Advanced One-Step Synthesis of 4H-5-(1-Hydroxy-1-Methylethyl)-2-Methyl-2-Furfural for Industrial Scale-Up
Introduction to Patent CN112125871A
The global demand for high-quality synthetic fragrance ingredients continues to drive innovation in process chemistry, particularly for complex heterocyclic compounds like 4H-5-(1-hydroxy-1-methylethyl)-2-methyl-2-furfural. This valuable molecule, known for its fruity and wine-like olfactory profile, is a critical component in fine perfumery and flavor formulations. Recent advancements documented in patent CN112125871A introduce a groundbreaking preparation method that fundamentally shifts the manufacturing paradigm from multi-step laboratory procedures to a streamlined, industrially viable one-step synthesis. By utilizing 6,7-epoxy citral as a readily available starting material and employing a mild sulfuric acid catalyst in dichloromethane, this technology eliminates the need for harsh reaction conditions and expensive stoichiometric oxidants. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for reliable synthetic flavors & fragrances supplier partnerships, ensuring consistent quality while drastically reducing the environmental footprint associated with traditional synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 4H-5-(1-hydroxy-1-methylethyl)-2-methyl-2-furfural was heavily reliant on the methodology reported by Grein et al. in 1994, which presented substantial barriers to commercial scalability and cost efficiency. The legacy process necessitated a cumbersome three-step sequence beginning with the epoxidation of linalool using meta-chloroperoxybenzoic acid (m-CPBA), followed by acid-catalyzed cyclization and subsequent reduction with borane. The final step involved oxidation using pyridinium chlorochromate (PCC), a toxic chromium-based reagent that poses severe environmental and disposal challenges. Furthermore, the requirement for HPLC separation of isomers and the generation of numerous by-products resulted in low overall yields and complex purification workflows. These factors collectively contributed to high production costs and inconsistent batch-to-batch quality, making cost reduction in flavor & fragrance manufacturing nearly impossible under the old regime.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data leverages a direct acid-catalyzed cyclization strategy that simplifies the entire production workflow into a single operational unit. By reacting 6,7-epoxy citral directly with an aqueous sulfuric acid solution in dichloromethane at room temperature, the process bypasses the need for intermediate isolation and hazardous redox reagents entirely. This one-pot methodology not only accelerates the reaction timeline but also inherently improves the impurity profile by minimizing side reactions associated with multiple workup stages. The ability to perform the reaction at ambient temperature without the need for cryogenic cooling or precise dropwise addition protocols significantly lowers energy consumption and equipment complexity. For supply chain heads, this translates to a robust and scalable process capable of commercial scale-up of complex fragrance intermediates, ensuring reliable delivery schedules and reduced dependency on specialized reagent supply chains.
Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this technological breakthrough lies in the efficient protonation of the epoxide moiety within the 6,7-epoxy citral substrate, which triggers a cascade of intramolecular rearrangements leading to the formation of the furan ring. Under the influence of the sulfuric acid catalyst, the epoxide oxygen is protonated, increasing the electrophilicity of the adjacent carbon atoms and facilitating ring opening. This generates a carbocation intermediate that is stabilized by the neighboring allylic system, allowing for a nucleophilic attack by the carbonyl oxygen of the aldehyde group. This intramolecular cyclization forms a hemiacetal intermediate, which subsequently undergoes dehydration to establish the aromatic character of the furan ring system. The mild acidity of the 20% sulfuric acid solution is critical; it is strong enough to drive the cyclization forward but sufficiently controlled to prevent the degradation of the sensitive aldehyde functionality or the dehydration of the tertiary alcohol group, which would lead to unwanted conjugated diene by-products.
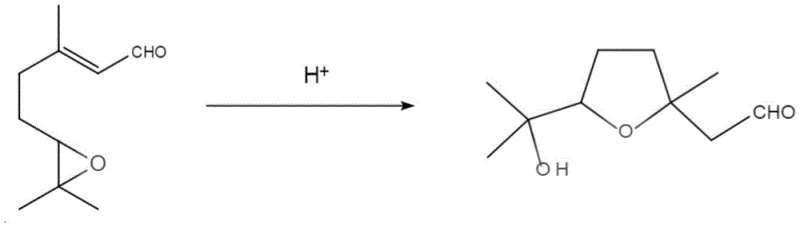
Controlling the impurity profile in this reaction is paramount for achieving the high-purity standards required in the fragrance industry, and the mechanistic pathway offers inherent selectivity advantages. The use of dichloromethane as a solvent provides an optimal polarity environment that stabilizes the transition states involved in the cyclization while keeping the organic product soluble for easy extraction. Unlike the oxidative conditions of the prior art, which often led to over-oxidation or chlorinated impurities from m-CPBA, this acid-mediated route produces a cleaner crude reaction mixture. The subsequent workup involving a simple alkaline wash effectively neutralizes residual acid and removes water-soluble impurities, while the final column chromatography step using petroleum ether and ethyl acetate ensures the removal of any remaining regioisomers. This rigorous control over the reaction environment ensures that the final product meets stringent purity specifications, typically exceeding 98.00%, which is essential for maintaining the delicate olfactory balance in high-end perfume compositions.
How to Synthesize 4H-5-(1-Hydroxy-1-Methylethyl)-2-Methyl-2-Furfural Efficiently
Implementing this synthesis route in a pilot or production plant requires adherence to specific operational parameters to maximize yield and safety. The process begins with the simultaneous addition of 6,7-epoxy citral and the aqueous acid catalyst into the solvent, a technique that prevents localized exotherms and ensures uniform reaction kinetics throughout the vessel. Following the reaction period of 1 to 3 hours at room temperature, the mixture is subjected to a straightforward aqueous workup to remove the catalyst, followed by drying and concentration. The detailed standardized synthesis steps, including specific molar ratios, washing protocols, and chromatography conditions, are outlined below to guide your technical team in replicating this high-efficiency process.
- Simultaneously mix 6,7-epoxy citral and 20% aqueous sulfuric acid in dichloromethane at room temperature and stir for 1-3 hours.
- Wash the reaction mixture with 5-20% sodium hydroxide solution to neutralize the acid and separate the organic phase.
- Extract with diethyl ether, dry over anhydrous magnesium sulfate, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis method offers transformative benefits for procurement managers and supply chain directors seeking to optimize their sourcing strategies for fragrance ingredients. The elimination of expensive and hazardous reagents such as m-CPBA, borane, and PCC directly correlates to a significant reduction in raw material costs and waste disposal fees. Furthermore, the simplification of the process from three distinct chemical transformations to a single step drastically reduces labor hours, reactor occupancy time, and utility consumption, leading to substantial cost savings in flavor & fragrance manufacturing. The robustness of the room-temperature operation also mitigates the risk of production delays caused by equipment failure in cooling systems, thereby enhancing the overall reliability of the supply chain.
- Cost Reduction in Manufacturing: The economic impact of switching to this one-step protocol is profound, primarily driven by the removal of stoichiometric oxidants and reductants which are both costly and difficult to handle on a large scale. By utilizing inexpensive sulfuric acid and common solvents like dichloromethane and diethyl ether, the variable cost per kilogram of the final product is drastically lowered. Additionally, the higher crude yields observed in this method, ranging significantly above traditional benchmarks, mean that less starting material is required to produce the same amount of finished goods, further amplifying the cost efficiency. This lean manufacturing approach allows for more competitive pricing structures without compromising on the quality of the high-purity fragrance intermediate delivered to the end user.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved due to the reliance on 6,7-epoxy citral, a commercially available and stable starting material that is easier to source in bulk quantities compared to the specialized reagents of the past. The simplified process flow reduces the number of potential bottlenecks, as there are fewer intermediate handling steps where material loss or contamination could occur. Moreover, the absence of strict temperature controls (such as sub-zero cooling) means that production can be maintained consistently across different seasons and geographical locations without requiring specialized infrastructure. This flexibility ensures reducing lead time for high-purity fragrance intermediates, allowing manufacturers to respond more agilely to market fluctuations and urgent customer demands.
- Scalability and Environmental Compliance: Scaling this process from laboratory benchtop to multi-ton production is inherently safer and more straightforward due to the mild reaction conditions and the absence of explosive or highly toxic reagents. The environmental profile of the process is superior, as it avoids the generation of heavy metal waste associated with chromium oxidants and reduces the overall solvent load through higher efficiency. This alignment with green chemistry principles facilitates easier regulatory compliance and permits acquisition in regions with strict environmental laws. Consequently, manufacturers can achieve commercial scale-up of complex fragrance intermediates with confidence, knowing that the process is sustainable, safe, and ready for long-term industrial deployment.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this advanced synthesis technology, we have compiled a set of frequently asked questions based on the patent data and practical implementation experience. These answers provide clarity on the operational benefits, purity profiles, and scalability of the method, helping stakeholders make informed decisions about integrating this route into their production portfolios. Understanding these details is crucial for maximizing the value proposition of this technology in a competitive market landscape.
Q: What are the primary advantages of this new synthesis method over the traditional Grein method?
A: The new method reduces the synthesis from three complex steps involving hazardous reagents like m-CPBA and PCC to a single room-temperature step, significantly lowering operational costs and safety risks.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates at room temperature without the need for cryogenic cooling or slow dropwise addition, making it highly scalable and energy-efficient for commercial manufacturing.
Q: What purity levels can be achieved with this protocol?
A: Following column chromatography purification, the final product achieves a purity greater than 98.00%, meeting stringent specifications for high-end fragrance applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4H-5-(1-Hydroxy-1-Methylethyl)-2-Methyl-2-Furfural Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial realities for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial volume is seamless and efficient. We are committed to delivering this high-value fragrance intermediate with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to manage complex chemistries like the acid-catalyzed cyclization described here positions us as a strategic partner for companies seeking to secure their supply of essential flavor and fragrance building blocks.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific product lines. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this method for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that you have all the necessary information to move forward with confidence in your sourcing strategy.
