Advanced Biocatalytic Route for High-Purity (S)-2-(3-pyridyl)pyrrolidine Production
Advanced Biocatalytic Route for High-Purity (S)-2-(3-pyridyl)pyrrolidine Production
The pharmaceutical industry's relentless pursuit of chiral intermediates has led to significant breakthroughs in biocatalysis, specifically highlighted in the recent patent CN112795603B. This intellectual property details a highly efficient method for preparing (S)-2-(3-pyridyl)pyrrolidine, a critical structural motif found in numerous neuroactive drugs targeting conditions such as Parkinson's and Alzheimer's disease. Unlike traditional chemical routes that struggle with stereocontrol, this invention leverages a specialized Imine Reductase (IRED) derived from Myxococcus fulvus to achieve exceptional enantioselectivity. For R&D directors and procurement specialists, this represents a paradigm shift towards greener, more cost-effective manufacturing of complex amine scaffolds. The technology not only simplifies the synthetic pathway but also aligns perfectly with modern regulatory demands for reduced heavy metal contamination and lower environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure 2-aryl(hetero)pyrrolidines has relied heavily on multi-step chemical transformations involving harsh reaction conditions and expensive chiral auxiliaries or transition metal catalysts. These conventional pathways often necessitate rigorous purification steps to remove toxic metal residues, which adds significant time and cost to the production lifecycle. Furthermore, achieving an optical purity exceeding 98.0% via chemical asymmetric synthesis is notoriously difficult, frequently resulting in racemic mixtures that require resolution, thereby capping the theoretical yield at 50%. The use of precious metal catalysts also introduces supply chain vulnerabilities and complicates waste disposal protocols, making these methods increasingly unattractive for large-scale commercial API intermediate manufacturing where margin compression is a constant pressure.
The Novel Approach
In stark contrast, the novel biocatalytic approach described in the patent utilizes a highly active imine reductase to directly reduce 2-pyridine-1-pyrroline compounds into the desired chiral amine with remarkable precision. This enzymatic route operates under mild physiological conditions, typically between 20°C and 40°C, eliminating the need for extreme temperatures or pressures that drive up energy consumption. The biological catalyst exhibits superior substrate tolerance, maintaining high conversion rates even at elevated substrate concentrations up to 90 g/L, which is a critical factor for industrial viability. By bypassing the need for chiral derivation reagents and heavy metal catalysts, this method offers a streamlined, single-step transformation that inherently produces high-purity products, fundamentally altering the economic model for producing these valuable nitrogen-containing heterocycles.
Mechanistic Insights into IRED-Catalyzed Asymmetric Reduction
The core of this technological advancement lies in the specific catalytic mechanism of the Imine Reductase (IRED) sourced from Myxococcus fulvus. This enzyme facilitates the stereoselective reduction of the C=N double bond in the 2-pyridine-1-pyrroline substrate, strictly controlling the spatial orientation of hydride transfer to ensure the formation of the (S)-enantiomer. The reaction relies on the cofactor NADPH as the hydride donor, which is oxidized to NADP+ during the reduction process. To make this process economically feasible for industrial applications, the patent integrates a coupled enzyme system where Glucose Dehydrogenase (GDH) regenerates the consumed NADPH back from NADP+ using inexpensive D-glucose as the terminal reductant. This creates a closed catalytic cycle where only a catalytic amount of the expensive cofactor is required, driving the reaction to completion while minimizing reagent costs.
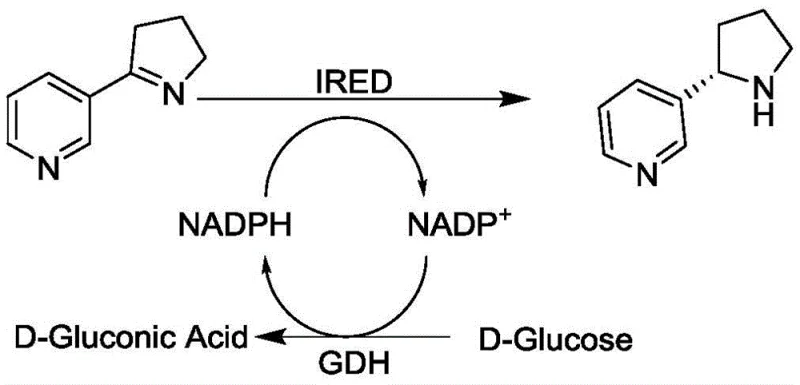
Furthermore, the impurity profile of the final product is significantly improved due to the high specificity of the enzyme. Chemical synthesis often generates regio-isomers or over-reduced byproducts that are structurally similar to the target molecule and difficult to separate. However, the enzyme's active site acts as a molecular sieve, accepting only the specific imine substrate and rejecting alternative reaction pathways. This intrinsic selectivity means that downstream purification is vastly simplified, often requiring only a standard extraction and crystallization rather than complex chromatography. For quality control teams, this translates to a more consistent batch-to-batch profile and a reduced risk of genotoxic impurities associated with chemical reagents, ensuring the final API intermediate meets stringent global pharmacopeia standards without extensive remediation.
How to Synthesize (S)-2-(3-pyridyl)pyrrolidine Efficiently
The implementation of this biocatalytic process involves a straightforward workflow that begins with the fermentation of genetically engineered E. coli strains expressing the target imine reductase. Following cell harvest and lysis to obtain the crude enzyme or whole cells, the biotransformation is conducted in a buffered aqueous system supplemented with glucose and the necessary cofactor. The reaction progress is monitored via TLC or HPLC, and upon completion, the product is isolated through pH adjustment and organic solvent extraction. This operational simplicity allows for easy integration into existing pilot plant facilities without the need for specialized high-pressure reactors.
- Cultivate genetically engineered E. coli expressing MsIR1 imine reductase in fermentation medium with IPTG induction.
- Prepare the biocatalytic reaction system using phosphate buffer (pH 7.5), glucose, NADP+, and GDH for cofactor recycling.
- Add 2-pyridine-1-pyrroline substrate (up to 90 g/L) and react at 28-30°C, followed by alkalization and organic extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic technology offers profound strategic benefits that extend beyond simple yield improvements. The elimination of transition metal catalysts removes a major cost center associated with both the purchase of precious metals and the subsequent validation of their removal from the final product. This shift not only reduces the direct cost of goods sold (COGS) but also mitigates the regulatory risks associated with heavy metal limits in pharmaceutical ingredients. Additionally, the reliance on renewable biological catalysts and benign reagents like glucose positions the supply chain for greater sustainability, aligning with the ESG (Environmental, Social, and Governance) goals that are increasingly mandatory for Tier 1 suppliers in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The implementation of the GDH/Glucose cofactor regeneration system fundamentally changes the economics of chiral amine synthesis by removing the need for stoichiometric quantities of expensive NADPH. In traditional biocatalysis, the cost of cofactors can be prohibitive, but this recycling loop ensures that minute amounts drive the entire reaction, leading to substantial raw material savings. Furthermore, the high substrate loading capacity of up to 90 g/L means that smaller reactor volumes can produce the same output, effectively increasing asset utilization and reducing capital expenditure requirements for new production lines.
- Enhanced Supply Chain Reliability: By utilizing a robust enzymatic process that operates under mild conditions, the manufacturing timeline is significantly compressed compared to multi-step chemical syntheses. The reduction in unit operations—from multiple reaction vessels and purification columns to a single biotransformation step—minimizes the potential for bottlenecks and equipment downtime. This streamlined workflow ensures a more predictable lead time for delivering high-purity pharmaceutical intermediates, allowing downstream API manufacturers to maintain leaner inventory levels and respond more agilely to market demand fluctuations without compromising on quality.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent's successful translation from milligram-scale screening to liter-scale fermentation and conversion. The aqueous nature of the reaction medium and the absence of volatile organic solvents during the reaction phase drastically reduce the facility's VOC (Volatile Organic Compound) emissions. This compliance with green chemistry principles simplifies the permitting process for new manufacturing sites and reduces the long-term liability associated with hazardous waste disposal, making it a future-proof solution for expanding production capacity in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this IRED-based synthesis platform. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential licensees or manufacturing partners. Understanding these nuances is essential for evaluating the feasibility of integrating this route into your current portfolio.
Q: What is the optical purity achieved in this biocatalytic process?
A: The process utilizing Imine Reductase from Myxococcus fulvus consistently achieves an enantiomeric excess (ee) of greater than 98%, often reaching 99.7% to 99.8%, which significantly exceeds typical chemical synthesis standards.
Q: How does the cofactor regeneration system impact production costs?
A: By employing a Glucose Dehydrogenase (GDH)/Glucose system, the expensive NADP+ cofactor is continuously regenerated in situ. This eliminates the need for stoichiometric amounts of costly cofactors, drastically reducing raw material expenses for large-scale manufacturing.
Q: Can this method handle high substrate concentrations for industrial scale-up?
A: Yes, the patented method demonstrates robust activity at substrate concentrations ranging from 10 g/L up to 90 g/L. This high tolerance allows for reduced reactor volumes and higher throughput compared to traditional enzymatic processes that often suffer from substrate inhibition.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-(3-pyridyl)pyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic route described in CN112795603B for the production of high-value chiral amines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the >99% ee values achievable with this specific imine reductase technology. We are committed to delivering pharmaceutical intermediates that meet the highest global standards for safety and efficacy.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced synthesis method for your pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive landscape of chiral drug development.
