Advanced Chiral Hydroxyl Amide Production: Leveraging Copper-Mediated Ring Opening for High-Purity Intermediates
Introduction to Patent CN102174006A: A Breakthrough in Chiral Synthesis
The landscape of asymmetric synthesis is constantly evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and fine chemical sectors. Patent CN102174006A introduces a sophisticated methodology for the preparation of a specific chiral compound, identified as N-2-beta-hydroxyl-1-benzyl-1-pyrrolacrylamide-2-one. This innovation represents a significant departure from traditional acid-catalyzed pathways, leveraging a unique metal-mediated ring-opening strategy. The core of this technology lies in the transformation of an oxazoline precursor into a valuable chiral hydroxyl amide using copper acetate monohydrate. For R&D directors and process chemists, this patent offers a compelling alternative to established protocols, promising enhanced control over stereochemistry and improved reaction efficiency. The detailed disclosure within the patent provides a robust framework for producing complex nitrogenous chiral compounds, which are critical building blocks in the development of bioactive molecules and advanced organic materials.
Furthermore, the utility of this compound extends beyond its role as a static intermediate; it exhibits remarkable catalytic performance in downstream reactions, specifically in the nitrile silication of aldehydes. This dual functionality—as both a target molecule and a catalyst precursor—adds substantial value to the synthetic route described in CN102174006A. By addressing the technical challenges associated with ring-opening reactions and stereoselectivity, this patent lays the groundwork for more efficient manufacturing processes. For supply chain leaders, understanding the nuances of this chemistry is vital, as it points towards potential optimizations in raw material utilization and waste reduction. The following analysis dissects the technical merits of this invention, highlighting its potential to redefine standards in reliable pharmaceutical intermediate supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral hydroxyl amides has been fraught with challenges related to reaction conditions and catalyst efficacy. Traditional methodologies often depend on strong acidic environments or heterogeneous catalysts like proton-exchanged montmorillonite to drive the formation of C-N bonds or the hydrolysis of precursors. These conventional approaches frequently suffer from issues such as poor atom economy, the generation of significant amounts of acidic waste, and difficulties in controlling the stereochemical outcome of the reaction. Moreover, the harsh conditions required can lead to the degradation of sensitive functional groups, resulting in lower overall yields and a complex impurity profile that necessitates extensive and costly purification steps. For procurement managers, these inefficiencies translate directly into higher operational costs and longer lead times, as the removal of trace metals or acid residues becomes a bottleneck in the production workflow.
The Novel Approach
In stark contrast, the methodology outlined in CN102174006A employs a milder, more controlled pathway utilizing Lewis acid catalysis. The process begins with the formation of an oxazoline intermediate through a reflux reaction between 2-pyrrolidone-phenylpropyl cyanide and L-phenylalaninol, catalyzed by anhydrous zinc chloride in chlorobenzene. This initial step is crucial for establishing the chiral center with high fidelity. 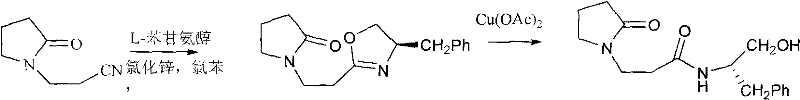 The subsequent transformation involves the reaction of this intermediate with copper acetate monohydrate in an ethanol mixed solvent. This copper-mediated step facilitates the ring-opening of the oxazoline moiety to yield the target chiral hydroxyl amide. The use of copper acetate not only acts as a catalyst but also participates in the structural reorganization of the molecule, leading to the formation of complex single crystals upon recrystallization. This novel approach eliminates the need for corrosive strong acids, thereby simplifying the workup procedure and enhancing the safety profile of the manufacturing process. The result is a cleaner reaction profile with a conversion rate reaching up to 99% in catalytic applications, demonstrating superior efficiency compared to legacy methods.
The subsequent transformation involves the reaction of this intermediate with copper acetate monohydrate in an ethanol mixed solvent. This copper-mediated step facilitates the ring-opening of the oxazoline moiety to yield the target chiral hydroxyl amide. The use of copper acetate not only acts as a catalyst but also participates in the structural reorganization of the molecule, leading to the formation of complex single crystals upon recrystallization. This novel approach eliminates the need for corrosive strong acids, thereby simplifying the workup procedure and enhancing the safety profile of the manufacturing process. The result is a cleaner reaction profile with a conversion rate reaching up to 99% in catalytic applications, demonstrating superior efficiency compared to legacy methods.
Mechanistic Insights into Copper-Mediated Oxazoline Ring Opening
The mechanistic elegance of this synthesis lies in the specific interaction between the copper species and the oxazoline ring. Unlike simple hydrolysis, the presence of copper acetate monohydrate suggests a coordination mechanism where the metal center activates the oxazoline nitrogen or the adjacent carbon atoms, rendering the ring susceptible to nucleophilic attack by solvent molecules or internal rearrangement. This metal-ligand interaction is pivotal for breaking the C-O or C-N bond within the heterocyclic ring without racemizing the chiral center established in the previous step. The patent data indicates that the reaction proceeds under reflux conditions for 48 hours in ethanol, allowing sufficient time for the thermodynamic equilibrium to favor the open-chain hydroxyl amide structure. 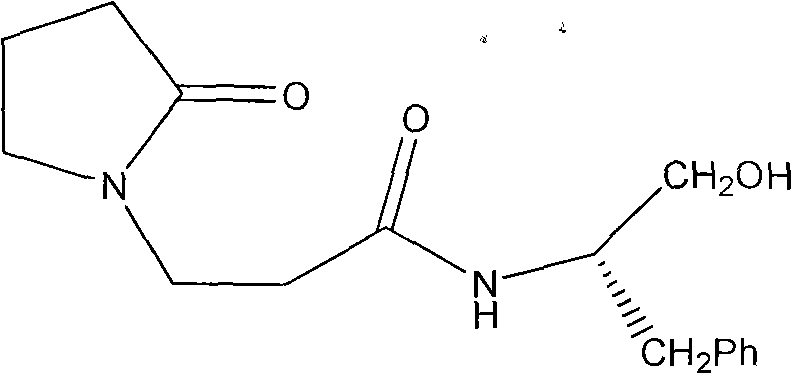 The isolation of the product as a monocrystal further confirms the high degree of structural order and purity achieved through this mechanism. For technical teams, understanding this coordination chemistry is essential for troubleshooting and optimizing reaction parameters such as temperature and solvent polarity to maximize yield.
The isolation of the product as a monocrystal further confirms the high degree of structural order and purity achieved through this mechanism. For technical teams, understanding this coordination chemistry is essential for troubleshooting and optimizing reaction parameters such as temperature and solvent polarity to maximize yield.
Beyond its synthesis, the resulting compound demonstrates exceptional utility as a catalyst itself, particularly in the silylation of nitriles. When applied to the reaction of 2-methylbenzaldehyde with trimethylsilyl cyanide (TMSCN), the compound facilitates the formation of 2-aminomethyl phenyl-2-(trimethylsiloxy) propionitrile with a transformation efficiency exceeding 99%.  This high conversion rate underscores the compound's effectiveness in activating electrophilic centers, likely through hydrogen bonding or Lewis acid interactions provided by the hydroxyl and amide functionalities. The ability to achieve such high conversion under mild conditions (20-30°C) highlights the potential for this chemistry to be adapted for other asymmetric transformations. For R&D directors, this mechanistic versatility opens avenues for developing new synthetic routes for complex API intermediates, reducing reliance on expensive noble metal catalysts.
This high conversion rate underscores the compound's effectiveness in activating electrophilic centers, likely through hydrogen bonding or Lewis acid interactions provided by the hydroxyl and amide functionalities. The ability to achieve such high conversion under mild conditions (20-30°C) highlights the potential for this chemistry to be adapted for other asymmetric transformations. For R&D directors, this mechanistic versatility opens avenues for developing new synthetic routes for complex API intermediates, reducing reliance on expensive noble metal catalysts.
How to Synthesize N-2-beta-hydroxyl-1-benzyl-1-pyrrolacrylamide-2-one Efficiently
The practical execution of this synthesis requires careful attention to anhydrous and oxygen-free conditions, particularly during the initial formation of the oxazoline intermediate. The protocol dictates a rigorous exclusion of moisture to prevent premature hydrolysis or side reactions that could compromise the integrity of the chiral center. Following the reflux period, the separation process involves removing the chlorobenzene solvent, dissolving the residue in water, and extracting with dichloromethane. This liquid-liquid extraction is critical for removing inorganic salts and polar impurities before the final purification via column chromatography. The detailed standardized synthesis steps for this high-value intermediate are provided in the guide below.
- Reflux 2-pyrrolidone-phenylpropyl cyanide with L-phenylalaninol in chlorobenzene using anhydrous ZnCl2 catalyst under oxygen-free conditions for 24 hours to form the oxazoline intermediate.
- Purify the intermediate via water dissolution, dichloromethane extraction, and column chromatography to remove solvent residues and impurities.
- React the purified intermediate with copper acetate monohydrate in ethanol under reflux for 48 hours, followed by recrystallization with ethanol and n-hexane to obtain the target chiral compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN102174006A offers tangible strategic benefits that extend beyond mere chemical novelty. The shift away from harsh acidic catalysts to a copper-zinc mediated system fundamentally alters the cost structure of production. By eliminating the need for specialized acid-resistant equipment and reducing the complexity of waste neutralization, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing. The use of commodity chemicals like ethanol, chlorobenzene, and copper acetate ensures that raw material sourcing remains stable and cost-effective, mitigating the risks associated with supply chain volatility for exotic reagents.
- Cost Reduction in Manufacturing: The streamlined purification process, which relies on standard extraction and recrystallization techniques rather than complex chromatographic separations on a large scale, drastically lowers operational expenditures. The high conversion rates observed in downstream applications mean less raw material is wasted, directly improving the overall material balance and yield of the final process. Furthermore, the avoidance of expensive noble metals or proprietary organocatalysts reduces the direct material cost per kilogram of product, enhancing the margin potential for commercial scale-up.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to greater supply chain continuity. Since the reagents involved are widely available industrial chemicals, the risk of supply disruption is minimized compared to processes relying on bespoke catalysts. The ability to produce the compound in high purity with consistent batch-to-batch reproducibility ensures that downstream customers receive a reliable chiral catalyst supplier product that meets stringent quality specifications. This reliability is crucial for maintaining production schedules in the fast-paced pharmaceutical industry.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates less hazardous waste due to the absence of strong mineral acids. This simplifies effluent treatment and helps facilities maintain compliance with increasingly strict environmental regulations. The scalability of the reflux and recrystallization steps is well-understood in chemical engineering, facilitating a smoother transition from laboratory benchtop to pilot plant and eventually to full commercial production. This ease of scale-up reduces the time-to-market for new products utilizing this chiral scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the limitations of traditional chiral hydroxyl amide synthesis methods?
A: Conventional methods often rely on harsh acidic conditions or proton-exchanged montmorillonite, which can lead to lower selectivity, difficult purification processes, and potential degradation of sensitive functional groups within the molecular structure.
Q: How does the copper acetate mediated method improve conversion rates?
A: The novel approach utilizes copper acetate monohydrate to facilitate the ring-opening of the oxazoline intermediate under mild reflux conditions, achieving conversion rates up to 99% in subsequent catalytic applications like nitrile silication, significantly outperforming traditional acid catalysts.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes common solvents like chlorobenzene and ethanol and avoids exotic reagents, making it highly suitable for scale-up. The robust purification steps involving extraction and recrystallization ensure consistent quality control essential for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Hydroxyl Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in driving innovation within the pharmaceutical and agrochemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN102174006A can be translated into industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify stereochemical integrity and impurity profiles. Our capability to handle complex metal-mediated reactions allows us to offer a reliable chiral hydroxyl amide supplier service that meets the exacting demands of global clients.
We invite you to collaborate with us to explore the full potential of this technology for your specific applications. Whether you require custom synthesis or process optimization, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs. Contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with our proven expertise in fine chemical manufacturing.
