Advanced Catalytic Synthesis of Alpha-Amino Acetamides for High-Efficiency Agrochemical Manufacturing
The chemical industry is constantly evolving towards greener and more efficient synthetic pathways, particularly for critical intermediates used in agrochemical production. A significant breakthrough in this domain is documented in patent CN102050754B, which details a novel method for preparing alpha-amino-dialkyl substituted acetamides. These compounds serve as vital precursors in the synthesis of imidazolinone herbicides, a class of agricultural chemicals known for their effectiveness in weed control. The patent introduces a catalytic oxidative hydrolysis process that utilizes hydrogen peroxide as the oxidant, replacing traditional, harsher reagents. This technological shift addresses long-standing challenges in the manufacturing of these intermediates, specifically focusing on improving reaction yields, enhancing product purity, and mitigating the environmental impact associated with legacy production methods. By leveraging specific ketone or amide catalysts under controlled alkaline conditions, this invention offers a robust solution for producing high-quality alpha-amino amides.
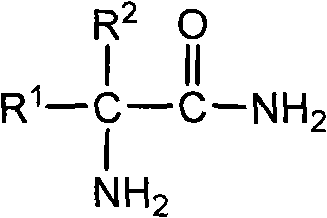
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-amino-dialkyl substituted acetamides has relied heavily on methods that pose significant operational and environmental risks. One prominent conventional technique involves the oxidation of alpha-amino-dialkyl substituted acetonitriles using concentrated sulfuric acid. While this method has been widely utilized, it suffers from severe drawbacks that impact both the economic and safety profiles of the manufacturing process. The use of concentrated sulfuric acid leads to substantial equipment corrosion, necessitating the use of expensive, corrosion-resistant materials for reactors and piping, which drives up capital expenditure. Furthermore, this acidic process generates a large volume of "three wastes" (waste water, waste gas, and waste residue), creating a heavy burden on waste treatment facilities and increasing the overall cost of environmental compliance. The handling of strong acids also introduces higher operational hazards for personnel, requiring stringent safety protocols that can slow down production throughput.
The Novel Approach
In stark contrast to the corrosive nature of sulfuric acid oxidation, the method disclosed in patent CN102050754B employs a much milder and environmentally friendly oxidative system. This novel approach utilizes hydrogen peroxide ($H_2O_2$) as the primary oxidizing agent in an alkaline medium, catalyzed by specific organic molecules such as ketones or amides. The transition to a hydrogen peroxide-based system fundamentally alters the reaction landscape, eliminating the need for strong mineral acids and thereby drastically reducing equipment corrosion. This allows manufacturers to utilize standard stainless steel equipment, lowering maintenance costs and extending the lifespan of production assets. Moreover, the byproducts of hydrogen peroxide oxidation are primarily water and oxygen, which significantly simplifies the waste stream and reduces the complexity of effluent treatment. The introduction of catalysts like acetone or phenylacetamide ensures that the reaction proceeds efficiently at moderate temperatures, typically between 5-30°C, further enhancing the safety and energy efficiency of the process.
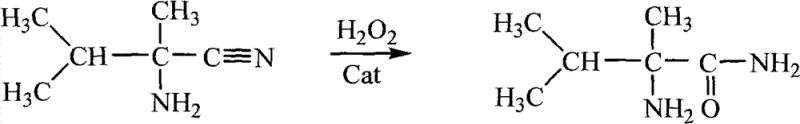
Mechanistic Insights into Catalytic Oxidative Hydrolysis
The success of this synthetic route lies in the intricate interplay between the catalyst, the oxidant, and the reaction medium. The patent elucidates that the addition of suitable amide or ketone catalysts is not merely incidental but is critical for the stabilization of hydrogen peroxide in the alkaline environment. In the absence of these catalysts, hydrogen peroxide tends to decompose rapidly, leading to poor conversion rates and low yields, as evidenced by comparative examples in the patent data. The proposed mechanism suggests that the lone pair of electrons on the nitrogen atom of the amide catalyst, or the carbanion generated from the methyl ketone under alkaline conditions, plays a pivotal role. These electron-rich species effectively interact with the hydrogen ions formed during the oxidative hydrolysis process, facilitating the nucleophilic attack on the nitrile group. This interaction promotes the continuous completion of the reaction, driving the conversion of the nitrile starting material to the desired amide product with high efficiency.
Furthermore, precise control of the reaction pH is a key mechanistic factor that distinguishes this process from non-catalytic variations. The patent specifies maintaining a pH range of 9-10, with an optimal window of 9.2-9.5. This specific alkalinity is crucial because it strikes a balance between activating the catalyst and preventing the excessive decomposition of hydrogen peroxide, which occurs more rapidly at higher pH levels. If the pH is too low, the catalytic activity of the ketone or amide is diminished; if too high, the oxidant is wasted through decomposition. By strictly adhering to this pH window, the process ensures that the electron cloud distribution of the intermediate species remains favorable for the hydrolysis reaction. This level of control results in a product with exceptional purity, often exceeding 97% as confirmed by gas chromatography, minimizing the formation of unwanted byproducts that could complicate downstream purification steps in herbicide synthesis.
How to Synthesize Alpha-Amino-Dialkyl Substituted Acetamide Efficiently
Implementing this patented technology requires a disciplined approach to reaction parameters to maximize yield and safety. The process begins with the preparation of an aqueous reaction mixture containing the alpha-amino-dialkyl substituted acetonitrile substrate and a catalytic amount of the chosen promoter, such as phenylacetamide or acetone. The reaction is conducted under vigorous stirring to ensure homogeneity, with the temperature carefully lowered to the 5-10°C range prior to the addition of reagents. Simultaneous dropwise addition of sodium hydroxide and hydrogen peroxide is then performed, with strict monitoring to maintain the pH within the critical 9-10 range. This controlled addition prevents local hotspots of high alkalinity that could degrade the oxidant. Following the addition, the mixture is held at low temperature for several hours to allow the primary conversion to occur, followed by a gradual warming to room temperature to drive the reaction to completion. Detailed standardized operating procedures for scaling this reaction are essential for consistent results.
- Prepare the reaction mixture by dissolving alpha-amino-dialkyl substituted acetonitrile in water with a catalytic amount of ketone (e.g., acetone) or amide (e.g., phenylacetamide).
- Maintain the reaction temperature between 5-30°C and adjust the pH to 9-10 using sodium hydroxide while slowly adding hydrogen peroxide.
- Stir the mixture for 6-8 hours until conversion is complete, then extract the product using an organic solvent like dichloromethane and purify.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic oxidative hydrolysis method presents a compelling value proposition centered on cost optimization and risk mitigation. The shift away from concentrated sulfuric acid removes a major source of operational expense related to equipment degradation and hazardous waste disposal. By utilizing a cleaner reaction profile, manufacturers can significantly reduce the overhead costs associated with environmental compliance and waste treatment infrastructure. This efficiency translates directly into a more competitive cost structure for the final agrochemical intermediate, allowing buyers to secure high-quality materials at sustainable price points. Additionally, the mild reaction conditions reduce the energy consumption required for heating or cooling, contributing to further operational savings.
- Cost Reduction in Manufacturing: The elimination of corrosive acids means that production facilities do not require specialized, high-cost alloy reactors, leading to substantial capital savings and reduced maintenance downtime. The simplified waste stream, consisting largely of water and benign organic residues, lowers the financial burden of effluent treatment compared to the heavy salt loads generated by acid neutralization processes. Furthermore, the high selectivity of the catalytic system minimizes the loss of raw materials to side reactions, ensuring that every kilogram of starting nitrile contributes maximally to the final product yield. This material efficiency is a key driver for reducing the overall cost of goods sold (COGS) in agrochemical manufacturing.
- Enhanced Supply Chain Reliability: The use of readily available and stable reagents like hydrogen peroxide and common ketones enhances the resilience of the supply chain. Unlike specialized or hazardous reagents that may face regulatory shipping restrictions or supply volatility, the inputs for this process are commodity chemicals with robust global availability. The safety profile of the process also reduces the risk of unplanned shutdowns due to safety incidents or regulatory inspections, ensuring a more consistent and reliable flow of intermediates to downstream customers. This stability is crucial for pharmaceutical and agrochemical companies that require just-in-time delivery to meet their own production schedules.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being manageable through standard cooling techniques and the absence of violent exotherms associated with strong acid mixing. This makes the technology suitable for commercial scale-up of complex agrochemical intermediates, from pilot plant batches to multi-ton production runs. From an environmental perspective, the reduction in hazardous waste generation aligns with increasingly stringent global regulations on industrial emissions. Adopting this green chemistry approach future-proofs the supply chain against tightening environmental laws, ensuring long-term operational continuity without the need for costly retrofits or process changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the advantages of using hydrogen peroxide over sulfuric acid for this synthesis?
A: The hydrogen peroxide method significantly reduces equipment corrosion and eliminates the generation of large volumes of acidic wastewater associated with traditional sulfuric acid oxidation, leading to lower environmental compliance costs.
Q: Which catalysts are most effective for this oxidative hydrolysis reaction?
A: Ketones containing methyl groups (like acetone) and amides without alpha-amino groups (like phenylacetamide or acetanilide) act as effective catalysts by stabilizing the hydrogen peroxide and facilitating proton transfer.
Q: What is the typical purity and yield achievable with this patented method?
A: Experimental data indicates that this method can achieve yields exceeding 95% with product purity levels around 97-98% as determined by GC analysis, ensuring high quality for downstream herbicide synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Amino-Dialkyl Substituted Acetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation herbicides. Our technical team has extensively analyzed the potential of the catalytic oxidative hydrolysis pathway described in CN102050754B and is fully prepared to support your project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market launch is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha-amino-dialkyl substituted acetamide meets the exacting standards required for agrochemical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both quality and cost for your agrochemical manufacturing operations.
