Advanced Synthesis of Vilanterol Intermediate: A Strategic Breakthrough for API Manufacturing
Advanced Synthesis of Vilanterol Intermediate: A Strategic Breakthrough for API Manufacturing
The pharmaceutical landscape for respiratory treatments continues to evolve, driven by the demand for more efficient and cost-effective manufacturing processes for critical active pharmaceutical ingredients (APIs). A pivotal development in this sector is detailed in patent CN109665959B, which discloses a novel synthetic method for producing 3-hydroxymethyl-4-hydroxymandelic acid, a crucial intermediate for Vilanterol. Vilanterol is a potent long-acting beta2-adrenergic receptor agonist widely used in combination therapies for asthma and chronic obstructive pulmonary disease (COPD). The traditional synthesis pathways have long been plagued by excessive step counts and complex protection-deprotection sequences, creating bottlenecks in supply chains and inflating production costs. This new technology offers a transformative approach by utilizing a direct electrophilic substitution reaction, thereby streamlining the entire value chain from raw material to final drug substance.
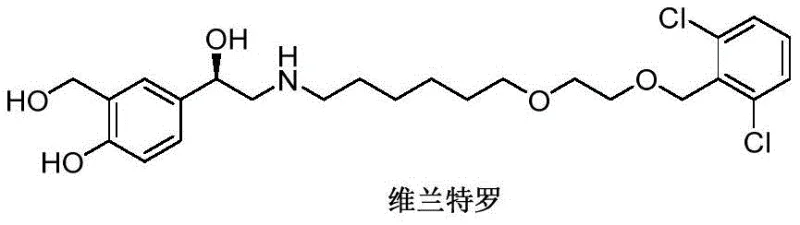
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the cumbersome construction of the chiral center and the aromatic substitution patterns that typically require harsh conditions. By starting with 4-hydroxymandelic acid and employing paraformaldehyde as an electrophilic source, the method achieves high regioselectivity and purity. This not only simplifies the purification protocols but also enhances the overall safety profile of the manufacturing process by reducing the inventory of hazardous intermediates. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic shifts is essential for evaluating the long-term viability and scalability of the supply base for respiratory medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, such as the route described in world patent WO2003024439, illustrates the historical challenges faced in Vilanterol synthesis. The conventional pathway typically involves more than ten distinct reaction steps to reach the final API. A major inefficiency in these legacy routes is the absolute necessity for introducing and subsequently removing protecting groups to mask reactive functional groups during intermediate transformations. Each protection and deprotection cycle adds at least two unit operations to the process, drastically reducing the overall atom economy and increasing the volume of solvent waste generated per kilogram of product. Furthermore, the construction of the specific chiral center required for biological activity in these older routes is notoriously difficult, often requiring six or more steps just to establish the correct stereochemistry with acceptable enantiomeric excess. These factors cumulatively result in low total yields and prohibitively high manufacturing costs, making the supply chain vulnerable to disruptions and price volatility.
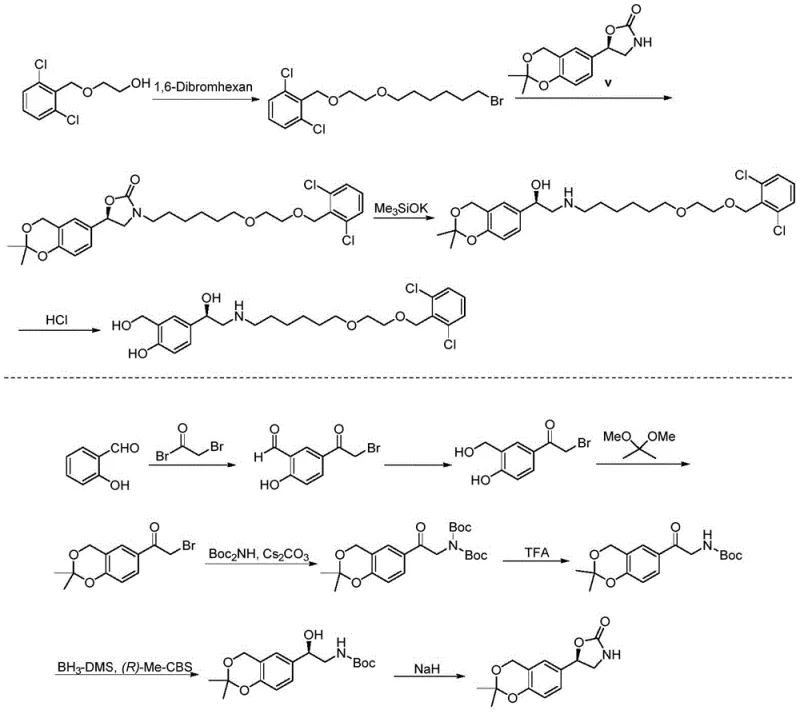
The Novel Approach
In stark contrast, the methodology presented in CN109665959B introduces a paradigm shift by focusing on the efficient synthesis of the key intermediate, 3-hydroxymethyl-4-hydroxymandelic acid. Once this intermediate is secured through the new one-step electrophilic substitution, the remaining path to Vilanterol is drastically shortened to merely three steps involving chiral resolution, condensation, and reduction. This reduction in step count is not merely a numerical improvement; it represents a fundamental enhancement in process robustness. By eliminating the need for protecting groups entirely in the formation of the core aromatic scaffold, the new route maximizes atom utilization and minimizes the generation of chemical waste. The simplicity of the operation allows for tighter control over impurity profiles, ensuring that the final API meets stringent regulatory standards with greater consistency. This approach effectively decouples the complexity of the synthesis from the cost of goods, offering a sustainable solution for cost reduction in API manufacturing.
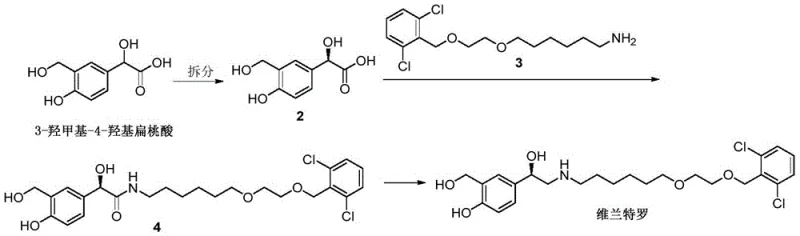
Mechanistic Insights into FeCl3-Catalyzed Electrophilic Substitution
The core innovation of this patent rests on a highly efficient electrophilic aromatic substitution reaction. The mechanism involves the activation of paraformaldehyde, which serves as a stable source of formaldehyde, by a Lewis acid catalyst such as Iron(III) Chloride (FeCl3) or Zinc Chloride (ZnCl2). In the presence of a polar protic solvent like trifluoroacetic acid or glacial acetic acid, the Lewis acid coordinates with the oxygen atoms of the formaldehyde oligomers, generating a highly reactive electrophilic species. This activated electrophile then attacks the electron-rich aromatic ring of 4-hydroxymandelic acid. Due to the directing effects of the hydroxyl and mandelic acid substituents, the substitution occurs selectively at the ortho-position relative to the phenolic hydroxyl group, yielding the desired 3-hydroxymethyl-4-hydroxymandelic acid. The reaction is typically conducted at moderate temperatures ranging from 60°C to 80°C, which provides sufficient energy to overcome the activation barrier without promoting side reactions or decomposition of the sensitive mandelic acid moiety.
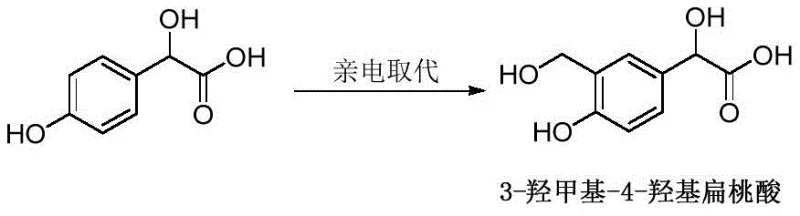
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or harsh acidic nitration/sulfonation alternatives. The mild Lewis acid conditions minimize the risk of racemization at the chiral center of the mandelic acid side chain, preserving the optical purity of the starting material. Furthermore, the use of paraformaldehyde ensures a controlled release of the electrophile, preventing poly-substitution which could lead to difficult-to-remove impurities. The workup procedure, involving simple concentration, water addition, and filtration, leverages the solubility differences between the product and unreacted starting materials or catalyst residues. Recrystallization from ethanol further purifies the solid, consistently achieving purity levels of 98% or higher as demonstrated in the patent examples. This high level of control is critical for downstream processing, as it reduces the burden on purification columns and crystallization steps in the subsequent synthesis of the final Vilanterol API.
How to Synthesize 3-Hydroxymethyl-4-hydroxymandelic Acid Efficiently
The practical implementation of this synthesis is designed for ease of operation in standard chemical reactors, making it accessible for both pilot-scale development and full commercial production. The process begins with the precise charging of 4-hydroxymandelic acid and paraformaldehyde into a reaction vessel, followed by the addition of the chosen solvent and catalyst system. The reaction mixture is then heated to the optimal temperature window and maintained for a fixed duration, typically around 4 hours, with progress monitored by thin-layer chromatography (TLC) to ensure complete conversion. Upon completion, the solvent is removed under reduced pressure, and the residue is treated with water to induce precipitation of the crude product. The final purification is achieved through recrystallization, yielding the high-purity intermediate ready for the next stage of Vilanterol synthesis. For a detailed breakdown of the specific operating parameters and stoichiometry, please refer to the standardized guide below.
- Combine 4-hydroxymandelic acid and paraformaldehyde in a solvent such as trifluoroacetic acid or glacial acetic acid with a catalyst like FeCl3 or ZnCl2.
- Heat the reaction mixture to 60-80°C and maintain for approximately 4 hours while monitoring progress via TLC.
- Cool the mixture, concentrate under vacuum, add water to precipitate the solid, and recrystallize from ethanol to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into tangible strategic benefits regarding cost stability and supply security. The primary driver of value is the drastic reduction in the number of unit operations required to produce the final API. By shortening the synthesis from over ten steps to a much more concise sequence, manufacturers can significantly reduce labor costs, utility consumption, and equipment occupancy time. This efficiency gain allows for a more competitive pricing structure without compromising on quality margins. Additionally, the elimination of protecting group chemistry removes the need for purchasing specialized and often expensive reagents dedicated solely to masking and unmasking functional groups. This simplification of the bill of materials (BOM) reduces the complexity of inventory management and mitigates the risk of supply shortages for niche reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the inherent efficiency of the chemistry. By avoiding the use of protecting groups, the process eliminates the associated reagent costs and the waste disposal costs linked to their removal. The high atom economy of the electrophilic substitution means that a larger proportion of the raw material mass ends up in the final product, reducing the cost per kilogram of the intermediate. Furthermore, the mild reaction conditions (60-80°C) lower energy consumption compared to processes requiring cryogenic temperatures or high-pressure hydrogenation. These factors combine to deliver substantial cost savings in pharmaceutical intermediate manufacturing, allowing for better margin management in a competitive generic drug market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and commodity-grade starting materials. 4-Hydroxymandelic acid and paraformaldehyde are produced on a large industrial scale globally, ensuring that there are no single-source bottlenecks for critical inputs. The robustness of the reaction, which tolerates minor variations in conditions without significant yield loss, ensures consistent batch-to-batch output. This reliability is crucial for maintaining continuous production schedules for life-saving respiratory medications. By reducing the lead time associated with complex multi-step syntheses, suppliers can respond more agilely to fluctuations in market demand, ensuring that high-purity pharmaceutical intermediates are available when needed most.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is significantly smaller than traditional routes, aligning with modern green chemistry principles and increasingly strict environmental regulations. The reduction in solvent usage and the avoidance of toxic protecting group reagents simplify wastewater treatment and hazardous waste disposal. The simple workup procedure involving filtration and recrystallization is inherently scalable, posing fewer engineering challenges when moving from kilogram to ton-scale production. This ease of commercial scale-up of complex pharmaceutical intermediates ensures that the technology can be rapidly deployed to meet global demand while maintaining compliance with environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the patent data and are intended to clarify the operational feasibility and quality implications for potential partners. Understanding these details is vital for making informed sourcing decisions and integrating this technology into existing production frameworks.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: The new route significantly shortens the synthesis pathway by eliminating the need for multiple protecting and deprotecting steps required in older methods. This leads to higher atom utilization, reduced waste generation, and improved overall yield, making it more suitable for large-scale industrial production.
Q: Which catalysts are most effective for this electrophilic substitution reaction?
A: The patent specifies that Lewis acids such as Zinc Chloride (ZnCl2) and Iron(III) Chloride (FeCl3) are highly effective catalysts. They facilitate the activation of paraformaldehyde for the electrophilic attack on the aromatic ring of 4-hydroxymandelic acid under mild conditions.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the process is designed for industrial scalability. It utilizes readily available raw materials, operates at moderate temperatures (60-80°C), and involves simple workup procedures like filtration and recrystallization, which are easily adaptable to large-scale reactor systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vilanterol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN109665959B for the global supply of asthma and COPD medications. As a leading CDMO and chemical manufacturer, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into robust, commercial-scale processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this streamlined synthesis are fully realized in terms of volume and consistency. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 3-hydroxymethyl-4-hydroxymandelic acid meets the highest international pharmacopoeia standards, providing our partners with absolute confidence in the quality of their supply chain.
We invite pharmaceutical companies and procurement leaders to collaborate with us to leverage this advanced technology for their Vilanterol projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your manufacturing strategy, reduce your cost of goods, and secure a reliable supply of high-quality intermediates for the future of respiratory care.
