Advanced One-Step Reduction Technology for Commercial Production of High-Purity Thiol Intermediates
The global demand for high-quality sulfur-containing terpenes, particularly those imparting tropical and fruity notes, has driven significant innovation in synthetic methodology. Patent CN112110840A introduces a transformative preparation method for 3,7-dimethyl-3-mercapto-6-octenol, a critical monoterpenoid thiol utilized extensively in the flavors, fragrances, and pharmaceutical sectors. This technology addresses long-standing inefficiencies in traditional synthesis routes by employing a robust one-step reduction strategy using lithium aluminum hydride (LiAlH4). Unlike conventional approaches that struggle with chemoselectivity and require multiple processing stages, this novel protocol achieves simultaneous reduction of aldehyde and acetylthio groups under mild conditions. For industry leaders seeking a reliable fragrance intermediate supplier, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms. The ability to synthesize this complex molecule at room temperature without the need for protective atmospheres or cryogenic cooling significantly lowers the barrier for commercial adoption. Furthermore, the process utilizes readily available raw materials and avoids toxic reagents like pyridine, aligning perfectly with modern green chemistry initiatives and rigorous environmental compliance standards required by top-tier multinational corporations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,7-dimethyl-3-mercapto-6-octenol has been plagued by significant technical hurdles that impede efficient large-scale production. Prior art, such as the methods described by Robert J. Canon et al., typically relies on a multi-step sequence involving the initial formation of a thioester followed by a selective reduction. A major bottleneck in these traditional routes is the use of sodium borohydride (NaBH4) as the reducing agent. While effective for reducing aldehydes, sodium borohydride lacks the necessary reducing power to cleave the acetylthio group, leading to incomplete conversion and the accumulation of unwanted intermediates. Consequently, manufacturers are forced to employ harsh reaction conditions, often requiring temperatures as low as 0°C and the use of environmentally unfriendly bases like pyridine. These苛刻 conditions not only increase energy consumption but also complicate the workup procedure, resulting in lower overall yields and a higher burden on waste treatment facilities. The necessity for slow, dropwise addition of reagents to control exothermicity further extends batch times, creating bottlenecks in production schedules and increasing labor costs. For procurement managers focused on cost reduction in flavor and fragrance manufacturing, these inefficiencies translate directly into higher unit prices and less predictable supply chains.
The Novel Approach
The patented methodology offers a decisive break from these limitations by leveraging the superior reducing capability of lithium aluminum hydride in anhydrous ether. This approach enables the concurrent reduction of both the aldehyde carbonyl and the acetylthio moiety in a single operational step, effectively collapsing a multi-stage process into a streamlined one-pot reaction. By operating at room temperature, the process eliminates the need for expensive cryogenic equipment and the associated energy costs of maintaining sub-zero environments. The protocol allows for the co-feeding of reactants, avoiding the time-consuming and safety-critical requirement of slow dropwise addition, which dramatically accelerates the reaction throughput. This simplification of the operational workflow not only enhances safety by minimizing operator exposure to reactive intermediates but also significantly improves the reproducibility of the synthesis. The result is a robust process capable of delivering high-purity products with yields ranging from 75.39% to 88.04% for the crude material, which can be further purified to exceed 98.00% purity. This technological leap provides a compelling value proposition for partners seeking the commercial scale-up of complex fragrance intermediates, ensuring a steady supply of high-quality material with reduced lead times.
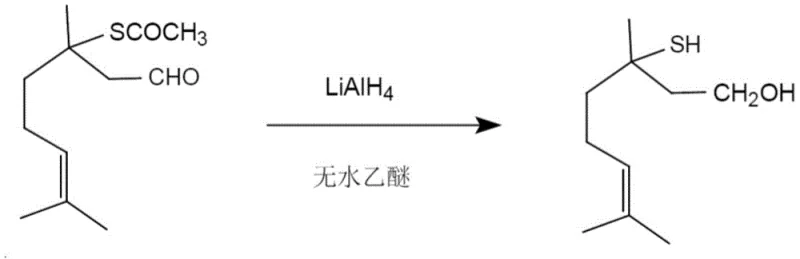
Mechanistic Insights into LiAlH4-Mediated Dual Reduction
The core of this technological advancement lies in the mechanistic efficiency of lithium aluminum hydride as a nucleophilic hydride source. In the anhydrous ether solvent system, LiAlH4 dissociates to provide highly reactive hydride ions that attack electrophilic centers within the substrate molecule. The reaction initiates with the nucleophilic addition of hydride to the aldehyde carbonyl carbon, forming an alkoxide intermediate that is subsequently protonated during the acidic workup to yield the primary alcohol functionality. Simultaneously, and crucially, the hydride species attacks the carbonyl carbon of the acetylthio group. This attack triggers the elimination of the thiolate leaving group, which is then protonated to form the free thiol (-SH) group. This dual-reduction mechanism is thermodynamically favorable and kinetically rapid at ambient temperatures, bypassing the activation energy barriers that hinder weaker reducing agents like sodium borohydride. The choice of anhydrous ether as the solvent is critical, as it stabilizes the aluminum complexes formed during the reaction and prevents premature decomposition of the hydride reagent by moisture. Understanding this mechanism is vital for R&D directors focusing on purity and impurity profiles, as it explains the absence of partially reduced by-products that typically contaminate batches produced via older methods.
Impurity control is inherently built into this synthetic design through the specificity of the reagent and the simplicity of the workup. Traditional methods often generate side products due to the prolonged exposure of sensitive functional groups to basic conditions or the incomplete reduction of the thioester. In contrast, the LiAlH4 reduction proceeds cleanly to the fully reduced alcohol-thiol state. The subsequent acid wash step serves a dual purpose: it quenches any unreacted lithium aluminum hydride safely and neutralizes the basic aluminum salts, facilitating their removal into the aqueous phase. The use of hydrochloric acid for this wash is particularly advantageous due to its low cost and effectiveness in breaking up emulsions that might form with aluminum residues. Following extraction and drying with agents like anhydrous magnesium sulfate, the crude product is obtained with a high degree of chemical integrity. Final purification via column chromatography using a petroleum ether and ethyl acetate gradient ensures the removal of any trace non-polar impurities or residual solvents. This rigorous purification protocol guarantees that the final 3,7-dimethyl-3-mercapto-6-octenol meets the stringent quality specifications demanded by the fine chemical industry, minimizing the risk of off-notes in the final fragrance application.
How to Synthesize 3,7-Dimethyl-3-Mercapto-6-Octenol Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and safety protocols, particularly given the reactivity of lithium aluminum hydride. The patent outlines a clear procedure where the molar ratio of the substrate to the reducing agent is optimized between 1:1 and 1:4 to balance cost and conversion efficiency. Operating outside this range can lead to either incomplete reaction or unnecessary expenditure on the more expensive hydride reagent. The reaction is conducted in anhydrous ether, necessitating strict moisture control to prevent hazardous hydrogen gas evolution. Once the reaction is complete, typically within 2 to 5 hours of stirring, the mixture is subjected to a controlled acid quench. The detailed standardized synthesis steps, including specific quantities for pilot and commercial scales, are outlined in the technical guide below.
- Co-feed 3,7-dimethyl-3-acetylthio-6-octenal and lithium aluminum hydride into anhydrous ether and stir at room temperature for 2 to 5 hours.
- Wash the reaction mixture with an acidic solution, such as hydrochloric acid, until the pH reaches neutrality to quench excess hydride.
- Extract the organic phase, dry over anhydrous magnesium sulfate, concentrate via rotary evaporation, and purify using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical superiority. The elimination of multi-step processing and the removal of toxic reagents like pyridine directly correlate to a significant reduction in operational expenditures. By consolidating the synthesis into a single reaction vessel, manufacturers can drastically reduce the consumption of solvents, energy, and labor hours per kilogram of product. This streamlining of the process flow enhances the overall asset utilization of production facilities, allowing for greater throughput without the need for capital-intensive infrastructure upgrades. Furthermore, the reliance on room-temperature conditions mitigates the risks associated with thermal runaways and simplifies the engineering controls required for safe operation. These factors collectively contribute to a more resilient and cost-competitive supply chain, ensuring that buyers can secure high-purity monoterpenoid thiols at a more stable price point. The robustness of the method also means fewer batch failures and less variability in quality, which is crucial for maintaining consistent production schedules in downstream applications.
- Cost Reduction in Manufacturing: The shift from a multi-step process involving expensive bases and specialized cooling to a one-pot room-temperature reaction fundamentally alters the cost structure of production. By removing the need for cryogenic cooling systems and reducing the total reaction time, energy costs are substantially lowered. Additionally, the avoidance of pyridine eliminates the costly disposal fees associated with hazardous nitrogenous waste streams. The use of lithium aluminum hydride, while potent, is utilized efficiently in a stoichiometric ratio that prevents waste, and the simplified workup reduces the volume of solvents required for extraction and purification. These cumulative efficiencies drive down the cost of goods sold, allowing for more competitive pricing strategies in the global market without compromising on margin.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that are prone to bottlenecks or equipment failures. This simplified protocol reduces the number of unit operations, thereby decreasing the probability of mechanical failure or human error during transfer steps. The use of common, commercially available solvents like diethyl ether and petroleum ether ensures that raw material sourcing remains stable and unaffected by niche supply shocks. Moreover, the shorter cycle time per batch means that inventory turnover can be accelerated, enabling suppliers to respond more rapidly to fluctuations in market demand. For supply chain planners, this translates to reduced lead times for high-purity thiol compounds and a lower safety stock requirement, freeing up working capital.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden challenges, but this method is inherently designed for industrialization. The exotherm is manageable at room temperature, and the one-pot nature minimizes the need for intermediate isolation, which is a common source of yield loss and contamination at scale. From an environmental perspective, the process generates less hazardous waste and avoids the use of persistent organic pollutants. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site. Companies prioritizing sustainability in their vendor selection criteria will find this production method highly attractive, as it supports corporate social responsibility goals while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,7-dimethyl-3-mercapto-6-octenol. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions regarding formulation compatibility and sourcing strategies. The answers reflect the consensus on best practices for handling sulfur-containing terpenes and optimizing their integration into complex fragrance accords.
Q: Why is Lithium Aluminum Hydride preferred over Sodium Borohydride for this synthesis?
A: Sodium borohydride lacks sufficient reducing power to cleave the acetylthio group, often resulting in incomplete reduction and requiring harsh multi-step conditions. Lithium aluminum hydride effectively reduces both the aldehyde and the thioester functionalities in a single operation at room temperature.
Q: What are the purity specifications achievable with this method?
A: Following column chromatography purification, the final product consistently achieves a purity greater than 98.00%, meeting stringent requirements for high-end fragrance and pharmaceutical applications.
Q: Does this process require cryogenic conditions?
A: No, a key advantage of this patented method is that the reaction proceeds efficiently at room temperature, eliminating the need for energy-intensive cooling below 10°C and simplifying industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,7-Dimethyl-3-Mercapto-6-Octenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced fragrance intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We understand the nuances of handling reactive hydride reagents and sulfur compounds, implementing stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards. Our facility is equipped to manage the specific safety requirements of this synthesis, providing a secure and reliable source for your critical raw materials.
We invite you to collaborate with us to leverage this cutting-edge technology for your product portfolio. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the superior quality and economic advantages of our 3,7-dimethyl-3-mercapto-6-octenol supply. Let us help you optimize your supply chain and bring exceptional tropical notes to your next flagship fragrance.
