Revolutionizing Indanone Production: A Cost-Effective Copper-Catalyzed Strategy for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and economically viable synthetic routes for high-value intermediates. A recent technological breakthrough, documented in patent CN108689815B, introduces a highly efficient method for synthesizing indanone compounds, a structural motif prevalent in numerous bioactive molecules. This innovation shifts the paradigm from traditional noble metal catalysis to a more sustainable copper-catalyzed system. Indanones serve as critical scaffolds in the development of therapeutic agents, including treatments for neurodegenerative diseases and viral infections. For instance, the structural core is found in compounds similar to Donepezil, used for Alzheimer's disease, and Indinavir, a protease inhibitor for HIV. The ability to construct these cores efficiently is paramount for maintaining a steady supply of life-saving medications.
The significance of this patent lies not just in the chemical transformation itself, but in its alignment with green chemistry principles and cost-efficiency goals. By utilizing earth-abundant copper instead of scarce precious metals, the process addresses both economic and environmental concerns simultaneously. This report analyzes the technical merits of this copper-catalyzed hydroacylation reaction, providing R&D directors, procurement managers, and supply chain heads with a comprehensive understanding of its potential impact on manufacturing strategies. The transition to base metal catalysis represents a strategic opportunity to decouple production costs from the volatile markets of rhodium, palladium, and iridium, ensuring greater stability in the long-term supply of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indanone skeleton via intramolecular hydroacylation of aldehydes and unsaturated organic compounds has relied heavily on transition metal catalysts derived from the platinum group. Metals such as rhodium, cobalt, ruthenium, iridium, and palladium have been the standard bearers for these transformations due to their high activity and selectivity. However, reliance on these noble metals introduces severe bottlenecks for large-scale industrial application. Firstly, the cost of these metals is exorbitant and subject to extreme geopolitical and market fluctuations, making budget forecasting for long-term projects difficult. Secondly, and perhaps more critically for the pharmaceutical sector, is the issue of residual heavy metals. Regulatory bodies impose strict limits on the ppm levels of toxic metals in Active Pharmaceutical Ingredients (APIs). Removing trace amounts of rhodium or palladium from the final product often requires complex, multi-step purification processes involving specialized scavengers or chromatography, which significantly increases processing time and waste generation. These factors collectively inflate the cost of goods sold (COGS) and extend the lead time for getting new drugs to market.
The Novel Approach
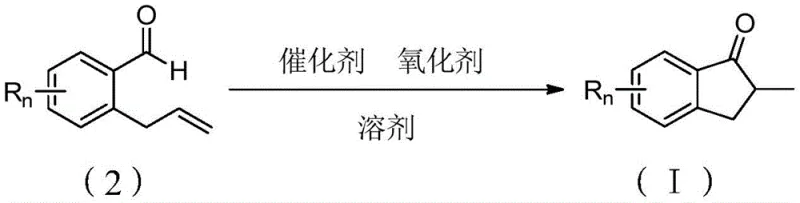
The methodology disclosed in patent CN108689815B offers a transformative solution by replacing these expensive noble metals with copper-based catalysts. Copper is not only orders of magnitude cheaper but also possesses a rich coordination chemistry that can be harnessed for radical-mediated transformations. The novel approach utilizes a copper catalyst in conjunction with tert-butyl hydroperoxide (TBHP) as an oxidant to facilitate the cyclization of 2-allylbenzaldehyde derivatives. This system operates effectively in common organic solvents like toluene at moderate temperatures (around 110°C). By shifting to a copper-catalyzed regime, manufacturers can bypass the need for expensive metal scavengers, thereby simplifying the downstream processing workflow. The reaction demonstrates excellent functional group tolerance, accommodating various substituents on the aromatic ring, which is essential for generating diverse chemical libraries during the drug discovery phase. This approach effectively democratizes access to complex indanone scaffolds, making them more accessible for both academic research and industrial scale-up.
Mechanistic Insights into Copper-Catalyzed Intramolecular Hydroacylation
The success of this synthesis relies on the unique ability of copper species to mediate radical pathways under oxidative conditions. Unlike the concerted mechanisms often seen with noble metals, the copper-catalyzed process likely proceeds through a radical cascade initiated by the interaction between the copper catalyst and the peroxide oxidant. The tert-butyl hydroperoxide (TBHP) serves a dual role: it acts as the terminal oxidant to regenerate the active copper species and potentially generates radical intermediates that facilitate the C-C bond formation. The reaction begins with the activation of the aldehyde C-H bond or the alkene moiety by the copper center. Subsequent intramolecular attack of the acyl radical or copper-acyl species onto the pendant alkene leads to the formation of the five-membered indanone ring. The presence of the oxidant ensures that the catalytic cycle is closed by re-oxidizing the reduced copper species back to its active state, allowing the reaction to proceed with catalytic amounts of metal. This mechanistic pathway is distinct from traditional hydrogenation-acylation reactions and offers a complementary reactivity profile that tolerates sensitive functional groups which might otherwise be reduced by hydrogenation conditions.
From an impurity control perspective, this mechanism offers distinct advantages. The use of TBHP as a clean oxidant results in tert-butanol as the primary byproduct, which is relatively easy to remove during aqueous workup compared to the complex ligand debris often left behind by noble metal catalysts. Furthermore, the reaction conditions (110°C in toluene) are robust enough to drive the reaction to completion while minimizing the formation of polymeric byproducts that can plague radical reactions. The screening data indicates that Copper(II) Oxide (CuO) is particularly effective, suggesting that the heterogeneous nature of the catalyst or the specific oxidation state of copper plays a crucial role in suppressing side reactions. This high level of chemoselectivity ensures that the resulting indanone products possess a clean impurity profile, a critical requirement for intermediates destined for GMP manufacturing. The ability to control the reaction outcome through simple adjustments in catalyst loading and oxidant stoichiometry provides process chemists with the necessary levers to optimize purity and yield consistently.

How to Synthesize 2-Methyl-1-indanone Efficiently
The practical implementation of this technology is straightforward and amenable to standard laboratory and plant equipment. The protocol involves a one-pot procedure where the catalyst, oxidant, substrate, and solvent are combined under an inert atmosphere. The simplicity of the operation reduces the risk of operator error and facilitates technology transfer from R&D to production scales. The process does not require specialized high-pressure equipment or cryogenic conditions, further lowering the barrier to entry for contract manufacturing organizations (CMOs). For detailed operational parameters, including precise stoichiometric ratios and workup procedures, please refer to the standardized synthesis guide below.
- Prepare the reaction system by adding a copper catalyst (preferably CuO) and tert-butyl hydroperoxide (TBHP) as the oxidant into a dry reaction vessel under inert atmosphere.
- Introduce the substrate, 2-allylbenzaldehyde (or its derivatives), and the solvent (preferably toluene) into the mixture, ensuring strict argon protection.
- Heat the reaction mixture to 110°C and maintain for approximately 5 hours to achieve optimal conversion, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology presents a compelling value proposition centered on cost stability and operational efficiency. The primary driver of value is the drastic reduction in raw material costs associated with the catalyst. By eliminating the dependency on rhodium, palladium, or iridium, companies can insulate their supply chains from the volatility of the precious metals market. This shift translates directly into improved margin protection for high-volume intermediates. Moreover, the simplified purification process required to remove copper residues, as opposed to noble metals, reduces the consumption of expensive scavenging resins and silica gel, leading to substantial savings in consumables and waste disposal costs. The overall process intensity is lowered, allowing for faster batch turnover and increased throughput in existing manufacturing facilities without the need for significant capital expenditure on new hardware.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts with copper oxide results in a fundamental restructuring of the cost base for indanone production. Copper is an earth-abundant base metal with a stable and low price point, contrasting sharply with the fluctuating and high costs of platinum group metals. This substitution eliminates the need for costly metal scavenging steps typically required to meet regulatory limits for heavy metals in pharmaceuticals. Consequently, the overall cost of goods is significantly reduced, not only through cheaper catalyst input but also through reduced downstream processing time and lower waste treatment expenses. This economic efficiency makes the production of complex indanone derivatives commercially viable even at lower price points, enhancing competitiveness in the generic drug market.
- Enhanced Supply Chain Reliability: Relying on copper rather than scarce noble metals mitigates supply risk. The global supply of rhodium and palladium is concentrated in few geographic regions, making it susceptible to geopolitical disruptions and mining constraints. Copper, being widely available and recycled, offers a secure and continuous supply stream. This reliability ensures that production schedules for critical pharmaceutical intermediates are not interrupted by catalyst shortages. Additionally, the reagents used, such as TBHP and toluene, are commodity chemicals with robust global supply chains, further reinforcing the resilience of the manufacturing process against external shocks. This stability is crucial for maintaining uninterrupted supply to downstream API manufacturers and ultimately to patients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been optimized in conditions that mimic industrial reactors (elevated temperatures in standard organic solvents). The use of toluene, a common industrial solvent, simplifies solvent recovery and recycling protocols. From an environmental standpoint, the avoidance of toxic heavy metals aligns with increasingly stringent global environmental regulations regarding heavy metal discharge and residue. The cleaner reaction profile reduces the burden on wastewater treatment facilities and lowers the environmental footprint of the manufacturing site. This compliance advantage facilitates easier regulatory approval for new manufacturing sites and supports corporate sustainability goals, which are becoming key criteria in vendor selection processes for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, aiming to clarify the feasibility and advantages of adopting this route for commercial production. Understanding these nuances is essential for making informed decisions about process development and vendor qualification.
Q: Why is copper preferred over noble metals like rhodium or palladium for this synthesis?
A: Copper catalysts are significantly cheaper and more abundant than noble metals, drastically reducing raw material costs. Furthermore, copper avoids the issue of toxic heavy metal residues often associated with rhodium or palladium, simplifying downstream purification and meeting stringent pharmaceutical purity standards.
Q: What are the optimal reaction conditions for maximizing yield in this process?
A: According to the patent data, the optimal conditions involve using Copper(II) Oxide (CuO) as the catalyst, tert-butyl hydroperoxide (TBHP) as the oxidant, and toluene as the solvent. The reaction should be conducted at 110°C for 5 hours under an argon atmosphere to achieve yields up to 79%.
Q: Can this method be applied to substrates with different substituents?
A: Yes, the method demonstrates broad substrate scope. It is effective for 2-allylbenzaldehydes bearing various substituents such as halogens, alkoxy groups, and alkyl groups on the aromatic ring, making it versatile for synthesizing diverse indanone libraries for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-1-indanone Supplier
The technological advancements detailed in patent CN108689815B underscore the potential for more efficient and sustainable manufacturing of indanone intermediates. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of such chemical innovations. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch meets stringent purity specifications, regardless of the complexity of the synthetic route. We understand that transitioning to a new catalytic system requires confidence in the supplier's ability to manage process variables and ensure consistent quality.
We invite you to leverage our technical expertise to evaluate the feasibility of this copper-catalyzed route for your specific project needs. Our team is ready to provide a Customized Cost-Saving Analysis comparing this novel method against your current supply chain arrangements. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and pilot batch samples. Let us collaborate to optimize your supply chain and accelerate your drug development timeline with reliable, high-quality intermediates.
