Advanced Cyanide-Free Synthesis of Drotaverine Hydrochloride Intermediates for Commercial Scale-up
Advanced Cyanide-Free Synthesis of Drotaverine Hydrochloride Intermediates for Commercial Scale-up
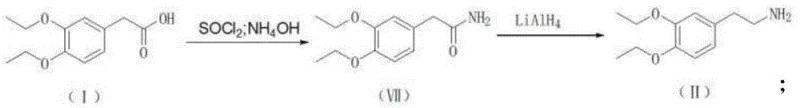
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally compliant synthetic routes for high-volume active pharmaceutical ingredients (APIs). A pivotal advancement in this domain is detailed in patent CN111170847B, which discloses a novel method for preparing drotaverine hydrochloride intermediates without relying on hazardous cyanide chemistry. Drotaverine, a potent spasmolytic agent widely used for treating smooth muscle spasms, traditionally requires the synthesis of 3,4-diethoxyphenylacetonitrile, a step fraught with safety and environmental challenges due to the use of sodium cyanide. This new technical disclosure offers a paradigm shift by directly synthesizing the critical precursors, 3,4-diethoxyphenylacetic acid and 3,4-diethoxyphenylethylamine, through safer Friedel-Crafts acylation and subsequent transformation pathways. For R&D directors and procurement strategists, this represents a significant opportunity to optimize supply chains while adhering to stricter global safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of drotaverine hydrochloride has been heavily dependent on the preparation of 3,4-diethoxyphenylacetonitrile as a central branching point. Conventional literature, including older patents like US4126615, describes routes involving the chloromethylation of o-phenetidine derivatives followed by nucleophilic substitution with sodium cyanide. This reliance on cyanide introduces severe operational hazards, requiring specialized containment infrastructure and rigorous operator protection protocols to prevent acute toxicity. Furthermore, the generation of cyanide-containing wastewater creates a substantial downstream burden, necessitating complex oxidation treatments to meet environmental discharge standards before effluent release. These factors not only inflate the operational expenditure (OPEX) due to safety compliance and waste management but also introduce potential supply chain bottlenecks if regulatory inspections tighten around cyanide usage facilities.
The Novel Approach
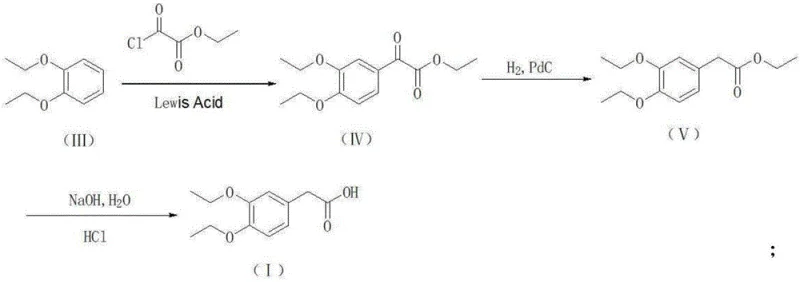
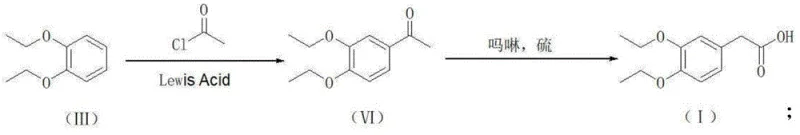
The innovative methodology presented in the patent data circumvents these legacy issues by entirely bypassing the nitrile intermediate. Instead, it employs two distinct, highly efficient pathways starting from the readily available 1,2-diethoxybenzene. The first pathway utilizes a Friedel-Crafts acylation with ethyl oxalyl chloride to form an alpha-keto ester, which is subsequently reduced via catalytic hydrogenation to yield the target acetic acid derivative. Alternatively, a second pathway employs acetyl chloride followed by a Willgerodt-Kindler reaction using morpholine and sulfur to achieve the same carboxylic acid functionality. Both routes converge on the formation of 3,4-diethoxyphenylacetic acid and its subsequent conversion to the amine via amide reduction. This strategic redesign eliminates the most toxic step of the entire value chain, thereby simplifying the process flow and enhancing the overall sustainability profile of the manufacturing operation.
Mechanistic Insights into Lewis Acid Catalyzed Acylation and Reduction
The core of this novel synthesis lies in the precise control of electrophilic aromatic substitution and subsequent functional group interconversions. In the initial Friedel-Crafts acylation step, a Lewis acid catalyst such as Aluminum Chloride (AlCl3) or Zinc Chloride (ZnCl2) activates the acyl chloride species, generating a highly reactive acylium ion. This electrophile attacks the electron-rich 1,2-diethoxybenzene ring, preferentially at the para-position relative to the ethoxy groups due to steric and electronic directing effects. The reaction conditions are meticulously controlled, typically maintaining temperatures between -10°C and 30°C depending on the specific acylating agent, to minimize poly-acylation side reactions and ensure high regioselectivity. Following acylation, the introduction of hydrogen gas in the presence of a palladium on carbon (Pd/C) catalyst facilitates the reduction of the ketone or glyoxylate moiety. This hydrogenolysis step is critical, as it cleaves the carbon-oxygen double bond to form the methylene group required for the phenylacetic acid structure, proceeding with high atom economy and minimal byproduct formation compared to stoichiometric metal reductions.
Impurity control is inherently superior in this cyanide-free design because it avoids the formation of nitrile-related degradation products and heavy metal cyanide complexes. In traditional nitrile routes, hydrolysis of the cyano group can sometimes lead to incomplete conversion or the formation of primary amide impurities that are difficult to separate. By contrast, the ester hydrolysis or Willgerodt-Kindler thioamide hydrolysis steps in the new process occur under well-defined alkaline conditions (e.g., using 5-6 mol/L NaOH), allowing for precise pH control during the final acidification to precipitate the product. The use of recrystallization from solvents like ethyl acetate or ethanol further ensures that the final 3,4-diethoxyphenylacetic acid meets stringent purity specifications (>98%), which is essential for preventing downstream contamination in the final API. This level of chemical cleanliness reduces the burden on purification units and increases the overall yield of the final drotaverine hydrochloride product.
How to Synthesize 3,4-Diethoxyphenylacetic Acid Efficiently
The synthesis of this critical intermediate involves a multi-step sequence that balances reaction kinetics with safety profiles. The process begins with the activation of the aromatic ring followed by chain extension and functional group modification. Operators must carefully manage the exothermic nature of the Lewis acid complexation and the handling of hydrogen gas during the reduction phase. The detailed standardized synthesis steps, including specific molar ratios, temperature ramps, and workup procedures, are outlined below to ensure reproducibility and safety in a GMP environment.
- Perform Friedel-Crafts acylation on 1,2-diethoxybenzene with ethyl oxalyl chloride or acetyl chloride using a Lewis acid catalyst like AlCl3 or ZnCl2.
- Execute catalytic hydrogenation on the resulting glyoxylate ester using Pd/C to form the ethyl ester, or utilize the Willgerodt-Kindler reaction with morpholine and sulfur.
- Hydrolyze the ester or thioamide intermediate under alkaline conditions followed by acidification to isolate high-purity 3,4-diethoxyphenylacetic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical feasibility. The elimination of sodium cyanide removes a Category 1 toxic substance from the bill of materials, which drastically simplifies logistics, storage, and handling requirements. Facilities no longer need to maintain specialized cyanide antidote kits or undergo frequent, high-cost safety audits specifically for cyanide storage, leading to a significant reduction in overhead operational costs. Furthermore, the raw materials for this new process, such as 1,2-diethoxybenzene and acetyl chloride or ethyl oxalyl chloride, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that often plague specialty reagent-dependent processes.
- Cost Reduction in Manufacturing: The removal of the cyanide step eliminates the need for expensive wastewater treatment systems designed to oxidize cyanide to harmless byproducts before discharge. This translates to substantially lower utility and waste disposal costs per kilogram of product. Additionally, the catalytic hydrogenation step uses recoverable palladium catalysts, and the Lewis acids can often be quenched and managed more economically than the complex salt wastes generated from nitrile hydrolysis. The overall process efficiency is improved by reducing the number of isolation steps, as the intermediates can often be telescoped or purified with simpler crystallization techniques, driving down the total cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: By relying on robust, non-hazardous chemistry, manufacturers can operate with greater continuity and less regulatory friction. The risk of production shutdowns due to safety incidents or environmental compliance violations is markedly decreased. The use of common solvents like dichloromethane, ethanol, and ethyl acetate ensures that solvent supply remains stable and cost-effective. Moreover, the scalability of Friedel-Crafts reactions is well-understood in the fine chemical industry, allowing for seamless technology transfer from pilot plants to multi-ton commercial reactors without the need for exotic equipment modifications.
- Scalability and Environmental Compliance: The process aligns perfectly with modern green chemistry initiatives, making it easier to obtain environmental permits for capacity expansion. The absence of persistent toxic pollutants in the effluent stream simplifies the environmental impact assessment (EIA) for new production lines. The Willgerodt-Kindler variant, while using sulfur, generates manageable sulfate wastes upon oxidation, which are far less regulated than cyanide. This environmental friendliness enhances the corporate social responsibility (CSR) profile of the supply chain, appealing to downstream pharmaceutical partners who are increasingly auditing their suppliers for sustainability metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel drotaverine intermediate synthesis. These answers are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on reaction conditions and expected outcomes.
Q: Why is the new method for Drotaverine intermediates considered safer than traditional routes?
A: Traditional methods rely on sodium cyanide to generate the nitrile intermediate, posing severe toxicity risks and requiring complex wastewater treatment. The novel process described in CN111170847B completely eliminates the use of cyanide, utilizing safer Friedel-Crafts acylation and hydrogenation or sulfur-based transformations instead.
Q: What are the key catalysts used in this novel synthesis pathway?
A: The process utilizes Lewis acids such as Aluminum Chloride (AlCl3), Zinc Chloride (ZnCl2), or Boron Trifluoride (BF3) for the initial acylation steps. Additionally, Palladium on Carbon (Pd/C) is employed for the critical hydrogenation step to reduce the glyoxylate derivative to the corresponding acetate ester.
Q: How does this process impact the environmental compliance of API manufacturing?
A: By avoiding cyanide-containing wastewater, the new method significantly reduces the environmental burden and regulatory compliance costs associated with hazardous waste disposal. The use of standard organic solvents and recoverable catalysts further aligns the process with green chemistry principles suitable for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Drotaverine Hydrochloride Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer, more efficient synthetic routes for high-demand pharmaceutical intermediates like those for Drotaverine Hydrochloride. Our technical team has extensively analyzed the pathways described in CN111170847B and possesses the expertise to implement these cyanide-free protocols at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our facilities are equipped with rigorous QC labs and advanced reactor systems capable of handling Lewis acid catalysis and high-pressure hydrogenation safely, guaranteeing stringent purity specifications for every batch delivered.
We invite global pharmaceutical partners to collaborate with us to leverage these technological advancements for their supply chains. By switching to our optimized manufacturing processes, you can achieve significant operational efficiencies and mitigate regulatory risks associated with toxic reagents. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term production goals for antispasmodic medications.
