Scalable Metal-Free Synthesis of Methylene Bridged Quinoline-Triazole Hybrids for Advanced Fungicide Development
The agricultural chemical industry is constantly seeking novel scaffolds to combat resistant fungal pathogens, and patent CN114957215B presents a significant breakthrough in this domain. This intellectual property discloses a class of methylene bridged quinoline and 1,2,3-triazole biheterocyclic compounds that exhibit potent bactericidal activity against crop diseases such as cucumber anthracnose and gray mold. Unlike traditional approaches that often rely on complex multi-step sequences or toxic transition metal catalysts, this invention introduces a streamlined, metal-free synthetic pathway. The core innovation lies in the strategic fusion of the quinoline ring, known for its broad-spectrum biological activity, with the 1,2,3-triazole pharmacophore via a robust methylene bridge. This structural hybridization not only enhances biological efficacy but also simplifies the manufacturing process, making it highly attractive for commercial scale-up. For R&D directors and procurement specialists, this patent offers a viable route to high-purity agrochemical intermediates that bypass the regulatory and technical hurdles associated with heavy metal residues.
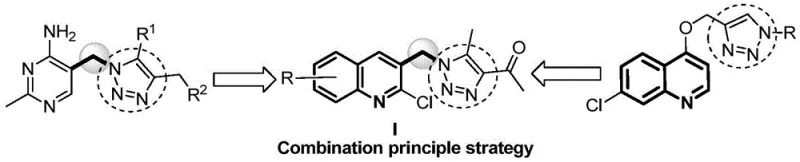
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,3-triazole rings has been dominated by the Copper(I)-catalyzed Azide-Alkyne Cycloaddition (CuAAC), commonly known as the Click reaction. While chemically efficient, this conventional methodology presents substantial drawbacks for large-scale agrochemical manufacturing. The primary concern is the reliance on transition metal catalysts, specifically copper, which poses significant challenges in downstream processing. Residual copper ions are difficult to remove completely and can introduce cytotoxicity, a critical failure point for bioactive compounds intended for environmental application. Furthermore, the CuAAC reaction typically requires the handling of potentially hazardous alkyne and azide precursors in separate steps, often necessitating the isolation of unstable intermediates. This increases operational complexity, solvent consumption, and waste generation, thereby inflating the cost of production and complicating supply chain logistics for high-volume manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in patent CN114957215B employs a ingenious "two-step one-pot" strategy that completely eliminates the need for transition metal catalysts. The process initiates with a nucleophilic substitution between 2-chloro-3-chloromethylquinoline and sodium azide, followed immediately by a cyclization reaction with acetylacetone in the same vessel. This telescoping of reactions avoids the isolation of the potentially explosive azide intermediate, significantly enhancing operational safety. By utilizing inexpensive inorganic bases like potassium carbonate or sodium carbonate instead of expensive ligand-metal complexes, the process drastically reduces raw material costs. The absence of metal catalysts means the final product is free from heavy metal contamination, simplifying quality control and ensuring compliance with stringent environmental regulations. This approach represents a paradigm shift towards greener, more cost-effective synthesis of complex heterocyclic fungicides.
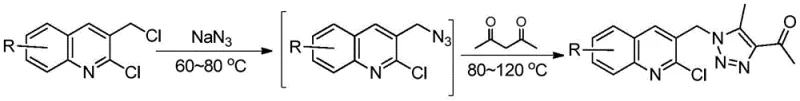
Mechanistic Insights into Metal-Free Cyclization
The reaction mechanism proceeds through a carefully orchestrated sequence of nucleophilic substitutions and condensations that maximize atom economy. Initially, the chloromethyl group on the quinoline ring undergoes nucleophilic attack by the azide ion (N3-) derived from sodium azide. This SN2-type substitution generates a 3-azidomethyl-2-chloroquinoline intermediate in situ. Crucially, this intermediate is not isolated; instead, upon the addition of acetylacetone and a base, the terminal nitrogen of the azide group attacks the carbonyl carbon of the acetylacetone. This triggers a cascade of intramolecular cyclization and dehydration steps, ultimately forming the stable 1,2,3-triazole ring fused to the quinoline scaffold via the methylene bridge. The use of polar aprotic solvents like DMF or acetonitrile facilitates the solubility of ionic species and stabilizes the transition states, ensuring high conversion rates even at moderate temperatures ranging from 60°C to 120°C.
From an impurity control perspective, this metal-free mechanism offers distinct advantages over catalytic methods. In traditional CuAAC reactions, side products often arise from oxidative homocoupling of alkynes or incomplete catalyst removal, leading to complex impurity profiles that require chromatographic purification. In this novel pathway, the primary byproducts are inorganic salts (such as sodium chloride) and water, which are easily removed during the aqueous workup. The specificity of the nucleophilic substitution ensures that the regioselectivity is inherently controlled by the structure of the starting quinoline derivative. This results in a crude product with high purity, often requiring only simple recrystallization or washing to meet commercial specifications. For process chemists, this translates to a robust and reproducible protocol that minimizes the risk of batch-to-batch variability.
How to Synthesize Methylene Bridged Quinoline-Triazole Efficiently
The synthesis protocol described in the patent is designed for operational simplicity and scalability, making it ideal for both laboratory optimization and industrial production. The process begins by dissolving the quinoline precursor and sodium azide in a lower alcohol solvent like ethanol and heating to reflux to drive the azide substitution to completion. Once the first step is verified by TLC, the solvent is removed under reduced pressure, and the residue is directly subjected to the cyclization conditions without purification. Acetylacetone and an inorganic base are added along with a high-boiling solvent like DMF, and the mixture is heated to promote ring closure.
- Dissolve 2-chloro-3-chloromethylquinoline and sodium azide in an organic solvent (e.g., ethanol) and heat to 60-80°C for nucleophilic substitution.
- Without isolating the intermediate, remove the solvent and add acetylacetone, an inorganic base (K2CO3 or Na2CO3), and a polar aprotic solvent (e.g., DMF).
- Heat the mixture to 80-120°C for 2-6 hours to effect cyclization, then precipitate the product with water and filter.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond mere chemical efficiency. The elimination of transition metal catalysts removes a significant cost driver, as noble metals and specialized ligands are subject to volatile market pricing and supply constraints. Furthermore, the "one-pot" nature of the synthesis reduces the number of unit operations required, which directly correlates to lower energy consumption and reduced labor costs per kilogram of product. The use of commodity chemicals such as sodium azide, acetylacetone, and simple quinoline derivatives ensures a stable and diversified supply base, mitigating the risk of raw material shortages that can plague specialty chemical manufacturing.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by removing the need for expensive copper catalysts and complex ligand systems, which are often single-use and difficult to recover. Additionally, the telescoping of two reaction steps into a single pot eliminates the isolation and purification of the intermediate azide, reducing solvent usage and waste disposal costs substantially. This streamlined workflow allows for higher throughput in existing reactor vessels, effectively increasing capacity without capital expenditure.
- Enhanced Supply Chain Reliability: The starting materials, particularly 2-chloro-3-chloromethylquinoline and acetylacetone, are widely available bulk chemicals with established global supply chains. This reduces dependency on niche suppliers and shortens lead times for raw material procurement. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, further ensures consistent production schedules even when facing minor variations in raw material quality or utility availability.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies the wastewater treatment process, as there is no need for specialized metal scavenging or complex effluent remediation protocols. This aligns with increasingly strict environmental regulations in major chemical manufacturing hubs. The solid product precipitates easily upon water addition, allowing for simple filtration and drying, which is highly scalable from pilot plant to multi-ton commercial production without the need for complex chromatography.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities.
Q: Why is the metal-free synthesis of quinoline-triazole hybrids advantageous for agrochemical applications?
A: Traditional Click chemistry often relies on copper catalysts, which can leave toxic metal residues unacceptable for agricultural use. This patent's metal-free approach eliminates heavy metal contamination risks, simplifies purification, and reduces environmental toxicity concerns.
Q: What are the key raw materials required for this synthesis?
A: The process utilizes readily available starting materials: 2-chloro-3-chloromethylquinoline derivatives, sodium azide, and acetylacetone. These are commodity chemicals with stable supply chains, ensuring consistent production capability.
Q: How does the 'one-pot' methodology impact production costs?
A: By avoiding the isolation of the unstable azide intermediate, the process reduces solvent consumption, labor time, and equipment usage. This telescoping of steps significantly lowers the overall cost of goods sold (COGS) compared to multi-step isolated processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methylene Bridged Quinoline-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of patent CN114957215B in the development of next-generation fungicides. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this metal-free synthesis, while our rigorous QC labs guarantee stringent purity specifications for every batch. We understand that consistency is key in agrochemical supply, and our dedicated process engineering team is ready to optimize this route for maximum yield and minimal environmental impact.
We invite you to collaborate with us to leverage this innovative chemistry for your product pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your time-to-market while reducing overall production costs. Let us be your strategic partner in bringing these high-efficacy quinoline-triazole hybrids to the global agricultural market.
