Advanced Alfacalcidol Manufacturing: Overcoming Trans-Isomer Impurities via Novel Diels-Alder Strategy
Introduction to Patent CN111072540A
The pharmaceutical industry continuously seeks robust synthetic routes for critical active metabolites, and the recent disclosure in patent CN111072540A presents a transformative approach to the manufacture of alfacalcidol. As a pivotal active metabolite of vitamin D3, alfacalcidol plays an indispensable role in calcium and phosphorus homeostasis and bone mineralization processes, making its high-purity production essential for treating osteoporosis and related metabolic bone diseases. This patent introduces a refined methodology that bypasses the traditional, cumbersome acetate intermediate steps, opting instead for a direct ring-opening hydrolysis followed by a highly selective purification strategy. By fundamentally altering the synthetic pathway, this innovation addresses long-standing challenges regarding yield, stability, and impurity control that have plagued previous generations of synthesis protocols.
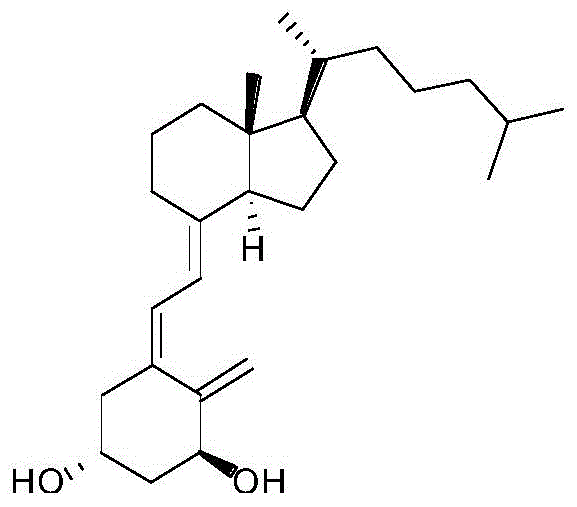
The significance of this technical advancement lies in its ability to streamline the production workflow while simultaneously enhancing the quality profile of the final active pharmaceutical ingredient. Traditional methods often struggle with the separation of geometric isomers, particularly the trans-isomer which shares similar physical properties with the target cis-alfacalcidol. The disclosed method ingeniously utilizes the differential reactivity of these isomers towards Diels-Alder dienophiles, effectively converting the problematic impurity into a separable adduct. For R&D directors and procurement specialists alike, this represents a shift towards more predictable, cost-effective, and scalable manufacturing capabilities for high-value vitamin D analogs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for alfacalcidol, as detailed in the background of the patent, rely heavily on a multi-step sequence involving the formation of a 3-acetate intermediate. This conventional pathway typically begins with the tosylation of vitamin D3, followed by cyclization and selenium dioxide oxidation to generate the 1α-hydroxy precursor. The critical bottleneck occurs during the ring-opening phase, where glacial acetic acid is used to open the 3,5-cyclized ring, necessitating a subsequent alkaline hydrolysis step using strong bases like sodium hydroxide or potassium hydroxide to remove the acetate group. This reliance on strong bases and high-temperature vacuum conditions to remove acetic acid solvents poses severe risks to the chemical integrity of the alfacalcidol molecule, which is inherently unstable and prone to degradation under such harsh environments.
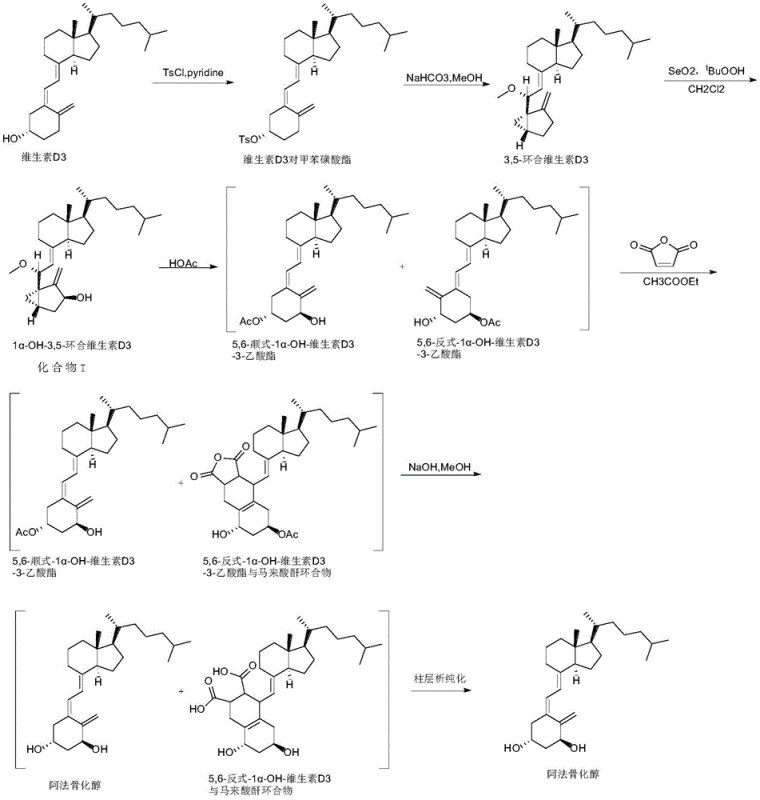
Furthermore, the conventional approach suffers from significant downstream processing inefficiencies. The generation of multiple impurities during the alkaline hydrolysis step complicates the purification landscape, often requiring repeated column chromatography or expensive preparative liquid phase chromatography to achieve pharmacopoeia-grade purity. The separation of the trans-isomer is particularly problematic; while some literature suggests photochemical conversion, this introduces additional equipment costs for photoreactors and high-pressure mercury lamps, along with the risk of generating new photo-reaction impurities. Consequently, the overall yield remains suboptimal, and the operational complexity hinders efficient scale-up, creating substantial bottlenecks for supply chain continuity and cost management in commercial manufacturing settings.
The Novel Approach
In stark contrast to the legacy methods, the novel approach disclosed in CN111072540A simplifies the synthesis by eliminating the acetate protection and deprotection cycle entirely. The process initiates with the direct ring-opening of 1α-OH-3,5-cyclized vitamin D3 (Compound I) in a mixed solvent system comprising dimethyl sulfoxide (DMSO) and an organic acid, such as acetic acid or p-toluenesulfonic acid. This reaction proceeds under mild thermal conditions, typically between 40°C and 60°C, which preserves the delicate stereochemistry of the vitamin D triene system. By avoiding the high-temperature vacuum removal of acetic acid and the subsequent strong base treatment, the new method drastically reduces the formation of degradation byproducts, leading to a cleaner crude reaction mixture that is far easier to handle in subsequent purification stages.
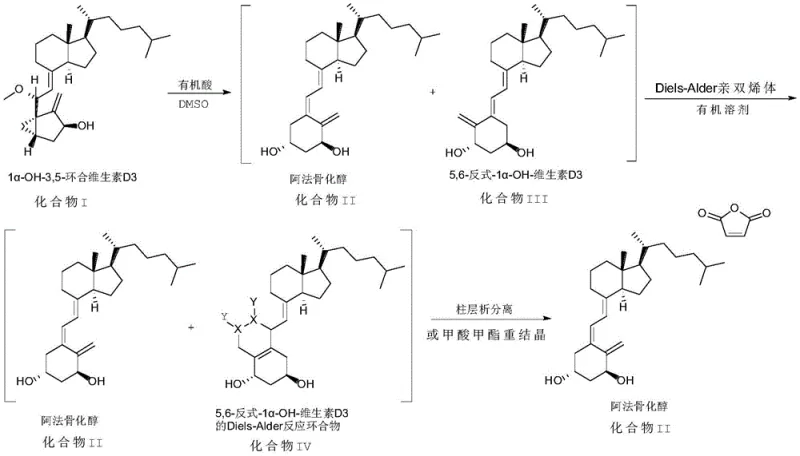
The true brilliance of this novel approach lies in its innovative purification strategy utilizing the Diels-Alder reaction. Instead of attempting to separate the cis-alfacalcidol from the trans-isomer through difficult physical means, the process chemically differentiates them. The trans-isomer selectively reacts with a Diels-Alder dienophile, such as maleic anhydride, to form a cyclic adduct, while the target cis-alfacalcidol remains inert under these conditions. This chemical derivatization creates a vast difference in polarity and molecular weight between the product and the impurity, enabling straightforward separation via standard column chromatography or even simple recrystallization with methyl formate. This strategic shift from physical separation to chemical differentiation significantly lowers the barrier to achieving high purity, making the process robust enough for industrial scale-up without the need for specialized preparative HPLC equipment.
Mechanistic Insights into DMSO-Mediated Ring Opening and Selective Diels-Alder Derivatization
The mechanistic foundation of this improved synthesis rests on the unique solvation properties of DMSO combined with the proton-donating capability of organic acids. In the ring-opening step, the polar aprotic nature of DMSO stabilizes the transition states involved in the cleavage of the 3,5-cyclo bond, while the organic acid provides the necessary protons to facilitate the hydrolysis without generating the harsh alkaline environment that triggers elimination reactions or isomerization. This mild acidic environment ensures that the 1α-hydroxyl group and the conjugated triene system remain intact, preventing the formation of pre-vitamin D or tachysterol byproducts that are common in thermal or basic treatments. The result is a high-fidelity conversion of the cyclized precursor into the desired seco-steroid framework with minimal structural erosion.
Following the ring opening, the selectivity of the Diels-Alder reaction serves as the critical impurity control mechanism. The trans-isomer of alfacalcidol possesses a s-cis diene conformation in its triene system that is sterically accessible and electronically favorable for cycloaddition with electron-deficient dienophiles like maleic anhydride or 4-phenyl-1,2,4-triazoline-3,5-dione. Conversely, the target cis-alfacalcidol adopts a conformation that is sterically hindered or electronically less reactive towards these specific dienophiles under the controlled reaction conditions (0-45°C). This kinetic selectivity allows for the quantitative conversion of the trans-impurity into a Diels-Alder adduct, effectively 'locking' it into a new chemical form.
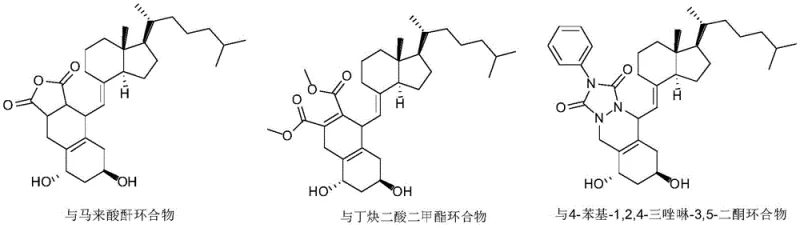
The resulting Diels-Alder adducts, as illustrated in the patent data, are structurally distinct polycyclic compounds that exhibit significantly different chromatographic retention times and solubility profiles compared to the free alfacalcidol. For instance, the adduct formed with maleic anhydride introduces rigid bicyclic structures and anhydride functionalities that alter the molecule's interaction with silica gel stationary phases. This divergence allows for the isolation of pure alfacalcidol with high efficiency. Moreover, the stability of these adducts ensures they do not revert to the trans-isomer during workup, providing a permanent sink for the impurity. This mechanistic understanding underscores the reliability of the process for producing high-purity material suitable for sensitive pharmaceutical applications.
How to Synthesize Alfacalcidol Efficiently
The synthesis of alfacalcidol via this improved method offers a practical roadmap for laboratories and manufacturing facilities aiming to optimize their production lines. The procedure begins with the dissolution of the cyclized precursor in a DMSO-organic acid mixture, followed by a controlled heating phase to effect ring opening. After neutralization and extraction, the crude mixture containing both isomers is subjected to the Diels-Alder reaction with a chosen dienophile. The final isolation step leverages the physicochemical differences created by the derivatization to yield the pure product. For detailed operational parameters, stoichiometry, and specific workup procedures, please refer to the standardized synthesis guide below.
- Perform ring-opening hydrolysis of 1α-OH-3,5-cyclized vitamin D3 in a DMSO and organic acid mixed solvent at 40-60°C to generate a mixture of alfacalcidol and its trans-isomer.
- React the resulting mixture with a Diels-Alder dienophile (such as maleic anhydride) in an organic solvent to selectively form an adduct with the trans-isomer.
- Purify the reaction mixture via column chromatography or methyl formate recrystallization to isolate pure alfacalcidol, leaving the trans-isomer adduct behind.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers profound advantages for procurement managers and supply chain heads focused on cost efficiency and reliability. The most immediate impact is the drastic simplification of the manufacturing workflow. By eliminating the acetylation and deacetylation steps, the process reduces the total number of unit operations, which directly correlates to lower labor costs, reduced solvent consumption, and decreased energy usage. The avoidance of high-temperature vacuum distillation for acetic acid removal further lowers utility costs and minimizes the thermal load on production equipment, extending asset life and reducing maintenance downtime. These cumulative efficiencies translate into a leaner cost structure for the production of this high-value intermediate.
Supply chain reliability is significantly enhanced through the robustness of the purification strategy. Traditional methods relying on preparative liquid chromatography are often bottlenecks due to low throughput and high solvent waste. The new method's reliance on standard column chromatography or recrystallization allows for much larger batch sizes and faster cycle times. Furthermore, the use of common, commercially available reagents like DMSO, acetic acid, and maleic anhydride ensures that raw material sourcing is stable and not subject to the volatility associated with specialized catalysts or exotic reagents. This stability in raw material supply mitigates the risk of production delays, ensuring a consistent flow of goods to downstream formulation partners.
Environmental compliance and scalability are also markedly improved. The reduction in solvent usage and the elimination of strong base waste streams simplify wastewater treatment protocols, aligning the process with increasingly stringent environmental regulations. The mild reaction conditions enhance operational safety, reducing the risk of thermal runaways or hazardous exotherms associated with strong base hydrolysis. This safety profile, combined with the simplified purification, makes the process highly amenable to scale-up from pilot plant to multi-ton commercial production. For organizations seeking a reliable pharmaceutical intermediates supplier, this technology represents a sustainable and scalable solution that balances economic performance with regulatory and environmental responsibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved alfacalcidol synthesis. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method resolves historical manufacturing pain points. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer or for procurement teams assessing the quality assurance capabilities of potential suppliers.
Q: How does the new method improve upon the traditional acetate intermediate route?
A: The new method eliminates the need for acetylation and subsequent strong base hydrolysis. By using a direct DMSO/organic acid ring-opening, it avoids harsh conditions that degrade the unstable alfacalcidol structure, thereby reducing impurity formation and simplifying the workflow.
Q: What is the key mechanism for removing the trans-isomer impurity?
A: The process leverages the structural difference between the cis-alfacalcidol and the trans-isomer. The trans-isomer selectively undergoes a Diels-Alder reaction with a dienophile to form a bulky adduct, while the target alfacalcidol remains unreacted, allowing for easy separation via chromatography or recrystallization.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable. It replaces complex preparative liquid chromatography with standard column chromatography or simple recrystallization. Furthermore, the mild reaction conditions (40-60°C) and avoidance of vacuum high-temperature solvent removal enhance operational safety and equipment longevity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alfacalcidol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the rigorous demands of the global pharmaceutical market. Our technical team has extensively analyzed the potential of the CN111072540A process, and we are well-positioned to leverage this technology for the commercial production of high-purity alfacalcidol. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the infrastructure and expertise to implement this DMSO-mediated ring-opening and Diels-Alder purification strategy efficiently. Our stringent purity specifications and rigorous QC labs ensure that every batch meets the highest international standards for active pharmaceutical ingredients.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain requirements. Whether you require a Customized Cost-Saving Analysis to quantify the economic benefits of switching to this newer method, or you need specific COA data to validate the purity profile against your internal standards, we are ready to assist. We encourage you to request route feasibility assessments today to explore how our advanced manufacturing capabilities can secure your supply of high-quality alfacalcidol and support your long-term product development goals.
