Advanced Asymmetric Conversion Technology for High-Purity L-2-Aminobutyric Acid Manufacturing
Advanced Asymmetric Conversion Technology for High-Purity L-2-Aminobutyric Acid Manufacturing
The pharmaceutical industry constantly seeks more efficient pathways for producing chiral amino acids, which serve as critical building blocks for modern therapeutics. A groundbreaking approach detailed in patent CN102060721A introduces a robust asymmetric conversion method for synthesizing L-2-aminobutyric acid, a key precursor for the antiepileptic drug Levetiracetam. Unlike conventional resolution techniques that are inherently capped at fifty percent theoretical yield, this innovative process leverages in-situ racemization coupled with diastereomeric crystallization to achieve yields exceeding eighty percent. For R&D directors and procurement specialists, this represents a paradigm shift in cost structure and material efficiency. By transforming the unwanted D-enantiomer back into the reactive pool, the method minimizes raw material waste and maximizes output per batch. This report analyzes the technical merits and commercial viability of this process, positioning it as a superior choice for reliable pharmaceutical intermediate supplier partnerships aiming for scalable and economical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure L-2-aminobutyric acid has relied heavily on classical chemical resolution, a process fraught with inherent economic inefficiencies. In traditional resolution, a racemic mixture of DL-2-aminobutyric acid reacts with a chiral resolving agent to form diastereomeric salts, which are then separated based on solubility differences. The fundamental flaw in this approach is the theoretical yield ceiling of fifty percent, as the unwanted D-enantiomer is typically discarded or requires complex, costly recycling steps to be useful. In practical industrial settings, losses during crystallization and washing often drive the actual total yield down to approximately thirty percent, resulting in significant raw material wastage. Furthermore, the separation efficiency is highly sensitive to temperature and solvent conditions, often leading to issues with optical purity where the unwanted enantiomer co-crystallizes, necessitating multiple recrystallization steps that further erode yield and increase processing time. These factors combine to create a high-cost manufacturing baseline that struggles to meet the demands of large-scale API production.
The Novel Approach
The asymmetric conversion method described in the patent data offers a transformative solution by integrating a dynamic racemization step directly into the resolution process. Instead of statically separating the enantiomers, this method employs an aromatic aldehyde catalyst to facilitate the continuous racemization of the amino acid in the reaction medium. As the desired L-2-aminobutyric acid forms an insoluble salt with D-tartaric acid and precipitates out of the solution, the equilibrium shifts, forcing the remaining D-enantiomer to racemize and eventually convert into the L-form to replenish the consumed species. This dynamic kinetic resolution effectively bypasses the fifty percent yield barrier, allowing for the conversion of nearly the entire starting material into the desired product. The process operates under relatively mild conditions using common acid solvents and achieves a stable yield improvement from the traditional thirty percent range to over eighty percent. This drastic increase in efficiency not only lowers the cost of goods sold but also simplifies the downstream purification requirements, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Aromatic Aldehyde-Catalyzed Asymmetric Conversion
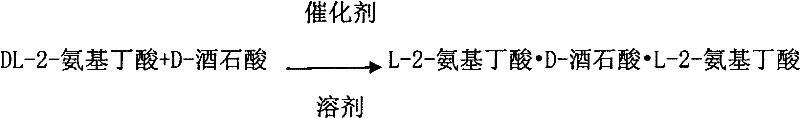
The core of this technology lies in the delicate interplay between catalytic racemization and selective crystallization thermodynamics. The reaction initiates with the mixing of DL-2-aminobutyric acid and D-tartaric acid in an acidic solvent, such as propionic acid or acetic acid, at elevated temperatures ranging from seventy to one hundred degrees Celsius. The addition of a catalytic amount of aromatic aldehyde, such as benzaldehyde or tolualdehyde, triggers the racemization of the amino acid via a Schiff base mechanism, although the exact transient intermediates are managed within the acidic milieu. As the reaction proceeds, the L-enantiomer selectively complexes with the D-tartaric acid to form a ternary salt, specifically L-2-aminobutyric acid•D-tartaric acid•L-2-aminobutyric acid, which exhibits extremely low solubility in the acidic solvent. Conversely, the salt formed by the D-enantiomer remains highly soluble. This solubility differential drives the precipitation of the L-complex, while the catalyst ensures that the D-enantiomer in the solution does not remain static but continuously racemizes to feed the formation of more L-product.
Following the isolation of the intermediate ternary salt, the process moves to the ammonolysis stage to liberate the free amino acid. This step relies on a straightforward acid-base neutralization mechanism where a stronger base displaces the weaker acid component of the salt. By treating the recrystallized L-2-aminobutyric acid•D-tartaric acid•L-2-aminobutyric acid complex with an ammonolysis agent such as ammonia gas, aqueous ammonia, or triethylamine in a solvent like methanol or water, the tartaric acid is converted into a soluble ammonium tartrate salt. The L-2-aminobutyric acid, being less soluble in the alcoholic or aqueous medium under the specific cooling conditions, precipitates as a high-purity solid. This mechanism avoids the use of harsh mineral acids or bases that could degrade the chiral center, ensuring the optical integrity of the final product is maintained throughout the transformation. The simplicity of this neutralization step allows for easy filtration and washing, yielding a product with chemical purity exceeding ninety-nine percent and high optical rotation values consistent with pharmacopeial standards.

How to Synthesize L-2-Aminobutyric Acid Efficiently
Implementing this synthesis route requires precise control over stoichiometry and temperature profiles to maximize the efficiency of the asymmetric conversion. The process begins by dispersing DL-2-aminobutyric acid and D-tartaric acid in a molar ratio between two-to-one and three-to-one within an acid solvent, followed by the addition of one to five percent aromatic aldehyde catalyst. The mixture is heated to promote the reaction and subsequent crystallization of the intermediate complex, which is then filtered and recrystallized from water to ensure high optical purity. The purified intermediate is subsequently subjected to ammonolysis in an alcohol or water solvent at ambient temperature, followed by cooling to induce precipitation of the final product. For detailed operational parameters, safety guidelines, and specific workup procedures required for GMP compliance, please refer to the standardized synthesis protocol below.
- Mix DL-2-aminobutyric acid and D-tartaric acid in a molar ratio of 2: 1 to 3:1 within an acidic solvent such as propionic acid or acetic acid.
- Add an aromatic aldehyde catalyst like benzaldehyde and react at 70 to 100 degrees Celsius for 6 to 8 hours to form the intermediate salt.
- Perform ammonolysis on the recrystallized intermediate using ammonia or triethylamine in alcohol or water to release the final L-2-aminobutyric acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this asymmetric conversion technology offers profound advantages over legacy resolution methods. The primary driver of value is the substantial increase in yield, which directly translates to a significant reduction in the consumption of raw materials per kilogram of finished product. By effectively utilizing both enantiomers of the starting racemate through in-situ conversion, manufacturers can drastically lower their dependency on bulk DL-2-aminobutyric acid, mitigating exposure to raw material price volatility. Furthermore, the process eliminates the need for expensive transition metal catalysts often found in enzymatic or hydrogenation routes, relying instead on inexpensive and readily available aromatic aldehydes. This simplification of the catalyst system reduces both the direct material cost and the complexity of heavy metal removal steps, streamlining the quality control workflow and reducing the burden on analytical laboratories.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the elimination of the fifty percent yield penalty associated with classical resolution. By achieving yields consistently above eighty percent, the effective cost per unit of active pharmaceutical ingredient is significantly lowered without compromising quality. The ability to recycle the mother liquor from the filtration step back into the reactor further enhances material efficiency, minimizing waste disposal costs and solvent consumption. Additionally, the use of common organic acids and alcohols as solvents ensures that procurement teams can source materials from a broad supplier base, avoiding single-source bottlenecks and leveraging competitive pricing for commodity chemicals.
- Enhanced Supply Chain Reliability: The robustness of this chemical process contributes to greater supply chain stability and predictability. The reaction conditions are not overly sensitive to minor fluctuations, allowing for consistent batch-to-batch performance which is critical for maintaining uninterrupted API production schedules. The starting materials, including D-tartaric acid and DL-2-aminobutyric acid, are commercially available in large quantities from established chemical producers, ensuring a secure supply line. Moreover, the simplified downstream processing, which involves standard filtration and washing rather than complex chromatography or distillation, reduces the risk of equipment downtime and accelerates the overall manufacturing cycle time, enabling faster response to market demand surges.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is straightforward due to the reliance on unit operations that are well-understood in the fine chemical industry, such as crystallization and filtration. The process design inherently supports green chemistry principles by reducing the generation of unwanted enantiomeric waste, thereby lowering the environmental footprint of the manufacturing site. The option to use water as a solvent in the ammonolysis step further aligns with sustainability goals by reducing the volume of volatile organic compounds released. This environmental compatibility simplifies regulatory compliance and waste treatment permitting, making it an attractive option for facilities operating under strict environmental regulations while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this asymmetric conversion technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the method's suitability for your specific production needs. Understanding these nuances is essential for R&D teams planning technology transfer and for procurement officers assessing the long-term viability of the supply chain. We encourage stakeholders to review these details to fully appreciate the operational flexibility and quality advantages offered by this novel synthetic route.
Q: How does this asymmetric conversion method improve yield compared to traditional resolution?
A: Traditional chemical resolution is theoretically limited to a 50 percent yield because it discards the unwanted enantiomer. This asymmetric conversion method utilizes a racemization catalyst to continuously convert the unwanted D-enantiomer into the L-form in situ, driving the theoretical yield significantly higher, often exceeding 80 percent in practice.
Q: What catalysts are suitable for this racemization process?
A: The patent specifies the use of aromatic aldehydes as effective catalysts for the racemization step. Specific examples include benzaldehyde, tolualdehyde, and 2-hydroxy naphthaldehyde, which facilitate the equilibrium shift without requiring expensive transition metals.
Q: Can the solvents be recycled to reduce environmental impact?
A: Yes, the process is designed for sustainability. The mother liquor from the filtration step, which contains unreacted materials and solvent, can be recycled directly back into the reaction system. Additionally, the ammonolysis step allows for the use of water or common alcohols, simplifying waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-2-Aminobutyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient chiral synthesis in the modern pharmaceutical landscape. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and robust. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of complex chiral amino acids or large-scale supply of established intermediates like L-2-aminobutyric acid, our infrastructure is designed to support your growth with reliability and precision.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced process chemistry. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your project's success and ensure a steady flow of high-quality materials for your global operations.
