Advanced Catalytic Dioxonation for Phenylpyruvic Acid: Commercial Scalability and Process Optimization
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for the production of alpha-keto acids, which serve as critical building blocks for a vast array of bioactive molecules. Patent CN1112346C introduces a transformative approach to synthesizing phenylpyruvic acid and its derivatives through a novel catalytic dioxonation process. This technology addresses long-standing challenges in carbonylation chemistry by utilizing a specialized cobalt pyridine carboxylate catalyst that exhibits exceptional stability and recyclability. Unlike traditional methods that rely on hazardous or expensive catalysts, this innovation enables the direct conversion of benzyl halides and carbon monoxide into high-value keto acids under moderate industrial conditions. For R&D directors and procurement managers, this patent represents a significant opportunity to optimize supply chains for reliable phenylpyruvic acid supplier networks, ensuring consistent quality and reduced operational risks in the manufacturing of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of phenylpyruvic acid via double carbonylation has been hindered by the inherent instability and handling difficulties of conventional catalysts. The most widely studied catalysts, such as cobalt octacarbonyl (Co2(CO)8), are notoriously sensitive to oxygen and moisture, requiring stringent inert atmosphere conditions that complicate reactor design and increase operational costs. Furthermore, the preparation of cobalt octacarbonyl itself often necessitates high-pressure conditions exceeding 10.0 MPa, creating significant safety hazards and energy burdens for manufacturing facilities. Alternative palladium-based catalysts, while active, are prohibitively expensive for large-scale commodity chemical production, rendering them economically unviable for cost-sensitive applications. The separation and recovery of these traditional transition metal catalysts from the reaction mixture are also technically challenging, often leading to product contamination with heavy metals and substantial waste generation, which conflicts with modern environmental compliance standards and green chemistry initiatives.
The Novel Approach
The methodology disclosed in patent CN1112346C fundamentally overcomes these barriers by introducing a cobalt pyridine carboxylate catalyst that is both air-stable and water-tolerant. This novel catalytic system allows the dioxonation reaction to proceed efficiently at significantly lower pressures, typically between 1.0 MPa and 5.0 MPa, and moderate temperatures ranging from 25°C to 100°C. The robustness of this catalyst simplifies the engineering requirements for the reactor, reducing capital expenditure and enhancing operational safety. Crucially, the catalyst can be easily recovered from the mother liquor after the product is isolated, allowing for multiple reuse cycles without significant loss of activity. This recyclability not only drastically reduces the consumption of cobalt resources but also minimizes the environmental footprint of the process. By eliminating the need for expensive palladium and the hazardous handling protocols associated with cobalt octacarbonyl, this approach offers a compelling pathway for cost reduction in pharmaceutical intermediates manufacturing, making it highly attractive for commercial scale-up of complex fine chemical intermediates.
Mechanistic Insights into Cobalt-Catalyzed Double Carbonylation
The core of this technological advancement lies in the unique coordination chemistry of the cobalt pyridine carboxylate complex, which facilitates the insertion of two carbon monoxide molecules into the carbon-halogen bond of the benzyl halide substrate. The reaction mechanism involves the oxidative addition of the benzyl halide to the cobalt center, followed by sequential migratory insertion of carbon monoxide to form an acyl-cobalt intermediate. The presence of a nucleophilic reagent, such as calcium hydroxide or other alkaline neutralizers, plays a critical role in trapping the acyl intermediate as a stable organic salt, thereby driving the equilibrium towards product formation and preventing side reactions. This in-situ neutralization strategy is key to achieving high selectivity for the alpha-keto acid structure over potential mono-carbonylation byproducts. The ligand environment provided by the pyridine carboxylate moiety stabilizes the cobalt center against decomposition, ensuring that the catalytic cycle remains active throughout the reaction duration even in the presence of trace impurities.
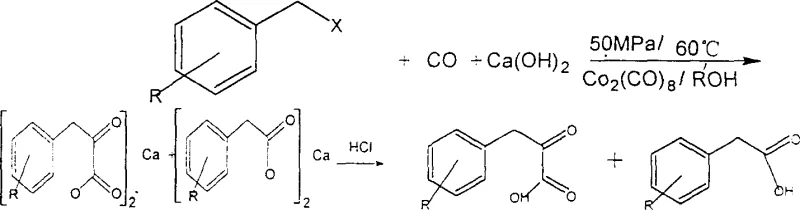
Impurity control is another critical aspect where this mechanism excels, particularly for R&D teams focused on purity and impurity profiles. The specific electronic properties of the cobalt pyridine carboxylate catalyst suppress the formation of common byproducts such as toluylic acid or ester derivatives that often plague traditional carbonylation processes. The reaction conditions allow for precise tuning of the selectivity by adjusting the solvent system, which can include methanol, ethanol, isoamyl alcohol, or aromatic hydrocarbons. The subsequent acidification step converts the isolated organic salt into the free phenylpyruvic acid with high purity, minimizing the need for extensive downstream purification. This streamlined workup process ensures that the final product meets the stringent purity specifications required for pharmaceutical applications, reducing the risk of batch rejection and ensuring consistent quality for high-purity phenylpyruvic acid supplies. The ability to tolerate various substituents on the benzyl ring further demonstrates the versatility of this mechanistic pathway for generating diverse chemical libraries.
How to Synthesize Phenylpyruvic Acid Efficiently
Implementing this synthesis route requires careful attention to the preparation of the reaction mixture and the control of gas-liquid mass transfer. The process begins by charging the reactor with the cobalt pyridine carboxylate catalyst, the benzyl halide substrate, and a suitable solvent system, often containing a mixture of alcohol and water to facilitate the neutralization step. Carbon monoxide is then introduced into the system, and the pressure is maintained within the optimal range of 1.0 MPa to 5.0 MPa while the temperature is controlled between 25°C and 100°C. The reaction progress is monitored until the conversion is complete, after which the mixture is cooled and the solid organic salt is separated via filtration. The detailed standardized synthesis steps, including specific molar ratios, stirring rates, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during technology transfer.
- Prepare the reaction mixture by dissolving the novel cobalt pyridine carboxylate catalyst and benzyl halide substrate in a suitable alcohol solvent such as methanol or isoamyl alcohol.
- Introduce carbon monoxide gas into the reactor and maintain the system pressure between 1.0 MPa and 5.0 MPa while heating the mixture to a temperature range of 25°C to 100°C.
- Neutralize the resulting reaction product with an alkaline agent like calcium hydroxide to form an organic salt, separate the salt, and subsequently acidify to isolate the final phenylpyruvic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic dioxonation technology offers substantial strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the significant reduction of raw material costs driven by the stability and recyclability of the novel cobalt catalyst. Unlike traditional catalysts that are consumed in single batches or require complex regeneration, this system allows for the recovery and reuse of the catalytic species from the mother liquor, leading to a drastic decrease in the per-kilogram cost of the catalyst. Furthermore, the moderate pressure and temperature conditions reduce energy consumption and lower the safety risks associated with high-pressure operations, which translates into lower insurance premiums and maintenance costs for the manufacturing facility. These factors combine to create a more resilient and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the ability to recycle the cobalt-based catalyst significantly lower the direct material costs associated with production. The process avoids the need for specialized high-pressure equipment required for cobalt octacarbonyl preparation, reducing capital expenditure and depreciation costs. Additionally, the simplified workup procedure reduces solvent usage and waste disposal fees, contributing to overall operational efficiency. These cumulative savings allow for a more competitive pricing structure for the final phenylpyruvic acid product, enhancing margin potential for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: The stability of the cobalt pyridine carboxylate catalyst against air and moisture simplifies logistics and storage, removing the need for specialized inert atmosphere shipping and handling. This robustness ensures that catalyst supplies are less prone to degradation during transit, guaranteeing consistent performance upon arrival at the manufacturing site. The use of readily available benzyl halides and carbon monoxide as starting materials further secures the supply chain against raw material shortages. By reducing lead time for high-purity phenylpyruvic acid derivatives, manufacturers can respond more agilely to market demand fluctuations and ensure continuous production schedules without unexpected interruptions.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard industrial unit operations and moderate reaction conditions that are easily managed in large-scale reactors. The reduced generation of heavy metal waste and the ability to recycle the catalyst align with increasingly strict environmental regulations, minimizing the risk of compliance violations. The aqueous workup and neutralization steps generate less hazardous waste compared to traditional organic extraction methods, simplifying effluent treatment. This environmental compatibility not only protects the company's reputation but also future-proofs the manufacturing process against tightening global sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic dioxonation technology. These answers are derived directly from the technical specifications and experimental data provided in the patent literature to ensure accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing lines or for sourcing high-quality intermediates from external partners. The insights provided here aim to clarify the operational advantages and technical capabilities of this novel synthetic route.
Q: What is the primary advantage of the cobalt pyridine carboxylate catalyst over traditional cobalt octacarbonyl?
A: The novel cobalt pyridine carboxylate catalyst offers superior stability against oxygen and moisture compared to cobalt octacarbonyl, which is extremely unstable and difficult to store. Additionally, the new catalyst can be easily recovered and recycled from the mother liquor, significantly reducing raw material costs and waste generation in large-scale manufacturing.
Q: What are the typical reaction conditions required for this dioxonation process?
A: According to patent CN1112346C, the reaction operates under moderate conditions, specifically at pressures ranging from 1.0 MPa to 5.0 MPa and temperatures between 25°C and 100°C. These conditions are generally safer and more energy-efficient than the high-pressure requirements often associated with traditional carbonylation methods.
Q: Can this process be adapted for substituted phenylpyruvic acid derivatives?
A: Yes, the process is highly versatile. By varying the substituents on the benzyl halide starting material (such as halogen, cyano, alkyl, or aryl groups) and adjusting the reaction conditions, manufacturers can synthesize a wide range of substituted phenylpyruvic acid derivatives, making it suitable for diverse pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylpyruvic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the global supply of essential pharmaceutical intermediates. Our technical team has extensively analyzed the catalytic dioxonation process described in patent CN1112346C and possesses the expertise to implement this technology at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and efficient. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of phenylpyruvic acid we produce. We are committed to delivering high-purity phenylpyruvic acid that meets the exacting standards of the international pharmaceutical industry.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains and reduce manufacturing costs. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Partnering with us ensures not only a reliable source of high-quality intermediates but also a strategic alliance focused on continuous process improvement and innovation. Let us help you navigate the complexities of chemical manufacturing with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
