Enzymatic Synthesis of Chiral Dihydrothiophenes: A Sustainable Route for Pharmaceutical Intermediates
Introduction to Green Enzymatic Catalysis in Fine Chemical Synthesis
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for sustainable and environmentally benign processes. Patent CN105755066B introduces a groundbreaking methodology for the synthesis of chiral dihydrothiophene compounds, utilizing porcine pepsin as a biocatalyst for asymmetric thia-Michael/aldol tandem reactions. This innovation represents a pivotal shift away from traditional transition-metal catalysis towards bio-organic synthesis, offering a pathway to high-value pharmaceutical intermediates with reduced ecological footprints. The patent details a robust protocol where alpha,beta-unsaturated aldehydes react with mercaptoacetaldehyde precursors under mild aqueous-organic conditions, achieving impressive stereocontrol without the toxicity associated with heavy metals. For R&D directors and procurement specialists, this technology signals a new era of cost reduction in pharmaceutical intermediate manufacturing, where biological efficiency meets chemical precision. By leveraging the inherent chirality of enzymes, this method bypasses the need for expensive chiral ligands, thereby streamlining the supply chain for complex heterocyclic structures.
Furthermore, the versatility of this enzymatic approach allows for the accommodation of various substrate types, including aromatic, heteroaromatic, and aliphatic aldehydes, making it a highly adaptable platform for diverse synthetic needs. The ability to generate dihydrothiophene scaffolds with high enantiomeric purity is crucial for the development of bioactive molecules, where stereochemistry often dictates pharmacological efficacy. This patent not only validates the catalytic proficiency of porcine pepsin beyond its natural digestive function but also establishes a reproducible framework for industrial application. As the industry moves towards greener chemistry standards, adopting such biocatalytic routes becomes a strategic imperative for maintaining competitiveness and regulatory compliance. The subsequent sections will delve deeper into the mechanistic nuances and commercial viability of this transformative synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral dihydrothiophenes has relied heavily on organocatalysts or transition-metal complexes, which often present significant challenges in terms of cost, toxicity, and waste management. Conventional methods frequently require stringent anhydrous conditions, expensive chiral ligands, and rigorous exclusion of oxygen and moisture, leading to complex operational protocols that hinder scalability. Moreover, the use of heavy metals raises serious concerns regarding residual contamination in the final product, necessitating additional purification steps that increase both production time and overall expenses. These traditional pathways often suffer from limited substrate scope, where electron-withdrawing or sterically hindered groups can drastically reduce yield and stereoselectivity. The environmental burden of disposing of metal-containing waste streams further complicates the sustainability profile of these legacy processes, making them less attractive in the current regulatory climate. Consequently, there is a pressing demand for alternative methodologies that can deliver high purity and selectivity without the associated logistical and environmental drawbacks.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN105755066B utilizes porcine pepsin, a readily available and inexpensive enzyme, to catalyze the asymmetric thia-Michael/aldol tandem reaction with remarkable efficiency. This biocatalytic method operates under mild conditions, typically at 30°C in a mixture of acetonitrile and phosphate buffer, eliminating the need for extreme temperatures or pressures. The use of water as a co-solvent not only enhances the green chemistry credentials of the process but also simplifies the reaction setup and workup procedures significantly. By employing 2,5-dihydroxy-1,4-dithiane as a stable precursor for mercaptoacetaldehyde, the method ensures a controlled release of the reactive thiol species, minimizing side reactions and improving overall yield. The enzymatic catalyst provides a chiral environment that induces high enantioselectivity, achieving up to 84% ee for certain substrates, which is competitive with many synthetic catalysts. This approach effectively addresses the limitations of conventional methods by offering a safer, more sustainable, and economically viable route for producing high-purity dihydrothiophene intermediates.
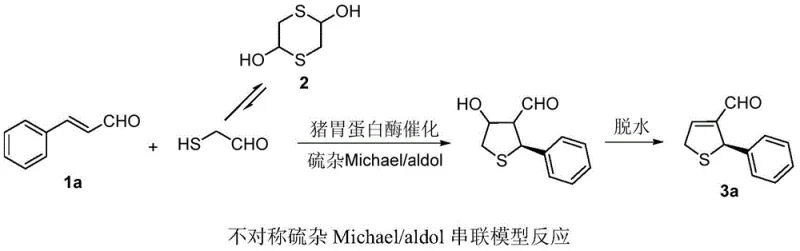
Mechanistic Insights into Porcine Pepsin-Catalyzed Tandem Reaction
The catalytic prowess of porcine pepsin in this transformation is rooted in its unique active site architecture, specifically involving two critical aspartic acid residues at positions 32 and 215. The mechanism initiates with the Asp32 residue acting as a general base to abstract a proton from the mercaptoacetaldehyde, which is generated in situ from the dithiane precursor. This deprotonation activates the thiol species, facilitating a nucleophilic attack on the beta-carbon of the alpha,beta-unsaturated aldehyde in a thia-Michael addition step. The resulting intermediate is then stabilized through protonation by the Asp215 residue, forming an enol structure that is primed for the subsequent intramolecular aldol cyclization. This cascade sequence proceeds with high stereochemical fidelity due to the rigid chiral pocket of the enzyme, which directs the spatial orientation of the reacting molecules. The final dehydration step yields the dihydrothiophene product with the observed configuration, demonstrating the enzyme's ability to control multiple bond-forming events in a single pot. Understanding this mechanistic pathway is essential for optimizing reaction parameters and expanding the substrate scope for future applications in complex molecule synthesis.
Impurity control in this enzymatic system is inherently superior due to the high specificity of the biocatalyst, which minimizes the formation of unwanted by-products commonly seen in non-enzymatic reactions. The mild reaction conditions prevent thermal degradation of sensitive functional groups, ensuring that the final product retains its structural integrity and purity. Furthermore, the absence of metal ions eliminates the risk of metal-catalyzed oxidation or polymerization side reactions, leading to a cleaner crude reaction mixture. This high level of chemoselectivity reduces the burden on downstream purification processes, allowing for simpler isolation techniques such as filtration and column chromatography. For quality assurance teams, this translates to a more consistent impurity profile and easier validation of the manufacturing process. The robustness of the enzyme under the specified pH and temperature conditions further contributes to batch-to-batch reproducibility, a critical factor for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Dihydrothiophene Compounds Efficiently
To implement this synthesis effectively, one must adhere to the optimized conditions established in the patent to maximize both yield and enantioselectivity. The process begins with the precise preparation of the solvent system, mixing acetonitrile and phosphate buffer at pH 6.5 in a 4:1 volume ratio to create the ideal medium for enzyme activity. Substrates are introduced at a specific molar ratio, typically favoring an excess of the dithiane precursor to drive the equilibrium towards product formation while maintaining low concentrations of the aldehyde to prevent self-polymerization. The reaction is maintained at a constant temperature of 30°C with continuous stirring to ensure homogeneous mixing and efficient mass transfer between the organic and aqueous phases. Detailed standardized synthesis steps follow below to guide the practical execution of this protocol in a laboratory or pilot plant setting.
- Prepare the reaction mixture by combining alpha,beta-unsaturated aldehyde and 2,5-dihydroxy-1,4-dithiane in a molar ratio of 1: 3.
- Add a solvent system consisting of acetonitrile and phosphate buffer (pH 6.5) in a volume ratio of 4: 1 to maintain substrate concentration.
- Introduce porcine pepsin catalyst and stir the reaction at 30°C for 96 to 168 hours to achieve optimal enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology offers substantial strategic benefits that extend beyond mere technical performance. The shift from precious metal catalysts to a ubiquitous biological enzyme like porcine pepsin drastically reduces raw material costs and mitigates the supply risks associated with fluctuating metal prices. Since the enzyme is derived from a renewable biological source, it aligns perfectly with corporate sustainability goals and reduces the regulatory burden related to hazardous substance handling. The simplified workup procedure, which avoids complex metal scavenging steps, leads to significant time savings in production cycles and lowers the consumption of auxiliary chemicals. This streamlined process enhances overall equipment effectiveness by reducing downtime between batches and minimizing the need for specialized containment infrastructure. Consequently, the total cost of ownership for manufacturing these intermediates is significantly lowered, providing a competitive edge in the global market.
- Cost Reduction in Manufacturing: The elimination of expensive chiral ligands and transition metals removes a major cost driver from the bill of materials, directly impacting the bottom line. Additionally, the reduced need for extensive purification to remove metal residues lowers solvent usage and waste disposal fees, contributing to further economic efficiency. The mild operating conditions also result in lower energy consumption compared to high-temperature or high-pressure alternatives, adding another layer of cost savings. By simplifying the process flow, labor costs associated with complex monitoring and handling are also minimized, making the overall operation more lean and efficient.
- Enhanced Supply Chain Reliability: Relying on commercially available enzymes and common solvents ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions. Unlike rare earth metals or specialized organocatalysts that may face supply bottlenecks, porcine pepsin is produced at a large scale for various industries, guaranteeing consistent availability. This reliability allows for better production planning and inventory management, reducing the risk of stockouts that could delay downstream drug development projects. Furthermore, the robustness of the enzyme allows for flexible sourcing strategies, as multiple suppliers can typically meet the quality specifications required for this application.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium facilitates easier scale-up from laboratory to commercial production without the safety hazards associated with volatile organic solvents alone. The biodegradability of the catalyst and the absence of toxic heavy metals simplify wastewater treatment and ensure compliance with increasingly strict environmental regulations. This green profile enhances the company's reputation and facilitates faster regulatory approvals for new drug applications that utilize these intermediates. The process is inherently safer for operators, reducing workplace health risks and associated liability costs, which is a critical consideration for modern manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial implementation of this enzymatic synthesis route. These answers are derived directly from the experimental data and findings presented in the patent documentation to provide accurate guidance. Understanding these aspects is crucial for stakeholders evaluating the integration of this technology into their existing manufacturing portfolios. The responses cover key areas such as catalyst stability, substrate compatibility, and process optimization parameters.
Q: What are the advantages of using porcine pepsin over traditional metal catalysts?
A: Porcine pepsin offers a green, biodegradable alternative that eliminates the need for toxic heavy metals, simplifying downstream purification and reducing environmental impact.
Q: What is the typical enantioselectivity achieved in this reaction?
A: Under optimized conditions, the reaction can achieve enantiomeric excess (ee) values up to 84%, particularly with branched aliphatic substrates.
Q: Is this method scalable for industrial production?
A: Yes, the use of commercially available enzymes and mild reaction conditions (30°C, aqueous/organic solvent mix) supports potential scale-up for fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydrothiophene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalytic processes like the one described in patent CN105755066B for the production of high-value fine chemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods can be successfully translated into robust industrial operations. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and enantiomeric excess. Our expertise in enzymatic reactions allows us to optimize conditions for maximum yield and selectivity, providing our partners with a reliable source of complex pharmaceutical intermediates. By choosing us, you gain access to a partner who understands the nuances of green chemistry and is dedicated to sustainable manufacturing practices.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements with tailored solutions. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to this enzymatic route for your supply chain. Our experts are ready to provide specific COA data and route feasibility assessments to help you make informed decisions. Let us collaborate to bring your next generation of therapeutic agents to market faster and more efficiently through our advanced synthesis capabilities.
