Advanced Crystalline Cephamycin Intermediate for Stable Antibiotic Synthesis
The pharmaceutical industry constantly seeks robust solutions to the challenges posed by unstable intermediates in antibiotic synthesis. Patent CN103193796B introduces a significant breakthrough in the preparation of cephamycin intermediates, specifically addressing the long-standing issues of hygroscopicity and chemical instability associated with traditional amorphous forms. This innovation details the preparation of a novel dicyclohexylamine salt of 7β-chloroacetamide-7α-methoxy-3-(1-methyl-1H-tetrazole-5-thiomethyl)-3-cephem-4-carboxylic acid. Unlike previous iterations that resulted in amorphous solids prone to degradation, this new compound exhibits exceptional crystalline properties, offering a reliable cephamycin intermediate supplier pathway for the production of critical antibiotics such as Cefminox sodium, Cefmetazole sodium, and Cefotetan disodium. The technical implications of shifting from an amorphous acid to a defined crystalline salt cannot be overstated, as it fundamentally alters the storage logistics and purity profiles achievable in large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cephamycin derivatives has been plagued by the physical instability of the key carboxylic acid intermediates. Prior art, such as the methods described in CN101302226A, often yielded the free acid or its triethylamine salt in an amorphous state. These amorphous forms possess a disordered molecular structure that makes them inherently thermodynamically unstable and highly hygroscopic, meaning they aggressively absorb moisture from the atmosphere. This moisture uptake accelerates hydrolytic degradation, leading to the formation of impurities that are difficult to remove in later stages. Furthermore, the triethylamine salt, while easier to handle than the free acid, still suffers from high water solubility and moisture sensitivity, making long-term storage risky and complicating inventory management for procurement teams. The lack of a defined crystal lattice in these conventional forms also hinders effective purification, often necessitating complex chromatographic steps that drive up costs and reduce overall yield in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology outlined in the patent presents a transformative solution by converting the unstable free acid into a dicyclohexylamine salt. This approach leverages the structural bulk and lipophilicity of the dicyclohexylamine cation to induce a highly ordered crystalline lattice. The process begins with the dissolution of the cephamycin free acid, structurally represented as Formula (II), in a specific solvent system.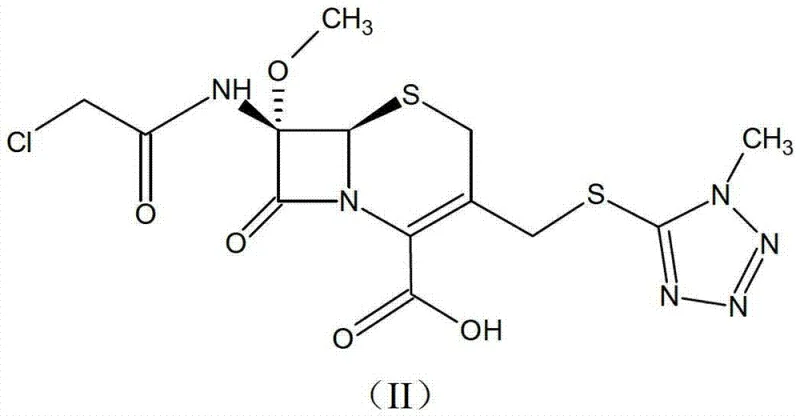
By reacting this precursor directly with dicyclohexylamine, the process bypasses the formation of unstable amorphous solids. The resulting product, shown as Formula (I), is a white crystalline powder that demonstrates remarkable resistance to moisture and degradation. This shift not only simplifies the isolation procedure—replacing complex purifications with straightforward filtration and drying—but also ensures that the intermediate maintains high purity levels exceeding 98% by HPLC, providing a solid foundation for the synthesis of high-quality final drug products.
Mechanistic Insights into Dicyclohexylamine Salt Formation and Crystallization
The core of this technological advancement lies in the acid-base chemistry and the subsequent crystallization kinetics driven by the choice of counter-ion. When the cephamycin carboxylic acid reacts with dicyclohexylamine, a proton transfer occurs, generating a carboxylate anion and a dicyclohexylammonium cation. Unlike smaller amines like triethylamine, the bulky cyclohexyl rings of dicyclohexylamine introduce significant steric hindrance and hydrophobic character to the salt complex. This structural feature disrupts the interaction with water molecules, drastically reducing the hygroscopic nature of the solid. The crystallization mechanism is further tuned by the selection of specific solvents, which modulate the solubility of the salt. By carefully controlling parameters such as temperature (ranging from -20°C to 50°C) and solvent polarity, the process encourages the nucleation and growth of well-defined crystals rather than the rapid precipitation of amorphous aggregates. This controlled growth is essential for trapping fewer impurities within the crystal lattice, effectively acting as a purification step in itself.
Furthermore, the stability of the final crystalline structure, depicted in Formula (I), is attributed to strong ionic interactions and potential hydrogen bonding networks within the lattice that lock the molecule into a low-energy state.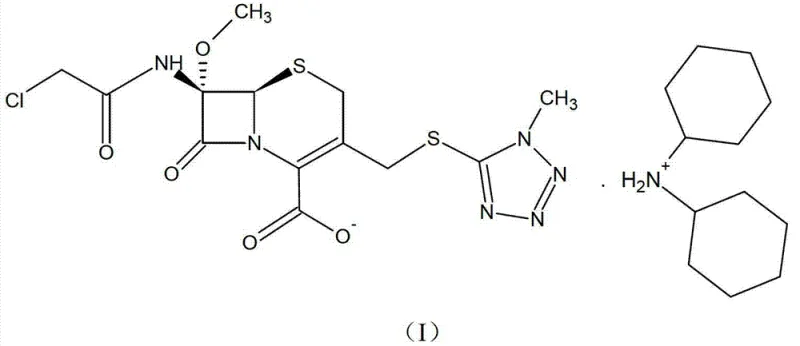
This thermodynamic stability is crucial for preventing the beta-lactam ring opening, a common degradation pathway for cephalosporins and cephamycins. The patent highlights that this salt form allows for vacuum drying at moderate temperatures (30-50°C) to achieve moisture content below 1%, a specification that is difficult to meet with amorphous forms without risking thermal decomposition. For R&D directors, understanding this mechanism provides confidence that the intermediate will remain chemically inert during storage and transport, ensuring that the reactivity required for the subsequent coupling reactions in antibiotic synthesis is preserved until the moment of use.
How to Synthesize Cephamycin Intermediate Dicyclohexylamine Salt Efficiently
The synthesis protocol described in the patent offers a versatile and scalable route suitable for industrial application. The process involves dissolving the cephamycin free acid in a variety of acceptable solvents, including acetone, ethyl acetate, DMF, or mixtures thereof, followed by the controlled addition of dicyclohexylamine. The reaction is exothermic and requires temperature management to ensure optimal crystal quality. Following the salt formation, the mixture is subjected to a crystallization period, often aided by cooling or seeding, to maximize yield. The solid is then isolated via filtration or centrifugation, washed to remove residual mother liquor, and dried under vacuum.
- Dissolve the cephamycin free acid (Formula II) in a specific organic solvent system such as acetone, ethyl acetate, or DMF mixtures at controlled temperatures between -20°C and 50°C.
- Add dicyclohexylamine directly to the solution to initiate the salt-forming reaction, maintaining a molar ratio of amine to acid between 0.5: 1 and 5:1 to ensure complete conversion.
- Induce crystallization by cooling the reaction mixture or adding anti-solvents, followed by filtration, washing with compatible solvents, and vacuum drying to obtain the high-purity crystalline salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this crystalline dicyclohexylamine salt represents a strategic opportunity to optimize costs and mitigate risk. The primary advantage lies in the drastic simplification of the purification workflow. By achieving high purity directly through crystallization, the need for expensive and time-consuming column chromatography is eliminated. This reduction in processing steps translates directly into lower operational expenditures and reduced solvent consumption, aligning with green chemistry initiatives. Additionally, the enhanced stability of the crystalline form significantly extends the shelf-life of the intermediate. This allows manufacturers to produce in larger batches and store inventory for longer periods without the fear of degradation, thereby smoothing out production schedules and reducing the pressure on just-in-time delivery models.
- Cost Reduction in Manufacturing: The elimination of complex purification steps such as column chromatography results in substantial cost savings. The process relies on standard unit operations like mixing, cooling, and filtration, which are far less capital-intensive and labor-intensive than chromatographic separations. Furthermore, the high recovery yields reported in the patent embodiments suggest that raw material utilization is maximized, reducing the cost per kilogram of the active intermediate. The ability to use a wide range of common industrial solvents also provides flexibility in sourcing, allowing procurement teams to leverage market fluctuations in solvent pricing to further drive down manufacturing costs.
- Enhanced Supply Chain Reliability: The hygroscopic nature of previous amorphous intermediates often led to supply chain disruptions due to product spoilage during transit or storage. The new crystalline salt is robust and stable, significantly reducing the risk of waste due to environmental exposure. This reliability ensures a consistent supply of high-quality material for downstream API production, minimizing the risk of batch failures in the final antibiotic formulation. For global supply chains, this stability is paramount, as it allows for safer and more predictable international shipping without the need for specialized climate-controlled logistics that amorphous materials might require.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory gram-scale experiments to multi-ton commercial production. The use of recyclable solvents like acetone and ethyl acetate, combined with the absence of heavy metal catalysts or hazardous reagents in the salt formation step, simplifies waste treatment and environmental compliance. The straightforward isolation of the product via filtration generates a solid waste stream that is easier to manage compared to the liquid waste streams associated with extensive chromatographic purification. This facilitates easier regulatory approval and supports sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cephamycin intermediate technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits for potential partners.
Q: Why is the dicyclohexylamine salt form preferred over the free acid or triethylamine salt for cephamycin intermediates?
A: The dicyclohexylamine salt offers superior physical stability compared to the amorphous free acid or the hygroscopic triethylamine salt. It exists as a stable crystalline solid that is less prone to moisture absorption and degradation during storage, ensuring higher purity for downstream antibiotic synthesis.
Q: What solvents are compatible with this crystallization process?
A: The process is highly versatile and supports a wide range of solvents including water, alcohols (methanol, ethanol, isopropanol), ketones (acetone, MEK, MIBK), esters (ethyl acetate, methyl carbonate), and polar aprotic solvents like DMF and acetonitrile, allowing for flexible process optimization.
Q: How does this intermediate improve the quality of final cephamycin antibiotics?
A: By providing a highly purified (>98% HPLC) and stable starting material, this intermediate minimizes the introduction of impurities during the subsequent synthesis steps for drugs like Cefminox sodium and Cefmetazole sodium, thereby enhancing the safety and efficacy profile of the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephamycin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team has extensively analyzed the pathway described in CN103193796B and is fully equipped to replicate and optimize this crystallization process for commercial needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial reality is seamless. Our facilities are governed by stringent purity specifications and supported by rigorous QC labs capable of verifying the crystalline nature and low moisture content of every batch, guaranteeing that the material you receive meets the highest standards for stability and reactivity.
We invite pharmaceutical manufacturers and contract research organizations to collaborate with us to leverage this advanced intermediate technology. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and solvent preferences. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain for cephamycin antibiotics is built on a foundation of stability, purity, and economic efficiency.
