Advanced One-Pot Synthesis of 2-Bromo-4,5-Dimethoxybenzyl Bromide for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical active pharmaceutical ingredient (API) intermediates. A significant advancement in this domain is detailed in patent CN107417501B, which discloses a novel preparation method for 2-bromo-4,5-dimethoxybenzyl bromide. This compound serves as a pivotal building block in the synthesis of Pinaverium Bromide, a widely prescribed calcium channel blocker used for treating gastrointestinal disorders. The traditional manufacturing pathways for this intermediate have long been plagued by safety hazards associated with handling elemental bromine and the high costs of specialized brominating agents. The disclosed technology addresses these challenges by introducing a controlled, in-situ bromination strategy that utilizes inexpensive inorganic salts. By shifting from hazardous liquid reagents to a solid-state redox system, this innovation not only enhances operator safety but also streamlines the purification process, offering a compelling value proposition for manufacturers aiming to optimize their supply chains for gastrointestinal therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-bromo-4,5-dimethoxybenzyl bromide has relied on methodologies that present substantial logistical and economic burdens. Conventional routes often utilize 3,4-dimethoxybenzyl alcohol or 3,4-dimethoxybenzaldehyde as starting materials, both of which command significantly higher market prices compared to simple toluene derivatives. Furthermore, the bromination steps in these legacy processes frequently employ liquid bromine or phosphorus tribromide. Liquid bromine is highly corrosive, volatile, and toxic, necessitating expensive containment infrastructure and rigorous safety protocols that inflate capital expenditure. Alternative methods using N-bromosuccinimide (NBS) avoid liquid bromine but suffer from poor atom economy and low effective bromine content, leading to excessive reagent consumption. Literature indicates that NBS-mediated routes often achieve yields as low as 38.6%, rendering them economically unviable for large-scale production. These inefficiencies create bottlenecks in the supply chain, resulting in higher costs for the final API and limiting the availability of affordable treatment options for patients.
The Novel Approach
The methodology described in patent CN107417501B represents a paradigm shift by employing a one-pot tandem reaction sequence starting from the much more affordable 3,4-dimethoxytoluene. Instead of introducing external bromine, the process generates the brominating agent in-situ through the redox reaction of sodium bromate and sodium bromide in the presence of sulfuric acid. This approach allows for precise control over the concentration of reactive bromine species, thereby minimizing side reactions. The entire transformation occurs in a non-polar solvent, typically carbon tetrachloride, facilitating a seamless transition from aromatic electrophilic substitution to benzylic radical substitution without isolating intermediates. 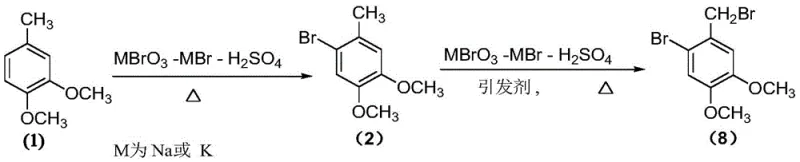 This consolidation of steps drastically reduces solvent waste and processing time. By eliminating the need for intermediate isolation and utilizing stable solid reagents, the novel approach significantly lowers the barrier to entry for commercial manufacturing, ensuring a more reliable and cost-effective supply of this critical pharmaceutical intermediate.
This consolidation of steps drastically reduces solvent waste and processing time. By eliminating the need for intermediate isolation and utilizing stable solid reagents, the novel approach significantly lowers the barrier to entry for commercial manufacturing, ensuring a more reliable and cost-effective supply of this critical pharmaceutical intermediate.
Mechanistic Insights into In-Situ Bromination and Radical Substitution
The chemical elegance of this process lies in its dual-mechanism design, which sequentially targets specific C-H bonds with high fidelity. The first stage involves the electrophilic bromination of the aromatic ring. The methoxy groups on the 3,4-dimethoxytoluene substrate are strong ortho-para directors, theoretically allowing bromination at multiple positions. However, the controlled release of bromine from the bromate-bromide-sulfuric acid system moderates the reaction kinetics. This kinetic control favors the formation of the 2-bromo isomer (Compound 2) over other potential regioisomers (Compounds 3-7). 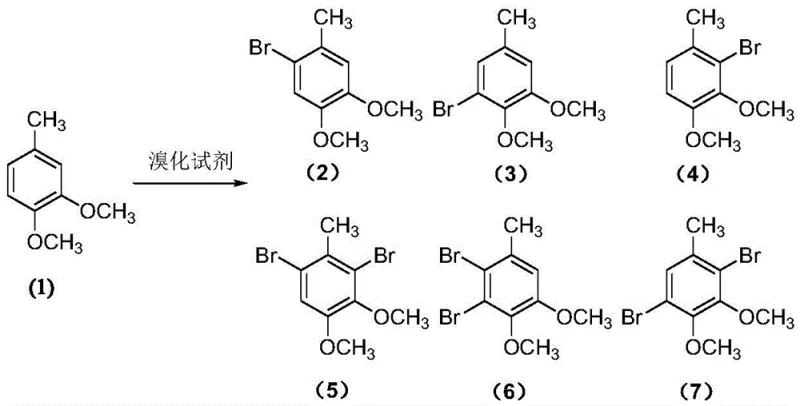 By avoiding a sudden surge in bromine concentration, the system prevents polybromination and ensures that the electrophilic attack occurs selectively at the position ortho to the methoxy group and meta to the methyl group, setting the stage for the subsequent transformation.
By avoiding a sudden surge in bromine concentration, the system prevents polybromination and ensures that the electrophilic attack occurs selectively at the position ortho to the methoxy group and meta to the methyl group, setting the stage for the subsequent transformation.
Following the completion of the aromatic substitution, the reaction conditions are modified to initiate the second stage: free radical bromination of the benzylic methyl group. Upon the addition of a radical initiator, such as azobisisoheptanenitrile (ABVN), and additional sulfuric acid, the reaction environment shifts to favor homolytic cleavage. The benzylic C-H bond is inherently weaker than aromatic C-H bonds, making it susceptible to radical abstraction. The generated bromine radicals selectively abstract a hydrogen atom from the methyl group, forming a stable benzylic radical intermediate which then reacts with molecular bromine to form the final benzyl bromide moiety. This sequential activation ensures that the aromatic ring remains intact while the side chain is functionalized, achieving the complex structural modification in a single vessel with high chemoselectivity.
How to Synthesize 2-Bromo-4,5-Dimethoxybenzyl Bromide Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and addition rates to maintain the delicate balance between the two reaction phases. The process begins by suspending the solid bromine sources in the non-polar solvent before heating. The gradual addition of sulfuric acid is critical; adding it too quickly could lead to uncontrolled bromine generation and loss of selectivity. Once the aromatic bromination is confirmed complete via monitoring techniques like gas chromatography, the system is primed for the radical phase.
- Dissolve 3,4-dimethoxytoluene, sodium bromate, and sodium bromide in carbon tetrachloride and heat to reflux.
- Slowly add sulfuric acid to generate bromine in-situ for selective electrophilic bromination of the aromatic ring.
- Add a radical initiator and additional sulfuric acid to facilitate benzylic bromination, yielding the final product after workup.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits beyond mere technical feasibility. The shift from expensive, specialized starting materials to commodity chemicals like 3,4-dimethoxytoluene fundamentally alters the cost structure of the intermediate. Since the raw materials are widely available and inexpensive, the volatility of the supply chain is significantly reduced, protecting the manufacturer from price spikes associated with niche reagents. Furthermore, the replacement of hazardous liquid bromine with stable solid salts simplifies storage and transportation logistics. Solid reagents do not require the same level of specialized containment as corrosive liquids, reducing warehousing costs and insurance premiums. This simplification of the input material profile enhances the overall resilience of the supply chain, ensuring consistent production schedules even during periods of market fluctuation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of costly reagents and the reduction of unit operations. By utilizing sodium bromate and sodium bromide, which are substantially cheaper than NBS or liquid bromine on a molar basis, the direct material cost is drastically lowered. Additionally, the one-pot nature of the synthesis removes the need for intermediate isolation, filtration, and drying steps. This consolidation reduces labor hours, energy consumption for heating and cooling cycles, and solvent usage for washing and recrystallization. The cumulative effect is a leaner manufacturing process that delivers substantial cost savings without compromising on the quality of the final product, allowing for more competitive pricing in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Reliability in the chemical supply chain is often compromised by the availability of hazardous or regulated substances. Liquid bromine is subject to strict transportation regulations and requires specialized tankers, creating potential bottlenecks. In contrast, the solid brominating agents used in this method are stable, non-volatile, and easy to ship using standard logistics channels. This ease of transport ensures that production facilities can maintain adequate inventory levels without facing regulatory hurdles. Moreover, the robustness of the reaction against minor variations in reagent quality means that manufacturers can source materials from a broader range of suppliers, further mitigating the risk of supply disruptions and ensuring continuous availability of the intermediate for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this methodology is inherently designed for industrial expansion. The in-situ generation of bromine means that only the amount needed for the reaction is produced at any given moment, minimizing the inventory of hazardous intermediates within the reactor. This "just-in-time" reagent generation significantly lowers the risk of runaway reactions or toxic releases. From an environmental perspective, the higher selectivity of the reaction reduces the formation of polybrominated byproducts, simplifying waste treatment and reducing the load on effluent processing systems. The ability to run the process in a single pot also minimizes solvent waste, aligning with modern green chemistry principles and helping manufacturers meet increasingly stringent environmental compliance standards.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its implementation. The following questions address common concerns regarding safety, yield, and scalability based on the data provided in the patent documentation. These insights are derived directly from the experimental examples and comparative analysis presented in the intellectual property, offering a clear picture of what partners can expect when adopting this technology for commercial production.
Q: Why is the in-situ bromination method safer than using liquid bromine?
A: The patented method generates bromine internally from solid sodium bromate and bromide salts upon acidification. This eliminates the need to handle, store, and transport hazardous liquid bromine, significantly reducing operational risks and environmental exposure.
Q: How does this process improve yield compared to NBS methods?
A: Prior art using N-bromosuccinimide (NBS) often suffers from low yields around 38.6% due to poor selectivity. The controlled release of bromine in this new method enhances regioselectivity for the 2-position, achieving yields up to 85%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the one-pot design minimizes unit operations and solvent usage. The use of stable solid reagents instead of volatile liquids simplifies logistics and reactor safety requirements, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-4,5-Dimethoxybenzyl Bromide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields reported in patent CN107417501B can be reliably replicated on an industrial scale. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-bromo-4,5-dimethoxybenzyl bromide meets the exacting standards required for pharmaceutical applications. We understand the critical nature of this intermediate in the Pinaverium Bromide supply chain and are committed to delivering consistent quality and volume.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology. By working together, we can optimize the production economics and secure a stable supply of high-quality intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented process can drive value and efficiency in your manufacturing operations.
