Advanced Synthesis of Tetraphenyl Vinyl Poly-Pyrazole Compounds for High-Performance Sensing Applications
Advanced Synthesis of Tetraphenyl Vinyl Poly-Pyrazole Compounds for High-Performance Sensing Applications
The rapid advancement of supramolecular chemistry has necessitated the development of sophisticated molecular architectures capable of precise environmental monitoring and material science applications. Patent CN113248437A introduces a groundbreaking methodology for the synthesis of 1,1,2,2-tetrakis(4-(pyrazol-4-yl)phenyl)ethylene, a nitrogen-containing heterocyclic compound with the chemical formula C38H28N8. This molecule represents a significant leap forward in the design of functional materials, specifically serving as a robust hydrogen-bond acceptor for constructing organic lattice structures and as a highly sensitive recognizer for metal ions. The technical breakthrough lies in its ability to exhibit exceptional sensitivity towards mercury ions in both water and organic solvent systems, maintaining high selectivity even in the presence of interfering anions. For industrial partners seeking reliable specialty chemical suppliers, this patent outlines a scalable route that balances complex molecular assembly with practical manufacturing considerations, ensuring high purity and consistent performance for downstream applications in environmental sensing and advanced material fabrication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of multi-functionalized tetraphenylethylene derivatives has been plagued by significant challenges regarding solubility, reaction efficiency, and the harsh conditions required to install multiple heterocyclic rings. Conventional methods often rely on pure organic solvents such as DMF or DMSO, which pose substantial difficulties in product isolation and waste treatment, leading to inflated production costs and environmental compliance issues. Furthermore, existing metal ion recognition molecules frequently suffer from poor selectivity, where the presence of competing transition metals like zinc, copper, or cadmium can mask the detection signal, rendering the sensor ineffective in complex real-world matrices. The lack of robust coordination sites in simpler aromatic systems often results in weak binding affinities, requiring higher concentrations of the sensing material to achieve detectable signals, which is economically unviable for large-scale deployment in water treatment or food safety monitoring sectors.
The Novel Approach
The innovative strategy detailed in the patent overcomes these hurdles by employing a strategic Suzuki-Miyaura cross-coupling reaction followed by a mild deprotection sequence. By utilizing 1,1,2,2-tetra(4-bromophenyl)ethylene as a central scaffold and reacting it with 1-(tetrahydropyran-4-yl)-1H-pyrazole-4-boronic acid pinacol ester, the process efficiently installs four pyrazole units in a single convergent step. This approach not only streamlines the synthetic pathway but also leverages the unique electronic properties of the pyrazole ring to enhance metal coordination capabilities. The subsequent removal of the tetrahydropyran protecting group under acidic conditions reveals the active NH sites essential for hydrogen bonding and metal chelation. This novel route ensures cost reduction in specialty chemical manufacturing by minimizing step count and utilizing readily available starting materials, thereby providing a commercially viable pathway for producing high-performance sensing materials that were previously difficult to access.
Mechanistic Insights into Suzuki-Miyaura Cross-Coupling and Deprotection
The core of this synthesis relies on the palladium-catalyzed cross-coupling mechanism, where tetrakis(triphenylphosphine)palladium acts as the catalyst to facilitate the bond formation between the aryl bromide and the boronic ester. The reaction proceeds through a classic catalytic cycle involving oxidative addition of the palladium species to the carbon-bromine bond of the tetraphenylethylene core, followed by transmetallation with the activated boronate species in the presence of the potassium carbonate base. The use of a mixed solvent system comprising water and n-butanol is critical here, as it enhances the solubility of the inorganic base while maintaining the organic reactants in solution, promoting efficient mass transfer and reaction kinetics. The steric bulk of the tetraphenylethylene core requires prolonged heating, typically between 48 to 96 hours at reflux temperatures around 120°C, to ensure complete conversion of all four bromine sites, preventing the formation of partially substituted impurities that could compromise the symmetry and functionality of the final sensor molecule.
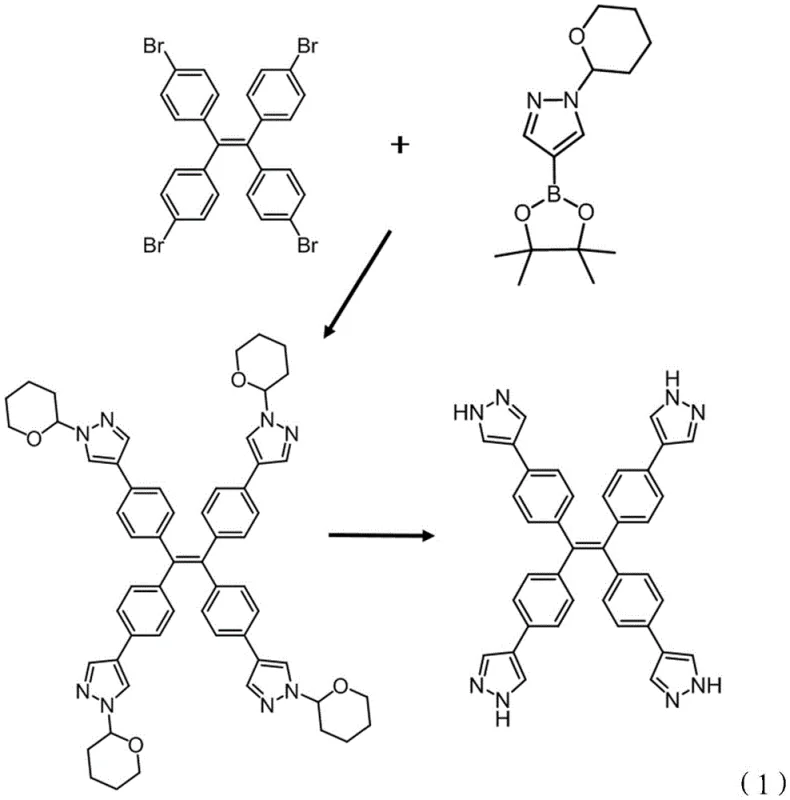
Following the coupling reaction, the mechanism shifts to an acid-catalyzed hydrolysis designed to cleave the ether linkage of the tetrahydropyran (THP) protecting groups. The addition of hydrochloric acid in a solvent like methanol or dichloromethane protonates the oxygen atom of the THP ring, facilitating the elimination of the dihydropyran byproduct and regenerating the free pyrazole NH group. This step is crucial for unlocking the molecule's hydrogen-bonding acceptor capabilities and its coordination sites for metal ions. The careful control of pH during the final neutralization step, adjusting to a range of 7-8 using sodium hydroxide, ensures that the product precipitates or remains stable without undergoing degradation or salt formation that could alter its spectroscopic properties. This precise mechanistic control allows for the production of high-purity OLED material precursors or sensing agents with minimal residual metal catalyst or acidic impurities.
How to Synthesize 1,1,2,2-Tetrakis(4-(pyrazol-4-yl)phenyl)ethylene Efficiently
The synthesis protocol described in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing the importance of reagent quality and atmospheric control. To achieve optimal yields, the reaction must be conducted under an inert gas blanket, typically nitrogen, to prevent oxidation of the palladium catalyst and the sensitive boronic ester species. The molar ratios are carefully balanced, with a slight excess of the boronic ester (8 to 16 equivalents relative to the bromide) driving the equilibrium towards the fully substituted product, while the base and catalyst loading are optimized to minimize side reactions. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during operation.
- Perform Suzuki-Miyaura coupling between 1,1,2,2-tetra(4-bromophenyl)ethylene and the boronic ester derivative using Pd(PPh3)4 catalyst in a water/n-butanol solvent system under reflux.
- Isolate the tetrahydropyran-protected intermediate via silica gel chromatography after aqueous workup.
- Execute acid hydrolysis using hydrochloric acid in methanol or dichloromethane to remove protecting groups, followed by neutralization to pH 7-8.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits in terms of operational efficiency and risk mitigation. The shift away from exotic reagents towards commodity chemicals like potassium carbonate and n-butanol significantly stabilizes the supply chain, reducing the vulnerability to market fluctuations associated with specialized fine chemicals. The ability to perform the reaction in a water-containing solvent system also simplifies the downstream processing, as the removal of inorganic salts becomes more straightforward compared to reactions performed in high-boiling polar aprotic solvents. This translates directly into reduced processing time and lower energy consumption during solvent recovery, enhancing the overall sustainability profile of the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive pure organic solvents in favor of a water/n-butanol mixture drastically lowers raw material costs and waste disposal fees. Furthermore, the use of a standard palladium catalyst, which can potentially be recovered or used in low loadings relative to the high value of the product, optimizes the cost-per-kilogram metric. By avoiding complex multi-step protection-deprotection sequences for the pyrazole ring prior to coupling, the overall process mass intensity is improved, leading to substantial cost savings in large-scale production runs without compromising the structural integrity of the final compound.
- Enhanced Supply Chain Reliability: The starting materials, specifically 1,1,2,2-tetra(4-bromophenyl)ethylene and the boronic ester derivative, are synthesized from widely available petrochemical feedstocks, ensuring a consistent and reliable supply flow. The robustness of the reaction conditions, which tolerate a range of temperatures and times without significant yield loss, provides flexibility in scheduling and batch planning. This reliability is critical for maintaining continuous production lines for downstream applications such as environmental monitoring kits or advanced polymer additives, where interruption in supply could halt critical R&D or manufacturing activities for global clients.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram commercial production due to the use of standard reflux equipment and common workup procedures like extraction and chromatography. The reduced reliance on chlorinated solvents during the main coupling step aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. Additionally, the high selectivity of the reaction minimizes the generation of hazardous byproducts, simplifying wastewater treatment and ensuring that the facility remains compliant with local and international environmental standards while delivering high-purity electronic chemical grades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this tetraphenyl vinyl poly-pyrazole compound. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on performance metrics and handling requirements. Understanding these factors is essential for integrating this material into existing quality control frameworks and application development pipelines.
Q: What is the primary application of 1,1,2,2-tetrakis(4-(pyrazol-4-yl)phenyl)ethylene?
A: This compound serves as a highly selective fluorescent sensor for mercury ions (Hg2+) in both aqueous and organic systems, exhibiting a distinct color change and significant fluorescence enhancement without interference from other metal ions.
Q: Why is the water/n-butanol solvent system preferred for the coupling reaction?
A: The use of a mixed solvent system comprising water and n-butanol significantly reduces reliance on expensive pure organic solvents, lowers environmental impact, and facilitates easier product isolation compared to traditional polar aprotic solvents.
Q: How does the pyrazole moiety contribute to metal ion recognition?
A: The pyrazole groups provide abundant coordination active sites that facilitate strong metal-organic coordination bonds, allowing the molecule to effectively bind and identify specific metal ions like mercury through supramolecular interactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1,2,2-Tetrakis(4-(pyrazol-4-yl)phenyl)ethylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance functional materials play in advancing environmental safety and material science technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our capability to handle complex heterocyclic syntheses positions us as a strategic partner for companies requiring reliable sources of advanced sensing materials and intermediate compounds.
We invite you to collaborate with us to explore the full potential of this technology in your specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D initiatives and help you secure a stable supply of this cutting-edge chemical entity.
