Revolutionizing Cefuroxime Intermediate Synthesis via Green Dehydration of 2-Keto-Sugar Acids
The pharmaceutical industry continuously seeks robust and environmentally sustainable pathways for synthesizing critical antibiotic intermediates, particularly those serving the massive cephalosporin market. Patent CN112724107A introduces a groundbreaking preparation method for alpha-oxo-2-furyl acetic acid and its esters, which serve as pivotal precursors for the synthesis of furylammonium salts used in producing second-generation cephalosporins like Cefuroxime. This technology represents a paradigm shift by utilizing 2-keto-sugar acids, readily available biological fermentation products, as the starting materials. By employing a direct acid-catalyzed dehydration strategy, this novel approach circumvents the severe safety hazards and environmental burdens associated with legacy synthetic routes. The structural relationship between the target acid and its downstream ammonium salt derivative is fundamental to understanding its value in the antibiotic supply chain.
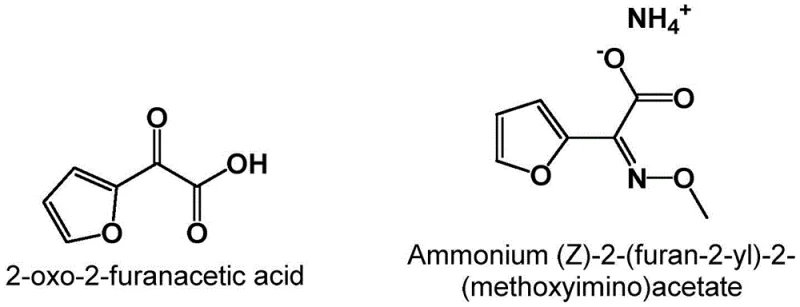
The strategic importance of this innovation lies in its ability to streamline the production of high-purity intermediates while adhering to increasingly stringent global environmental regulations. For R&D directors and process chemists, the transition from multi-step hazardous syntheses to a concise dehydration protocol offers a compelling opportunity to optimize manufacturing efficiency. The patent details a versatile methodology that accommodates various solvent systems, including water, organic solvents, and ionic liquids, providing flexibility for scale-up operations. This report analyzes the technical merits of this dehydration technology, evaluating its potential to redefine the cost structure and supply reliability of alpha-oxo-2-furyl acetic acid for the global pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of alpha-oxo-2-furyl acetic acid has been plagued by significant safety and environmental challenges inherent to the traditional cyano-recarburization and oxidation pathways. The conventional route typically initiates with furoic acid or furan, proceeding through a chlorination step to generate furoyl chloride, followed by a dangerous cyanidation reaction using sodium cyanide or potassium cyanide to form furoyl cyanide. This reliance on cyanide reagents introduces extreme toxicity risks, requiring specialized containment infrastructure and rigorous safety protocols to prevent lethal exposure. Furthermore, the subsequent hydrolysis step generates substantial quantities of hydrogen chloride and sulfur dioxide gases, necessitating complex scrubbing systems to manage corrosive off-gases.

Alternative oxidative methods, such as the oxidation of acetylfuran using nitrites, present a different set of formidable obstacles. These processes often suffer from poor selectivity, leading to deep oxidation side reactions that drastically reduce overall yield. A critical drawback of these oxidation routes is the concomitant generation of nitrogen oxides (NOx), which are potent greenhouse gases and regulated pollutants. Additionally, the wastewater generated from these traditional processes is characterized by high salt content and Chemical Oxygen Demand (COD) levels that frequently exceed regulatory discharge standards. The combination of toxic reagents, hazardous gas emissions, and difficult-to-treat wastewater renders these conventional methods increasingly unsustainable in the modern regulatory landscape.
The Novel Approach
In stark contrast to the hazardous legacy processes, the novel method disclosed in the patent utilizes a direct dehydration of 2-keto-sugar acids, offering a remarkably cleaner and safer synthetic trajectory. This innovative route leverages the structural proximity of 2-keto-sugar acids to the target furan ring system, allowing for a concise transformation driven by acid catalysis. By simply heating the 2-keto-sugar acid substrate in the presence of an acid catalyst within a suitable solvent, the molecule undergoes dehydration to directly form the alpha-oxo-2-furyl acetic acid skeleton or its corresponding ester. This one-step transformation eliminates the need for toxic cyanide sources, chlorinating agents, and nitrite oxidants entirely.
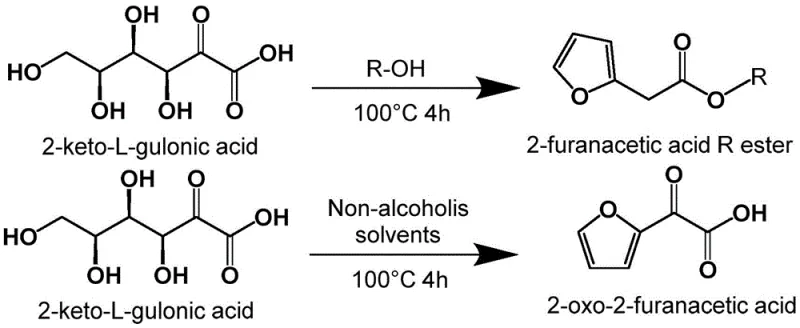
The versatility of this new approach is further enhanced by its compatibility with a wide range of reaction media, including alcoholic solvents for direct ester formation or non-alcoholic solvents for acid production. The ability to tune the solvent system allows manufacturers to optimize the process for either direct isolation of the acid or the ester, depending on downstream requirements. Moreover, because the starting materials are fermentation products, the process inherently aligns with green chemistry principles by utilizing renewable feedstocks. The elimination of heavy metal catalysts and toxic reagents significantly simplifies the purification workflow, reducing the burden on downstream processing units and minimizing the generation of hazardous solid waste.
Mechanistic Insights into Acid-Catalyzed Dehydration
The core of this technological advancement rests on the efficient acid-catalyzed dehydration mechanism that converts the polyhydroxy structure of 2-keto-sugar acids into the aromatic furan ring of the target product. The reaction proceeds through a series of protonation and elimination steps where the acid catalyst facilitates the removal of water molecules from the sugar acid backbone. Strong acids, such as concentrated sulfuric acid, p-toluenesulfonic acid, or methanesulfonic acid, act as proton donors to activate the hydroxyl groups, making them better leaving groups. As the reaction temperature is elevated, typically between 80°C and 130°C, the kinetic energy drives the elimination of water, leading to cyclization and aromatization to form the stable furan ring system.
Crucially, the patent highlights the broad substrate scope of this dehydration mechanism, demonstrating its applicability across various stereoisomers of 2-keto-sugar acids. The process effectively converts 2-keto-L-gulonic acid, 2-keto-D-gluconic acid, 2-keto-D-galactonic acid, and 2-keto-D-allose acid into the desired product. Among these, 2-keto-L-gulonic acid and its salts, which are key intermediates in Vitamin C production, exhibit superior reactivity and yield profiles. This mechanistic robustness ensures that manufacturers can utilize commercially abundant fermentation intermediates without being constrained by specific stereochemical requirements, thereby enhancing supply chain flexibility.
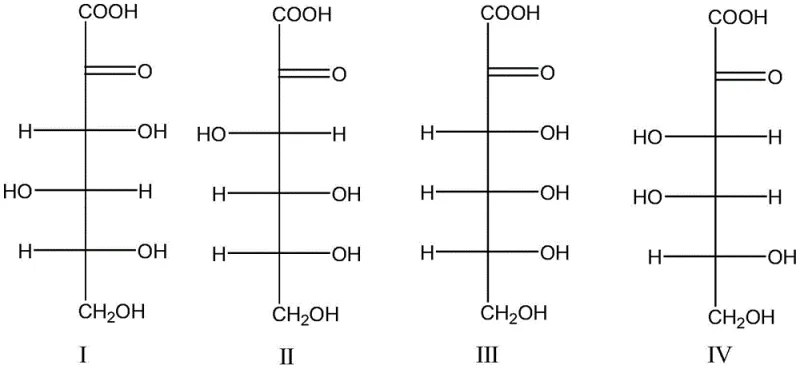
Impurity control in this dehydration process is inherently superior to oxidation routes due to the absence of radical-mediated side reactions. In traditional nitrite oxidations, uncontrolled radical species often attack the furan ring or the side chain, generating complex mixtures of over-oxidized byproducts that are difficult to separate. In the acid-catalyzed dehydration pathway, the reaction is governed by ionic mechanisms that are more predictable and controllable. While excessive acidity or temperature can lead to carbonization or polymerization, these parameters are easily optimized. The patent data indicates that maintaining a specific molar ratio of acid catalyst to substrate and controlling the temperature window prevents severe degradation, resulting in a cleaner crude product profile that requires less intensive purification.
How to Synthesize Alpha-Oxo-2-Furyl Acetic Acid Efficiently
The operational simplicity of this dehydration protocol makes it highly attractive for immediate adoption in pilot and commercial plants. The general procedure involves dissolving the selected 2-keto-sugar acid substrate in a solvent such as n-butanol (for esters) or dimethyl sulfoxide (for acids), followed by the addition of a catalytic amount of strong acid. The mixture is then heated under reflux or in a pressure-resistant vessel to drive the dehydration to completion. Detailed standard operating procedures regarding specific stoichiometry, temperature ramping, and workup protocols are essential for maximizing yield and ensuring reproducibility.
- Select a suitable 2-keto-sugar acid substrate, such as 2-keto-L-gulonic acid, and dissolve it in an appropriate solvent system like n-butanol or dimethyl sulfoxide.
- Introduce a strong acid catalyst, such as concentrated sulfuric acid or p-toluenesulfonic acid, to the reaction mixture to initiate the dehydration process.
- Heat the reaction mixture to a temperature between 80°C and 130°C for several hours to facilitate the conversion, followed by standard extraction and purification protocols.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this dehydration technology translates into tangible strategic advantages regarding cost stability and regulatory compliance. The most significant benefit is the complete elimination of cyanide-based reagents from the supply chain. Traditional methods require the procurement, storage, and disposal of highly regulated toxic chemicals, which incur substantial costs related to safety infrastructure, insurance, and hazardous waste management. By switching to a process that relies on benign acid catalysts and fermentation-derived sugars, manufacturers can drastically reduce their operational expenditure related to EHS (Environment, Health, and Safety) compliance.
- Cost Reduction in Manufacturing: The economic model of this new process is strengthened by the removal of expensive and hazardous reagents. Traditional cyano-recarburization requires stoichiometric amounts of sodium cyanide and chlorinating agents like thionyl chloride, the prices of which are volatile and subject to strict regulatory controls. Furthermore, the waste treatment costs for cyanide-containing effluent are exorbitant due to the need for specialized detoxification processes. In contrast, the dehydration route generates wastewater with significantly lower toxicity and salt content, allowing for more straightforward and cost-effective treatment. The simplified purification process also reduces solvent consumption and energy usage during distillation and crystallization steps.
- Enhanced Supply Chain Reliability: Relying on fermentation products like 2-keto-L-gulonic acid as raw materials insulates the supply chain from the volatility of petrochemical feedstocks. These sugar acids are produced on a massive global scale for the Vitamin C industry, ensuring a stable and abundant supply. Unlike the oxidation route, which depends on nitrites and generates NOx gases that can trigger production shutdowns due to environmental violations, this dehydration process operates cleanly. This reliability minimizes the risk of unplanned downtime caused by regulatory inspections or waste disposal bottlenecks, ensuring consistent delivery of critical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The scalability of this process is supported by its use of standard unit operations such as heating, stirring, and liquid-liquid extraction, which are easily transferable from laboratory to industrial scale. The absence of gaseous byproducts like hydrogen cyanide or nitrogen oxides simplifies the reactor design, removing the need for complex gas scrubbing towers. This facilitates faster scale-up timelines and reduces capital expenditure for new production lines. Additionally, the process aligns with green chemistry metrics by utilizing renewable feedstocks and generating less hazardous waste, positioning the manufacturer favorably for sustainability audits and eco-label certifications required by major pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dehydration technology. These answers are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a factual basis for decision-making.
Q: What are the primary safety advantages of this new dehydration method over traditional cyanide routes?
A: The new method eliminates the use of highly toxic sodium cyanide or potassium cyanide required in traditional cyano-recarburization processes, thereby removing the risk of cyanide exposure and the generation of hazardous hydrogen cyanide gas.
Q: Which 2-keto-sugar acid substrates are compatible with this synthesis protocol?
A: The process is versatile and supports various fermentation-derived substrates including 2-keto-L-gulonic acid, 2-keto-D-gluconic acid, 2-keto-D-galactonic acid, and 2-keto-D-allose acid, with 2-keto-L-gulonic acid showing particularly high yields.
Q: How does this process impact environmental compliance regarding wastewater?
A: Unlike oxidation routes that generate nitrogen oxides (NOx) and high-salt wastewater with excessive COD, this dehydration route produces significantly cleaner effluent, simplifying waste treatment and reducing environmental liability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Oxo-2-Furyl Acetic Acid Supplier
The transition to greener, safer, and more efficient synthetic routes is no longer optional but a strategic imperative for the modern pharmaceutical supply chain. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced process chemistry to deliver high-value intermediates with unmatched consistency. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of this dehydration technology can be seamlessly translated into robust industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-oxo-2-furyl acetic acid meets the exacting standards required for antibiotic synthesis.
We invite forward-thinking partners to collaborate with us to capitalize on the efficiencies offered by this novel patent technology. By integrating this dehydration route into your supply chain, you can achieve significant operational improvements and cost optimizations. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can support your production goals with reliability and excellence.
