Revolutionizing Chiral Synthesis: Scalable Biocatalytic Production of (R)-1,3-Butanediol
Revolutionizing Chiral Synthesis: Scalable Biocatalytic Production of (R)-1,3-Butanediol
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards sustainable manufacturing, driven by the urgent need for greener synthetic routes and higher stereochemical fidelity. A pivotal development in this landscape is documented in patent CN109749968B, which discloses a novel Bacillus velezensis strain, specifically designated as SWGC31011 (CGMCC No. 13354), capable of高效 biocatalytic synthesis of (R)-1,3-butanediol. This chiral diol serves as a critical building block for a vast array of high-value compounds, including carbapenem antibiotics, fragrances, and agrochemicals. The strategic importance of this molecule cannot be overstated, as it acts as a key precursor in the synthesis of life-saving beta-lactam antibiotics, addressing the global challenge of antibiotic resistance. By leveraging the intrinsic enzymatic machinery of this prokaryotic strain, manufacturers can bypass the severe limitations of traditional chemical synthesis, achieving conversion rates exceeding 95% with exceptional optical purity.
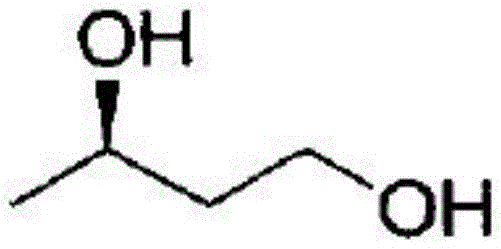
For R&D directors and procurement strategists, the implications of this technology extend far beyond simple yield improvements. The ability to produce high-purity (R)-1,3-butanediol through a biological fermentation process represents a fundamental upgrade in process chemistry. Traditional methods often struggle with the separation of enantiomers or require stoichiometric amounts of chiral auxiliaries, leading to excessive waste and inflated costs. In contrast, the enzymatic approach described in the patent utilizes a carbonyl reductase expressed by the bacteria to perform a highly stereoselective asymmetric reduction of 4-hydroxy-2-butanone. This specificity ensures that the final product meets the rigorous quality standards required for active pharmaceutical ingredient (API) intermediates, thereby reducing the burden on downstream purification units and enhancing overall process efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of (R)-1,3-butanediol has relied heavily on chemical synthesis routes that are fraught with inefficiencies and environmental hazards. One prevalent method utilizes L-threonine as a starting material, subjecting it to a multi-step sequence involving nitrosation deamination, methyl esterification, hydrogenation, and decalcification. This convoluted pathway not only suffers from a dismal overall yield of approximately 64% but also generates significant quantities of toxic byproducts and hazardous waste streams. Furthermore, the reliance on harsh reaction conditions and expensive chiral starting materials drives up the cost of goods sold (COGS), making the final product economically vulnerable to raw material price fluctuations. Another chemical alternative involves the asymmetric hydrogenation of 4-hydroxy-2-butanone using ruthenium-based catalysts. While this method can achieve high yields, the prohibitive cost of noble metal catalysts and the stringent requirement for removing trace heavy metal residues from the final product render it less attractive for large-scale pharmaceutical applications where regulatory compliance is paramount.
The Novel Approach
The biocatalytic strategy introduced in patent CN109749968B offers a transformative solution to these entrenched problems by employing a whole-cell biocatalyst system. The core innovation lies in the use of the Bacillus velezensis SWGC31011 strain, which naturally expresses a highly active carbonyl reductase. This enzyme facilitates the direct asymmetric reduction of the prochiral ketone substrate, 4-hydroxy-2-butanone, into the desired chiral alcohol with remarkable precision. Unlike the multi-step chemical synthesis, this biological process consolidates the transformation into a single, efficient reaction step conducted under mild aqueous conditions. The elimination of toxic reagents and the avoidance of heavy metal catalysts not only simplifies the workflow but also aligns perfectly with modern green chemistry principles. For a reliable pharmaceutical intermediate supplier, adopting this route means transitioning from a pollution-intensive operation to a clean, fermentation-based manufacturing model that is inherently safer and more sustainable.
Mechanistic Insights into Carbonyl Reductase-Catalyzed Asymmetric Reduction
At the heart of this technological breakthrough is the sophisticated enzymatic mechanism employed by the Bacillus velezensis strain. The carbonyl reductase produced by SWGC31011 exhibits exquisite substrate specificity and stereocontrol, recognizing the prochiral center of 4-hydroxy-2-butanone and delivering a hydride ion exclusively to one face of the carbonyl group. This results in the formation of the (R)-enantiomer with an optical purity that can reach 100% ee, a level of fidelity that is exceptionally difficult to achieve with small-molecule organocatalysts without extensive optimization. The reaction proceeds via a classic NADH-dependent reduction mechanism, where the enzyme utilizes the reduced form of nicotinamide adenine dinucleotide as a cofactor to drive the thermodynamic equilibrium towards the alcohol product. To sustain this catalytic cycle in an industrial setting, the process incorporates an auxiliary substrate system—such as glucose, isopropanol, or xylose—which serves to regenerate the consumed NADH back to its active reduced state. This cofactor recycling loop is critical for maintaining high catalytic turnover numbers and ensuring that the reaction remains economically viable by minimizing the need for exogenous cofactor addition.
From an impurity control perspective, the biological nature of this synthesis provides inherent advantages over chemical counterparts. In traditional chemical reductions, side reactions such as over-reduction, dehydration, or non-selective reduction of other functional groups can lead to complex impurity profiles that are challenging to separate. However, the enzymatic active site acts as a molecular sieve, sterically hindering access to unwanted reaction pathways and ensuring that only the target transformation occurs. The patent data indicates that the conversion rate can exceed 95%, leaving minimal unreacted starting material and virtually no detectable (S)-enantiomer impurity. This high selectivity drastically reduces the complexity of the workup procedure, often allowing for straightforward extraction and distillation to obtain the final product. For quality assurance teams, this translates to a more robust and predictable manufacturing process with fewer batch failures and a consistently high-quality output that meets stringent pharmacopoeial specifications.
How to Synthesize (R)-1,3-Butanediol Efficiently
Implementing this biocatalytic route requires a precise understanding of the fermentation and bioconversion parameters to maximize productivity. The process begins with the cultivation of the Bacillus velezensis SWGC31011 strain in a nutrient-rich medium optimized for enzyme expression. Following cell harvest, the wet biomass is suspended in a buffered solution containing the substrate and a co-substrate for cofactor regeneration. The reaction is then allowed to proceed under controlled temperature and pH conditions to ensure optimal enzyme activity and stability. The detailed standard operating procedures for scaling this reaction from laboratory benchtop to pilot plant volumes are critical for ensuring reproducibility and safety during technology transfer.
- Cultivate Bacillus velezensis SWGC31011 in optimized fermentation media containing glucose and yeast extract at 28-30°C.
- Harvest wet cells via centrifugation and suspend in a buffered solution with 4-hydroxy-2-butanone substrate.
- Conduct asymmetric reduction at mild temperatures (20-50°C) with auxiliary substrates like glucose to regenerate cofactors.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this biocatalytic technology presents a compelling value proposition centered on cost efficiency and risk mitigation. The shift from chemical synthesis to biological fermentation fundamentally alters the cost structure of producing (R)-1,3-butanediol. By eliminating the need for expensive noble metal catalysts like ruthenium and avoiding the use of hazardous chemical reagents, the direct material costs are significantly reduced. Furthermore, the simplified downstream processing—resulting from the high selectivity of the enzyme—lowers the consumption of solvents and energy required for purification. These factors combine to deliver substantial cost savings in API manufacturing, allowing companies to improve their margins or offer more competitive pricing to their customers without compromising on quality. The economic benefits are further amplified by the use of readily available, low-cost carbon sources such as glucose and yeast extract in the fermentation media, which are far cheaper and more stable in supply than specialized chiral chemical starting materials.
- Cost Reduction in Manufacturing: The transition to a whole-cell biocatalytic system removes the dependency on precious metal catalysts, which are subject to volatile market prices and supply constraints. Additionally, the mild reaction conditions (20-50°C and near-neutral pH) reduce the energy load associated with heating, cooling, and pressure management, leading to lower utility costs. The high conversion efficiency minimizes raw material waste, ensuring that a greater proportion of the input substrate is converted into saleable product, thereby optimizing the overall material balance and reducing the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Relying on a robust bacterial strain for production enhances supply chain resilience compared to complex multi-step chemical syntheses that may depend on scarce reagents. The fermentation process is highly scalable and can be easily adapted to existing bioreactor infrastructure, reducing the lead time for capacity expansion. Moreover, the stability of the lyophilized or frozen bacterial cells ensures a consistent supply of the biocatalyst, mitigating the risk of production delays caused by catalyst degradation or availability issues often seen with sensitive chemical catalysts.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic reaction significantly reduces the generation of organic solvent waste, simplifying wastewater treatment and lowering environmental compliance costs. This green profile is increasingly important for meeting corporate sustainability goals and regulatory requirements in major markets. The process is inherently safer, operating at atmospheric pressure and moderate temperatures, which reduces the risk of industrial accidents and lowers insurance premiums. This combination of scalability and environmental stewardship makes the technology ideal for the commercial scale-up of complex fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic process. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing production lines. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. These insights clarify the operational parameters and expected outcomes, facilitating a smoother transition from R&D evaluation to commercial adoption.
Q: What is the optical purity achievable with this biocatalytic method?
A: The patented Bacillus velezensis strain SWGC31011 achieves an optical purity of up to 100% ee, significantly outperforming traditional chemical resolution methods.
Q: How does this biological route compare to ruthenium-catalyzed synthesis?
A: Unlike ruthenium-based chemical catalysis which involves expensive heavy metals and complex removal steps, this biocatalytic process operates under mild aqueous conditions, eliminating heavy metal contamination risks.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of a robust prokaryotic strain (Bacillus velezensis) allows for high-density fermentation and simplified downstream processing, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-1,3-Butanediol Supplier
As the demand for high-purity chiral intermediates continues to surge, partnering with an experienced CDMO is crucial for navigating the complexities of process development and scale-up. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to maintain stringent purity specifications for sensitive pharmaceutical intermediates. We understand the critical nature of supply continuity in the global pharmaceutical market and are committed to delivering products that meet the highest international standards.
We invite you to collaborate with our technical team to explore how this innovative biocatalytic route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of scientific excellence and commercial viability.
