Revolutionizing Tetrazolone Synthesis: A One-Step Palladium-Catalyzed Route for High-Purity Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly those serving as bioisosteres for carboxylic acids. Patent CN112574133B introduces a groundbreaking synthetic methodology for producing 1,3-diaryl substituted tetrazolone inner salts, a critical structural motif found in numerous bioactive molecules such as the analgesic alfentanil and various beta-3 adrenoreceptor agonists. This innovation represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-step palladium-catalyzed aerobic oxidation process. By utilizing readily available diarylsemicarbazide reagents as the starting material, the invention effectively bypasses the need for hazardous precursors like acid chlorides and trimethylsilylazide, which have historically plagued the supply chain with safety concerns and high costs. The technical breakthrough lies in the dual role of the palladium catalyst and atmospheric oxygen, which facilitates both the oxidative formation of an azo intermediate and its subsequent cyclization rearrangement under remarkably mild conditions. For R&D directors and process chemists, this patent offers a robust platform for generating diverse tetrazolone derivatives with high purity and efficiency, directly addressing the industry's demand for greener and more sustainable manufacturing protocols.
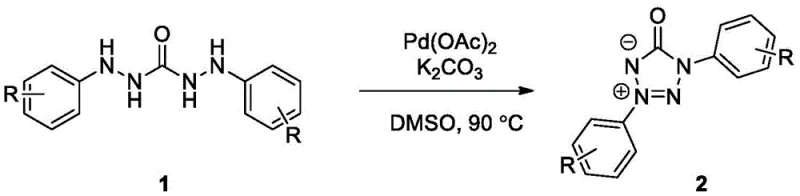
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetrazolone ring system has been fraught with significant synthetic challenges that hinder large-scale production and increase the overall cost of goods. Conventional routes often rely on the reaction between acid chlorides and trimethylsilylazide, a process that necessitates high temperatures and strictly anhydrous conditions to prevent premature decomposition of sensitive reagents. Furthermore, these traditional methods are generally limited to the synthesis of monosubstituted tetrazolones, failing to provide a versatile pathway for the 1,3-diaryl substituted variants which are crucial for specific pharmacological activities. The reliance on hazardous azide sources poses severe safety risks in a manufacturing environment, requiring specialized equipment and rigorous safety protocols that drive up capital expenditure. Additionally, the multi-step nature of older synthetic strategies inevitably leads to cumulative yield losses and increased waste generation, creating a substantial burden on environmental compliance teams. The complexity of purification in these legacy processes often results in difficult-to-remove impurities, compromising the quality of the final active pharmaceutical ingredient and necessitating costly additional refining steps.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method disclosed in CN112574133B utilizes a direct oxidative cyclization strategy that transforms simple diarylaminourea reagents into the desired tetrazolone inner salts in a single operational step. This novel approach leverages the power of palladium catalysis, specifically using palladium acetate in conjunction with potassium carbonate as a base, to activate the substrate under an ambient air atmosphere. The elimination of external oxidants, relying instead on molecular oxygen from the air, drastically simplifies the reaction setup and removes the cost associated with purchasing and handling stoichiometric oxidizing agents. The reaction proceeds smoothly in polar aprotic solvents like dimethyl sulfoxide (DMSO) at moderate temperatures ranging from 70 to 100 degrees Celsius, conditions that are easily achievable in standard glass-lined reactors without the need for cryogenic cooling or extreme heating. This simplicity translates directly into operational excellence, allowing manufacturers to reduce batch times and minimize the footprint of the production facility while maintaining high conversion rates. The versatility of this method is further evidenced by its tolerance to a wide range of functional groups, enabling the synthesis of diverse derivatives without the need for protecting group strategies.
Mechanistic Insights into Pd-Catalyzed Aerobic Oxidative Cyclization
The core of this technological advancement lies in the intricate interplay between the palladium catalyst and the molecular oxygen present in the air, which drives the transformation through a unique mechanistic pathway. Initially, the diarylaminourea substrate undergoes a palladium-catalyzed oxidation where atmospheric oxygen serves as the terminal electron acceptor, facilitating the formation of a reactive azo intermediate species. This oxidative step is critical as it generates the necessary nitrogen-nitrogen double bond character required for the subsequent ring closure, effectively mimicking the functionality of more dangerous azide reagents but through a much safer oxidative manifold. Once the azo intermediate is generated, the palladium center continues to play a pivotal role by coordinating with the nitrogen atoms, promoting an intramolecular cyclization rearrangement that constructs the five-membered tetrazolone ring. The presence of potassium carbonate is essential in this phase, acting as a base to deprotonate intermediate species and drive the equilibrium towards the formation of the inner salt product. This mechanism not only ensures high atom economy but also minimizes the formation of side products, as the reaction pathway is highly selective for the desired cyclization over other potential oxidative degradation routes. Understanding this mechanism allows process chemists to fine-tune reaction parameters such as oxygen flow or stirring rates to maximize efficiency in a commercial setting.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over traditional acid chloride-based routes. Because the reaction avoids the use of highly reactive acylating agents, there is a significant reduction in the formation of hydrolysis by-products and oligomeric impurities that typically complicate downstream purification. The mild basic conditions provided by potassium carbonate help to neutralize any acidic by-products formed during the oxidation, preventing acid-catalyzed decomposition of the sensitive tetrazolone ring. Furthermore, the use of DMSO as a solvent provides excellent solubility for both the starting materials and the polar transition states, ensuring a homogeneous reaction mixture that promotes consistent heat transfer and reduces the risk of hot spots which can lead to thermal degradation. The resulting crude reaction mixtures are generally cleaner, allowing for simpler work-up procedures involving aqueous quenching and organic extraction. This inherent cleanliness of the reaction profile means that the final product can often be isolated with high purity through standard column chromatography or even crystallization, reducing the need for extensive preparative HPLC purification which is often a bottleneck in pilot plant operations.
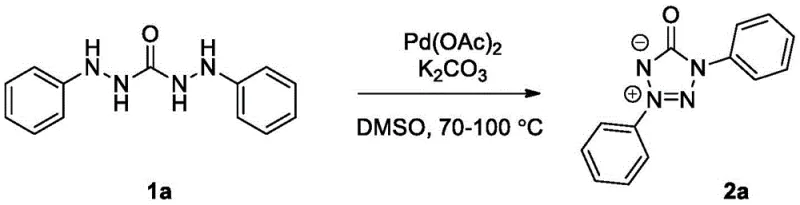
How to Synthesize 1,3-Diaryl Tetrazolone Inner Salt Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that ensure maximum yield and reproducibility. The process begins with the precise weighing of the diarylaminourea substrate, which serves as the foundational building block for the tetrazolone scaffold. It is crucial to maintain the stoichiometric balance between the substrate, the palladium catalyst, and the base to prevent catalyst deactivation or incomplete conversion. The choice of solvent is also paramount, with dimethyl sulfoxide demonstrating superior performance compared to other solvents like tetrahydrofuran or dioxane, likely due to its ability to stabilize the polar intermediates and facilitate oxygen solubility. Detailed standardized operating procedures for this synthesis, including specific mixing times and temperature ramping rates, are essential for technology transfer and scale-up success.
- Mix 0.5 mmol of diarylaminourea with 10 mol% palladium acetate catalyst and 1.0 mmol potassium carbonate base in a suitable solvent like DMSO.
- Heat the reaction mixture to 70-100°C (optimally 90°C) under an air atmosphere with magnetic stirring for 2-4 hours until TLC indicates completion.
- Quench with water, extract with dichloromethane, dry over anhydrous sodium sulfate, and purify via column chromatography to isolate the pure tetrazolone inner salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple yield improvements. The primary driver of value is the drastic simplification of the raw material portfolio; by shifting from hazardous acid chlorides and silylated azides to stable, commercially abundant diarylsemicarbazides, companies can significantly mitigate supply risk and reduce raw material inventory costs. These semicarbazide precursors are widely available from multiple global suppliers, fostering a competitive sourcing environment that prevents vendor lock-in and ensures long-term price stability. Moreover, the elimination of expensive and dangerous reagents translates directly into lower logistics and storage costs, as there is no longer a need for specialized containment systems for pyrophoric or toxic substances. The operational simplicity of the one-step process also reduces the demand for skilled labor and complex reactor configurations, allowing manufacturing sites to increase throughput without proportional increases in capital investment. This efficiency gain is critical for maintaining margins in the highly competitive generic pharmaceutical and agrochemical markets.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the replacement of costly stoichiometric oxidants with free atmospheric oxygen. In traditional synthesis, the purchase and disposal of oxidizing agents represent a significant line item in the cost of goods sold; by utilizing air, this expense is virtually eliminated. Additionally, the high selectivity of the palladium-catalyzed reaction minimizes the formation of waste by-products, thereby reducing the costs associated with waste treatment and environmental compliance. The ability to run the reaction at moderate temperatures (70-100 degrees Celsius) also lowers energy consumption compared to high-temperature protocols, contributing to a smaller carbon footprint and reduced utility bills. Furthermore, the simplified work-up procedure requires fewer solvents and less time for purification, leading to substantial savings in solvent procurement and recovery operations. These cumulative factors create a leaner, more cost-effective manufacturing model that enhances overall profitability.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of this synthetic method. The use of common, non-proprietary reagents like potassium carbonate and palladium acetate ensures that production is not vulnerable to shortages of exotic or single-source chemicals. The tolerance of the reaction to various substituents, including halogens and alkyl groups, means that a single manufacturing platform can be used to produce a wide library of tetrazolone derivatives, increasing asset utilization and flexibility. This versatility allows manufacturers to respond rapidly to market demands for different API intermediates without the need for extensive process re-validation or equipment modification. The stability of the starting materials also permits longer shelf-life and easier transportation, reducing the risk of spoilage during transit. Consequently, companies can maintain lower safety stock levels while still guaranteeing continuous supply to their downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the benign nature of the reaction conditions and the absence of explosive hazards. The use of air as an oxidant removes the risk of runaway exotherms associated with strong chemical oxidizers, making the process inherently safer for large-scale reactors. From an environmental standpoint, the reduction in hazardous waste generation aligns perfectly with modern green chemistry principles and increasingly stringent regulatory frameworks. The lower E-factor (mass of waste per mass of product) achieved through this high-yield, one-step process simplifies the permitting process for new manufacturing lines. Additionally, the potential for catalyst recovery and recycling in palladium-catalyzed systems offers further opportunities to minimize heavy metal discharge and reduce the consumption of precious metals. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the corporate brand image among environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tetrazolone synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production pipelines.
Q: What are the key advantages of this Pd-catalyzed method over traditional tetrazolone synthesis?
A: Unlike traditional methods requiring hazardous acid chlorides and trimethylsilylazide at high temperatures, this patent describes a mild, one-step aerobic oxidation using readily available diarylsemicarbazides, significantly improving safety and operational simplicity.
Q: What is the substrate scope for this tetrazolone synthesis reaction?
A: The method demonstrates excellent compatibility with various substituents including electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens like chlorine and fluorine, yielding products with isolation yields ranging from 65% to 84%.
Q: Is this process scalable for commercial production of API intermediates?
A: Yes, the use of air as the terminal oxidant, combined with common solvents like DMSO and standard bases like potassium carbonate, eliminates the need for expensive or hazardous reagents, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Diaryl Tetrazolone Inner Salt Supplier
As the global demand for complex heterocyclic intermediates continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM becomes a strategic imperative for pharmaceutical companies. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the highest standards of safety and precision, utilizing stringent purity specifications and rigorous QC labs to guarantee that every batch meets your exacting requirements. We understand the critical nature of API intermediates in the drug development timeline and are committed to delivering high-purity 1,3-diaryl tetrazolone inner salts that accelerate your path to market. Our technical team is adept at optimizing reaction parameters to maximize yield and minimize impurities, leveraging the insights from patents like CN112574133B to drive innovation in your supply chain.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener, more efficient methodology. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics. Let us help you optimize your supply chain and reduce your time to market with our superior manufacturing capabilities and dedication to quality excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
