch of S-3-cyclohexene carboxylic acid meets the >99% ee requirement essential for Edoxaban synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this optimized resolution technology can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this recycling-enabled process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
