Advanced CO2 Catalytic Synthesis Of Sec-Butyl Urea For Scalable Agrochemical Intermediate Production
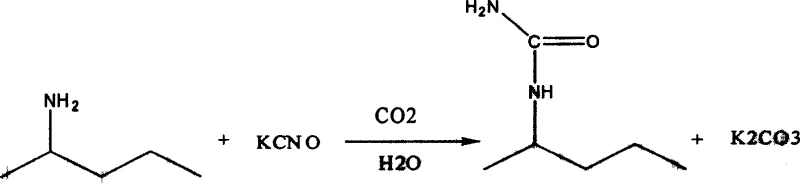
The chemical industry is constantly evolving towards more sustainable and economically viable synthesis pathways, and the production of sec-butyl urea stands as a prime example of this technological shift. Patent CN100357268C introduces a groundbreaking CO2 catalytic method that fundamentally alters the manufacturing landscape for this critical agrochemical intermediate. This innovative approach replaces the traditional, corrosive sulfuric acid catalysis with a greener, more efficient carbon dioxide-mediated process, addressing long-standing issues regarding waste management and by-product valorization. For R&D directors and procurement managers alike, understanding the nuances of this patent is essential, as it offers a pathway to significantly reduced operational expenditures while maintaining high purity standards required for downstream herbicide synthesis. The method leverages the unique reactivity of potassium cyanate and sec-butylamine in an aqueous environment, facilitated by CO2, to achieve high conversion rates without the environmental burden associated with legacy methods. This report provides a deep technical and commercial analysis of this synthesis route, highlighting its potential to transform supply chain reliability and cost structures for global chemical manufacturers seeking a reliable sec-butyl urea supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of sec-butyl urea has relied heavily on sulfuric acid catalysis, a method that, while effective in achieving high yields, presents substantial economic and environmental drawbacks that hinder modern manufacturing efficiency. In the traditional process, potassium cyanate and sec-butylamine are reacted in the presence of 35% sulfuric acid, which acts as the catalyst to drive the urea formation. Although this method can achieve yields around 95%, the resulting by-product is potassium sulfate, a compound with relatively low market value and limited industrial application compared to other potassium salts. This low-value by-product creates a significant waste management burden, requiring additional processing steps for disposal or finding niche markets, which ultimately inflates the overall production cost. Furthermore, the use of concentrated sulfuric acid introduces severe safety hazards, including corrosion of reactor equipment and the need for specialized handling protocols, which increases capital expenditure on maintenance and safety infrastructure. The acidic nature of the reaction medium also complicates the work-up procedure, often requiring neutralization steps that generate additional saline waste streams, thereby negatively impacting the environmental footprint of the manufacturing facility. For supply chain heads, these factors translate into higher volatility in pricing and potential regulatory risks associated with hazardous waste disposal, making the conventional method less attractive for long-term strategic sourcing.
The Novel Approach
In stark contrast to the legacy sulfuric acid route, the novel CO2 catalytic method described in the patent offers a paradigm shift by utilizing carbon dioxide as a benign and effective promoter for the synthesis reaction. This approach eliminates the need for corrosive mineral acids, thereby reducing equipment corrosion and extending the lifespan of reactor vessels, which is a critical factor for cost reduction in agrochemical intermediate manufacturing. The reaction proceeds in an aqueous medium where CO2 is bubbled through the mixture of sec-butylamine and potassium cyanate, facilitating the formation of the urea linkage under mild conditions. One of the most compelling advantages of this new method is the generation of potassium carbonate as the primary by-product, a chemical with substantial market value and wide-ranging applications in the glass, detergent, and fertilizer industries. This high-value by-product can be easily isolated via hot filtration with alcohol, creating an additional revenue stream that offsets the production costs of the sec-butyl urea itself. Moreover, the process allows for the recovery and recycling of excess sec-butylamine through distillation, ensuring that raw material utilization is maximized and waste is minimized. This closed-loop capability not only enhances the sustainability profile of the process but also provides a buffer against raw material price fluctuations, offering a more stable and predictable cost structure for procurement teams.
Mechanistic Insights into CO2-Catalyzed Urea Formation
The core of this technological advancement lies in the unique mechanistic role that carbon dioxide plays in the aqueous reaction system, acting not merely as a reactant but as a catalyst that modulates the pH and reactivity of the amine and cyanate species. When CO2 is introduced into the aqueous solution containing sec-butylamine, it forms carbamate species in situ, which alters the nucleophilicity of the amine and facilitates its attack on the cyanate ion. This mechanism avoids the harsh protonation effects seen in strong acid catalysis, preserving the integrity of the reactants and minimizing side reactions that could lead to impurity formation. The reaction conditions are carefully optimized, with temperatures maintained between 30°C and 60°C and pressures ranging from 0.1 MPa to 5 MPa, ensuring that the CO2 remains dissolved and active within the liquid phase. This mild thermal profile is crucial for preventing the decomposition of sensitive intermediates and ensures that the energy consumption of the process remains low, contributing to the overall energy efficiency of the plant. The stoichiometry of the reaction is also flexible, allowing for an excess of sec-butylamine which drives the equilibrium towards the product side, and this excess can be subsequently recovered, demonstrating the robustness of the chemical engineering design.
Impurity control is another critical aspect where this CO2 catalytic method excels, particularly when compared to the sulfuric acid route which often generates sulfonated by-products or salt contaminants that are difficult to remove. In the new process, the primary impurities are unreacted starting materials and the potassium carbonate by-product, both of which are easily separable through physical means such as distillation and filtration. The use of alcohol for hot filtration effectively precipitates the potassium carbonate, leaving the sec-butyl urea in the solution, which is then concentrated under reduced pressure to yield the final product. This simplicity in purification reduces the need for complex chromatographic separations or extensive washing steps, which are often bottlenecks in commercial scale-up of complex agrochemical intermediates. Furthermore, the aqueous nature of the reaction medium ensures that any inorganic salts remain dissolved or precipitate cleanly, preventing the entrapment of impurities within the crystal lattice of the product. For R&D directors focused on purity and impurity profiles, this mechanism offers a clear path to achieving high-purity sec-butyl urea that meets the stringent specifications required for downstream herbicide synthesis, reducing the risk of batch failures in subsequent chemical transformations.
How to Synthesize Sec-Butyl Urea Efficiently
The practical implementation of this synthesis route involves a series of well-defined unit operations that are easily adaptable to existing chemical manufacturing infrastructure, ensuring a smooth transition from laboratory scale to industrial production. The process begins with the preparation of the reactant solutions, where potassium cyanate is dissolved in water and sec-butylamine is diluted in a separate vessel, allowing for precise control over the molar ratios and concentration levels. These solutions are then combined in a reactor equipped with gas sparging capabilities, where CO2 is introduced at a controlled flow rate to maintain the desired pressure and reaction kinetics. The reaction mixture is stirred vigorously to ensure mass transfer of the gas into the liquid phase, and the temperature is regulated to stay within the optimal 30°C to 60°C window. Following the reaction period, which typically lasts between 2 to 6 hours depending on the specific pressure and temperature settings, the mixture undergoes a distillation step to remove water and recover the excess sec-butylamine for reuse. The remaining residue is then treated with an alcohol, such as ethanol or methanol, to precipitate the potassium carbonate by-product, which is filtered off while hot. Finally, the filtrate is concentrated under vacuum to remove the alcohol solvent, yielding the pure sec-butyl urea product ready for packaging or further processing.
- Dissolve potassium cyanate in water to prepare a homogeneous aqueous solution for the reaction mixture.
- Mix sec-butylamine with water in a reactor and add the potassium cyanate solution under stirring.
- Bubble CO2 gas into the solution at 30-60°C and 0.1-5 MPa, then isolate the product via filtration and concentration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CO2 catalytic synthesis method presents a compelling value proposition that extends far beyond simple chemical conversion, offering tangible benefits in cost structure and supply reliability. The elimination of sulfuric acid removes a major hazardous material from the supply chain, reducing the regulatory burden and insurance costs associated with storing and handling corrosive acids. Additionally, the generation of potassium carbonate as a saleable by-product creates a new revenue stream that can significantly subsidize the production cost of the sec-butyl urea, effectively lowering the net cost of goods sold. This economic advantage is further amplified by the ability to recycle excess sec-butylamine, which reduces the consumption of this key raw material and insulates the manufacturing process from market volatility in amine pricing. The mild reaction conditions also translate to lower energy consumption and reduced wear and tear on equipment, leading to lower maintenance costs and higher overall equipment effectiveness. These factors combined result in a more resilient supply chain capable of withstanding market fluctuations and delivering consistent value to downstream customers.
- Cost Reduction in Manufacturing: The shift from sulfuric acid to CO2 catalysis fundamentally alters the cost equation by replacing a waste-generating reagent with a value-generating one. The production of high-value potassium carbonate instead of low-value potassium sulfate means that the waste stream is monetized, directly offsetting production expenses. Furthermore, the recycling of excess sec-butylamine ensures that raw material costs are minimized, as the unreacted amine is not lost but rather reintroduced into the process. This closed-loop material flow significantly reduces the variable cost per kilogram of product, allowing for more competitive pricing strategies in the global market. The reduction in equipment corrosion also leads to substantial long-term savings on capital maintenance and replacement, further enhancing the financial viability of the process.
- Enhanced Supply Chain Reliability: The simplicity and robustness of the CO2 catalytic method contribute to a more reliable supply chain by reducing the number of potential failure points in the manufacturing process. The use of common and readily available raw materials such as CO2, water, and potassium cyanate ensures that supply disruptions are minimized, as these commodities are widely sourced and stable. The ability to operate under mild conditions reduces the risk of thermal runaways or pressure-related incidents, ensuring continuous operation and consistent output. For supply chain heads, this reliability translates to reduced lead time for high-purity agrochemical intermediates, as the process is less prone to unplanned downtime or quality deviations that could delay shipments. The consistent quality of the product also reduces the need for extensive incoming quality control testing by customers, streamlining the entire procurement-to-payment cycle.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is ideally suited for large-scale commercial production due to its green chemistry credentials and ease of scale-up. The absence of hazardous acidic waste streams simplifies wastewater treatment and reduces the environmental footprint of the facility, ensuring compliance with increasingly stringent global environmental regulations. The use of CO2, a greenhouse gas, as a reactant also contributes to carbon utilization efforts, enhancing the sustainability profile of the manufactured product. The process design, involving simple mixing, gas sparging, and filtration, is easily scalable from pilot plant to multi-ton production without the need for complex engineering modifications. This scalability ensures that supply can be rapidly ramped up to meet surging demand, providing a strategic advantage in dynamic market conditions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the CO2 catalytic synthesis of sec-butyl urea, providing clarity on the process capabilities and advantages. These insights are derived directly from the patent data and technical analysis, ensuring that stakeholders have accurate information for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines or sourcing strategies.
Q: What is the primary advantage of the CO2 catalytic method over sulfuric acid catalysis?
A: The CO2 catalytic method generates potassium carbonate as a by-product, which has significantly higher market value and utility compared to the low-value potassium sulfate produced by the traditional sulfuric acid method.
Q: Can the excess sec-butylamine be recovered in this process?
A: Yes, the process allows for the distillation and recovery of excess sec-butylamine, which can be recycled back into the reactor, thereby improving overall atom economy and reducing raw material costs.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction typically operates under mild conditions with temperatures ranging from 30°C to 60°C and pressures between 0.1 MPa and 5 MPa, ensuring safety and ease of scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sec-Butyl Urea Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern chemical industry, and we are fully equipped to leverage the CO2 catalytic technology for the commercial production of sec-butyl urea. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of sec-butyl urea meets the exacting standards required for agrochemical applications. Our commitment to quality and consistency makes us a trusted partner for global enterprises seeking to optimize their supply chains with high-performance intermediates.
We invite you to collaborate with us to explore how this advanced synthesis method can drive value for your organization. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient and environmentally responsible.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
