Advanced Metal-Free Synthesis of S-Aryl Isothiourea Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds, particularly those containing sulfur-nitrogen motifs which are prevalent in bioactive molecules and organocatalysts. Patent CN113735752A, published in late 2021, introduces a groundbreaking metal-free strategy for the preparation of S-aryl isothiourea compounds, addressing critical bottlenecks in traditional synthetic routes. This innovation utilizes substituted iodobenzene and thiourea derivatives as primary substrates, reacting them in the presence of a metal hydride base to achieve direct C-S bond formation. Unlike conventional methods that rely heavily on transition metal catalysis, this approach operates under mild conditions, typically at room temperature, thereby significantly reducing energy consumption and operational complexity. For R&D directors and process chemists, this represents a paradigm shift towards greener, more efficient synthesis of valuable building blocks used in drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-S bonds to form S-aryl isothioureas has been dominated by transition metal-catalyzed cross-coupling reactions, such as the Chan-Lam coupling or copper-catalyzed protocols. While effective in many contexts, these traditional methodologies suffer from inherent and often severe limitations that hinder their industrial applicability. A primary concern is the phenomenon of catalyst poisoning; sulfur-containing species are notorious for their ability to rapidly and irreversibly deactivate various types of metal catalysts, necessitating high catalyst loading capacities to drive the reaction to completion. Furthermore, these processes often require harsh reaction conditions, including elevated temperatures, strong bases, and air-sensitive environments, which complicate process safety and increase operational costs. The reliance on expensive transition metals like copper or palladium also introduces significant downstream purification challenges, as removing trace metal residues to meet stringent pharmaceutical purity standards adds multiple costly steps to the manufacturing workflow.
The Novel Approach
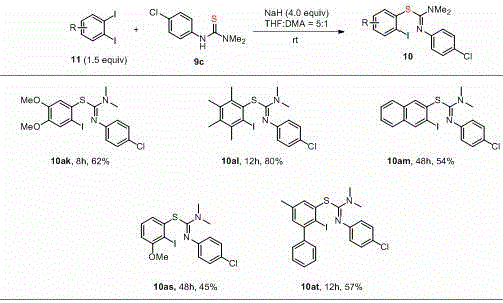
In stark contrast to the complexities of metal catalysis, the method disclosed in CN113735752A offers a streamlined, metal-free alternative that leverages nucleophilic addition chemistry. By employing sodium hydride (NaH) as a promoter in a mixed solvent system of THF and DMA, the process facilitates the direct coupling of substituted iodobenzenes with thiourea derivatives without the need for any transition metal catalyst. This novel approach not only circumvents the issue of catalyst deactivation but also allows the reaction to proceed efficiently at room temperature over a period of 8 to 50 hours. The simplicity of the reagent system—using cheap and easily obtained raw materials like NaH and commercially available iodobenzenes—drastically simplifies the workup procedure. As illustrated in the reaction scope below, this method exhibits remarkable versatility, successfully synthesizing various S-(substituted iodoaryl) isothiourea derivatives with good yields and high regioselectivity, even for sterically hindered or electronically diverse substrates.
Mechanistic Insights into NaH-Promoted Nucleophilic Substitution
The core mechanistic advantage of this technology lies in the activation of the thiourea nucleophile through deprotonation by the strong base, sodium hydride. In the absence of a metal catalyst to lower the activation energy for oxidative addition, the reaction relies on the intrinsic nucleophilicity of the sulfur atom in the thiourea anion. The base abstracts a proton from the thiourea, generating a highly reactive thiolate species that attacks the electron-deficient carbon of the aryl iodide. This nucleophilic aromatic substitution pathway is particularly effective because the iodine atom serves as an excellent leaving group, facilitating the displacement even under mild thermal conditions. The patent data indicates that the reaction possesses excellent functional group tolerance, meaning that sensitive moieties such as methoxy groups, halogens, and fused aromatic systems remain intact during the transformation. This selectivity is crucial for multi-step synthesis where protecting group strategies add unnecessary cost and time.
Furthermore, the process demonstrates impressive regioselectivity, particularly when dealing with poly-halogenated substrates like ortho-substituted diiodobenzenes. The ability to control the site of C-S bond formation ensures that the desired isomer is produced as the single major product, minimizing the formation of impurities that would otherwise complicate purification. The structural diversity of the substrates is well-defined, as shown in the general formula where R can be selected from halogens, alkyl, alkoxy, or phenyl groups. This broad substrate scope confirms that the electronic and steric properties of the aromatic ring do not prohibit the reaction, making it a universally applicable tool for synthesizing complex isothiourea libraries. The elimination of metal-ligand complexes also means there are no competing coordination equilibria to manage, resulting in a more predictable and reproducible reaction profile suitable for large-scale manufacturing.
How to Synthesize S-Aryl Isothiourea Efficiently
Implementing this metal-free synthesis in a laboratory or pilot plant setting requires careful attention to the handling of reactive hydrides and the maintenance of anhydrous conditions to ensure optimal yields. The general protocol involves suspending the base in anhydrous tetrahydrofuran, followed by the controlled addition of the thiourea derivative and the iodobenzene substrate. The reaction mixture is then stirred at ambient temperature, allowing the nucleophilic substitution to proceed to completion as monitored by thin-layer chromatography. Detailed standardized operating procedures regarding stoichiometry, solvent ratios, and quenching methods are essential for reproducibility. For a comprehensive, step-by-step technical guide on executing this synthesis safely and effectively, please refer to the standardized protocol section below.
- Prepare the reaction system by suspending sodium hydride (NaH) in anhydrous THF under inert atmosphere protection.
- Add the thiourea derivative dissolved in DMA dropwise to the base suspension, followed by the addition of the substituted iodobenzene substrate.
- Stir the mixture at room temperature for 8 to 50 hours, monitor by TLC, and quench with ice water followed by standard extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this metal-free methodology offers transformative benefits that directly impact the bottom line and operational resilience. The most immediate advantage is the drastic reduction in raw material costs associated with eliminating expensive transition metal catalysts and specialized ligands. Traditional copper or palladium-catalyzed processes often require precious metals that are subject to volatile market pricing and supply constraints; by replacing these with commodity chemicals like sodium hydride, manufacturers can achieve substantial cost savings and insulate themselves from supply chain shocks. Additionally, the simplified reaction conditions reduce the need for specialized equipment capable of handling high temperatures or rigorous air-free environments, further lowering capital expenditure requirements for production facilities.
- Cost Reduction in Manufacturing: The economic implications of removing transition metals from the process extend beyond just the price of the reagents. In pharmaceutical manufacturing, the removal of heavy metal residues is a mandatory and costly step, often requiring specialized scavengers or extensive chromatography to meet regulatory limits. By utilizing a metal-free route, the downstream purification process is significantly streamlined, reducing solvent consumption, waste generation, and processing time. This leads to a leaner manufacturing process with higher overall throughput and lower cost of goods sold (COGS), making the final isothiourea intermediates more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain stability is paramount for continuous API production, and reliance on scarce catalytic metals poses a significant risk. Sodium hydride and substituted iodobenzenes are widely available commodity chemicals with robust global supply networks, ensuring consistent availability and shorter lead times compared to specialized catalysts. Furthermore, the mild reaction conditions reduce the risk of batch failures due to thermal runaway or sensitivity issues, enhancing the reliability of production schedules. This stability allows procurement managers to negotiate better terms with suppliers and maintain lower safety stock levels, optimizing working capital.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often exposes hidden inefficiencies, particularly those related to heat management and waste disposal. This room-temperature process inherently minimizes energy consumption for heating and cooling, aligning with green chemistry principles and reducing the facility's carbon footprint. The absence of heavy metal waste also simplifies environmental compliance and waste treatment protocols, avoiding the high costs associated with hazardous waste disposal. These factors combined make the technology highly scalable, enabling seamless transition from kilogram-scale development to multi-ton commercial production without the need for major process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-free isothiourea synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction scope, safety, and scalability. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the metal-free approach superior for synthesizing sulfur-containing heterocycles?
A: Traditional metal-catalyzed C-S coupling often suffers from catalyst poisoning because sulfur species irreversibly deactivate transition metals like copper or palladium. This metal-free method eliminates that risk entirely.
Q: What are the typical reaction conditions for this isothiourea synthesis?
A: The reaction proceeds efficiently at room temperature using sodium hydride as a base in a THF/DMA solvent system, avoiding the high temperatures and harsh ligands required by conventional Chan-Lam couplings.
Q: Does this method support diverse functional groups on the benzene ring?
A: Yes, the patent demonstrates excellent functional group tolerance, successfully accommodating halogens, alkoxy groups, and fused ring systems like naphthalene without compromising yield or selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Aryl Isothiourea Supplier
As the demand for high-quality heterocyclic building blocks continues to grow in the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO is essential for successful project execution. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of S-aryl isothiourea intermediates we produce. We understand the critical nature of supply continuity and are committed to delivering consistent quality that meets the exacting standards of the global life sciences industry.
We invite you to collaborate with our technical team to explore how this innovative metal-free synthesis can optimize your specific project requirements. By leveraging our expertise in process optimization and scale-up, we can help you realize the full commercial potential of this technology. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, specific COA data, and detailed route feasibility assessments tailored to your target molecules. Let us be your strategic partner in driving efficiency and innovation in your chemical supply chain.
