Advanced Synthesis of N-Nitrone Aromatic Amide Derivatives for Pharmaceutical Applications
Advanced Synthesis of N-Nitrone Aromatic Amide Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with operational efficiency. Patent CN109438288B introduces a significant breakthrough in the preparation of N-nitrone substituted aromatic amide derivatives, compounds that serve as critical building blocks in modern drug discovery. These molecules feature a unique structural motif where a tert-butyl nitrone group and an amide group are positioned adjacently on a benzene ring, conferring dual functionality that is highly valued in medicinal chemistry. The patent details a novel catalytic system that enables the direct construction of this scaffold under remarkably mild conditions, addressing long-standing challenges in substrate compatibility and reaction control. For R&D directors and procurement specialists, understanding the nuances of this technology is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering complex scaffolds with consistent quality.
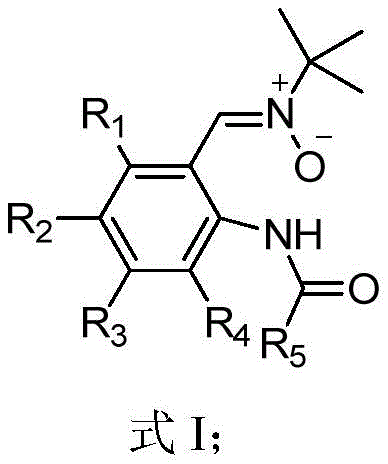
The core innovation lies in the versatility of the substitution pattern, allowing for a wide range of R groups including halogens, alkyls, and trifluoromethyl groups. This structural diversity is crucial for optimizing the pharmacokinetic properties of final drug candidates. By leveraging this patented methodology, manufacturers can access a broader chemical space for developing next-generation therapeutics, ranging from antimalarial agents to antioxidants. The ability to introduce these functional groups efficiently without compromising the integrity of the sensitive nitrone moiety represents a substantial advancement in synthetic organic chemistry, paving the way for more cost-effective production of high-value active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for aromatic amide derivatives often suffer from significant drawbacks that hinder large-scale industrial adoption. Conventional methods typically require harsh reaction conditions, including elevated temperatures and the use of strong acids or bases, which can lead to the decomposition of sensitive functional groups like nitrones. Furthermore, existing processes frequently involve multiple synthetic steps, necessitating the isolation and purification of unstable intermediates, which drastically reduces overall yield and increases waste generation. The reliance on stoichiometric oxidants in older C-H activation protocols not only escalates raw material costs but also creates substantial environmental burdens due to the generation of heavy metal waste and toxic byproducts. These inefficiencies result in prolonged lead times and higher production costs, making it difficult for supply chain managers to maintain consistent inventory levels for critical intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN109438288B utilizes a transition metal-catalyzed C-H amination strategy that operates efficiently at room temperature under an air atmosphere. This approach eliminates the need for external oxidants, acids, or alkalis, thereby streamlining the reaction workflow and minimizing the risk of side reactions. The use of 1,4,2-dioxazol-5-one derivatives as amidating reagents allows for the direct installation of the amide group with high regioselectivity, driven by the coordinating ability of the nitrone oxygen. This green chemistry protocol not only enhances reaction specificity but also simplifies the downstream processing, as the reaction mixture can often be purified via simple filtration and chromatography. For procurement teams, this translates to a more predictable manufacturing timeline and reduced dependency on hazardous reagents, aligning perfectly with modern sustainability goals.
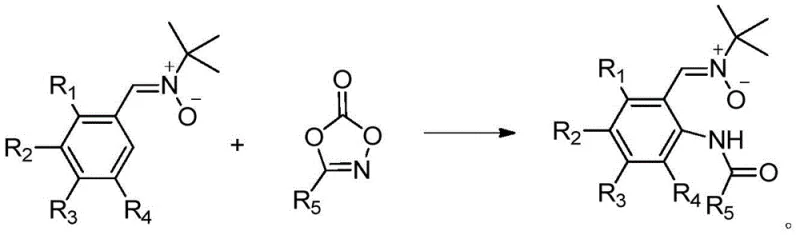
Mechanistic Insights into Transition Metal-Catalyzed C-H Amination
The success of this synthesis relies heavily on the precise interplay between the transition metal catalyst and the substrate's directing group. The mechanism likely involves the initial coordination of the nitrone oxygen to the metal center, such as Iridium(III) or Rhodium(III), forming a stable metallacycle intermediate. This coordination activates the ortho-C-H bond of the aromatic ring, facilitating its cleavage and subsequent insertion of the nitrene species generated from the dioxazolone. The mild conditions prevent the premature decomposition of the nitrone group, which is notoriously unstable under acidic or basic environments. Understanding this mechanistic pathway is vital for R&D directors aiming to optimize reaction parameters for specific substrates, ensuring that the delicate balance between catalyst activity and substrate stability is maintained throughout the process.
Impurity control is another critical aspect addressed by this catalytic system. The high selectivity of the catalyst minimizes the formation of regioisomers and over-oxidized byproducts, which are common issues in non-directed C-H functionalization. The patent specifies the use of silver salts as additives, which likely play a role in abstracting halide ligands from the metal precursor to generate the active cationic catalytic species. This fine-tuning of the catalytic cycle ensures that the reaction proceeds with high fidelity, yielding products with purity profiles suitable for pharmaceutical applications without the need for extensive recrystallization. Such control over the impurity spectrum is essential for meeting the stringent regulatory requirements of global health authorities.
How to Synthesize N-Nitrone Aromatic Amide Derivatives Efficiently
The practical implementation of this synthesis is designed for scalability and ease of operation, making it accessible for both laboratory research and commercial production. The protocol involves mixing the aromatic nitrone and the dioxazolone derivative in a suitable organic solvent such as 1,2-dichloroethane, followed by the addition of the catalyst and additive. The reaction proceeds rapidly at ambient temperature, typically reaching completion within 0.5 to 2 hours, which significantly reduces energy consumption compared to thermal methods. Detailed standardized synthesis steps are provided below to guide process chemists in replicating these results with high precision.
- Mix aromatic nitrone (Formula II) and 1,4,2-dioxazol-5-one derivative (Formula III) with a transition metal catalyst such as Cp*IrCl2 and a silver salt additive in a solvent like 1,2-dichloroethane.
- Stir the reaction mixture at room temperature under air atmosphere for 0.5 to 2 hours to facilitate the amination reaction without external heating.
- Filter the reaction solution through diatomite to remove catalyst residues, concentrate the filtrate, and purify the crude product via column chromatography to obtain the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers tangible benefits that extend beyond mere chemical novelty. The elimination of harsh reaction conditions and expensive oxidants directly contributes to a reduction in operational expenditures, as there is no need for specialized corrosion-resistant equipment or complex waste treatment systems. The simplicity of the workup procedure, involving basic filtration and concentration, allows for faster turnaround times between batches, enhancing the overall agility of the supply chain. This efficiency is particularly valuable when responding to fluctuating market demands for specialized pharmaceutical intermediates, ensuring that production schedules remain robust and uninterrupted.
- Cost Reduction in Manufacturing: The transition to a room-temperature catalytic process eliminates the energy costs associated with heating and cooling cycles, leading to substantial savings in utility expenses. Furthermore, the high atom economy of the reaction means that raw materials are utilized more effectively, reducing the cost per kilogram of the final product. By avoiding the use of stoichiometric oxidants, the process also lowers the cost of waste disposal and environmental compliance, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials, such as aromatic nitrones and dioxazolones, ensures a consistent supply of feedstock without the risk of shortages associated with exotic reagents. The robustness of the reaction under air atmosphere removes the need for inert gas protection, simplifying the operational requirements and reducing the risk of batch failures due to equipment malfunction. This reliability is crucial for maintaining continuous production flows and meeting strict delivery deadlines for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns with increasingly stringent environmental regulations, minimizing the generation of hazardous waste and reducing the carbon footprint of the manufacturing process. The scalability of the reaction has been demonstrated through various examples in the patent, showing that yields remain high even as the scale increases. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to expand production capacity without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of N-nitrone substituted aromatic amide derivatives. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their supply chains.
Q: What are the primary advantages of this synthesis method over conventional aromatic amide production?
A: The patented method utilizes a direct C-H amination strategy that operates under mild conditions (room temperature, air) without requiring harsh oxidants or acids. This significantly simplifies the post-treatment process and reduces energy consumption compared to traditional multi-step syntheses.
Q: Which catalysts are most effective for this N-nitrone substitution reaction?
A: The patent identifies several high-efficiency transition metal catalysts, including dichloro(pentamethylcyclopentadienyl)iridium(III), palladium acetate, and rhodium complexes. These catalysts enable high selectivity and yield while minimizing the formation of side products.
Q: What is the pharmaceutical potential of N-nitrone substituted aromatic amides?
A: These derivatives serve as versatile intermediates for producing vitamins, hormones, and antimalarial drugs. Additionally, the tert-butyl nitrone moiety acts as a free radical scavenger, offering potential applications in developing antioxidants and anti-platelet aggregation agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Nitrone Aromatic Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN109438288B for the production of high-purity pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the highest industry standards. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-nitrone aromatic amide delivered is free from critical impurities and ready for immediate use in drug development pipelines.
We invite global pharmaceutical companies and research institutions to collaborate with us to leverage this advanced technology for their specific projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient synthesis can optimize your budget. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
