Advanced Topiroxostat Manufacturing: High-Purity Routes for Global Pharma Supply Chains
Introduction to Advanced Topiroxostat Synthesis Technology
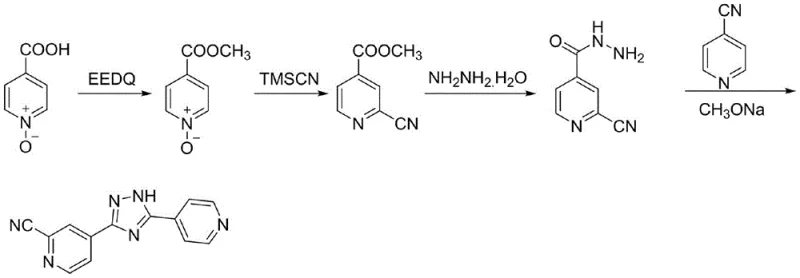
The pharmaceutical industry is constantly seeking more efficient and safer pathways for the production of critical active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is documented in Chinese patent CN114315800B, which details a novel preparation method for Topiroxostat, a potent selective xanthine oxidoreductase (XOR) inhibitor used for treating gout and hyperuricemia. This patent outlines a robust synthetic strategy that addresses long-standing challenges in yield optimization and impurity control. By leveraging a unique combination of nucleophilic substitution and low-temperature amidation, the disclosed technology achieves a final product purity of 99.8%, setting a new benchmark for quality in pharmaceutical intermediates manufacturing. For global procurement leaders and R&D directors, understanding the nuances of this pathway is essential for securing a reliable supply chain and ensuring the economic viability of generic drug production.
The significance of this technology extends beyond mere academic interest; it represents a tangible solution to the bottlenecks faced in current industrial practices. Traditional methods often rely on harsh conditions that compromise the integrity of sensitive functional groups, such as the cyano group, leading to substantial material loss and complex waste streams. In contrast, the approach described in CN114315800B emphasizes mild reaction conditions and high atom economy. This shift not only enhances the safety profile of the manufacturing process but also aligns with the increasing regulatory pressure for greener chemistry solutions. As we delve deeper into the technical specifics, it becomes clear that this methodology offers a compelling value proposition for stakeholders focused on cost reduction in API manufacturing and operational excellence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Topiroxostat have been plagued by inefficiencies that hinder scalable production. For instance, the route described in patent CN1561340 utilizes isonicotinic acid-N-oxide as a starting material, undergoing methylation, cyanation, hydrazinolysis, and cyclization. While conceptually sound, the practical execution suffers from dismal yields, with the hydrazinolysis step achieving only 49.2% and the subsequent cyclization merely 57.6%. Such low efficiency translates directly into higher raw material consumption and increased waste generation, creating a heavy burden on both cost structures and environmental compliance teams. Furthermore, other reported methods, such as those in CN105130958A, attempt to avoid toxic cyanides but result in a catastrophic total route yield of only 9.5%, rendering them commercially unviable for large-scale operations.
Beyond yield issues, safety remains a paramount concern in legacy synthesis routes. Many conventional processes rely on the use of highly toxic sodium cyanide as a cyanide source, necessitating stringent safety protocols and specialized equipment to handle hazardous reagents. Additionally, these methods often require high-temperature reactions over extended periods, which significantly increases the risk of cyano group hydrolysis. This side reaction not only degrades the desired product but also generates difficult-to-remove impurities that complicate downstream purification. The cumulative effect of these drawbacks is a fragile supply chain susceptible to disruptions, quality failures, and escalating production costs, highlighting the urgent need for a more robust and reliable alternative.
The Novel Approach
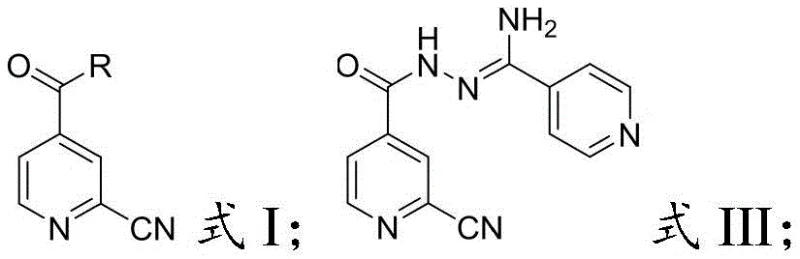
The methodology presented in CN114315800B offers a transformative solution by reimagining the construction of the triazole core. Instead of relying on multi-step sequences with poor convergence, this novel approach employs a convergent strategy centered on the coupling of two key fragments: a functionalized pyridine derivative (Formula I) and an amidrazone intermediate (Formula II). This strategic pivot allows for the direct formation of the critical amide bond under controlled conditions, bypassing the inefficient steps seen in prior art. The result is a streamlined process where individual step yields consistently exceed 90%, dramatically improving the overall mass balance and reducing the cost of goods sold (COGS) for the final API.
A defining feature of this new route is the implementation of low-temperature amidation, typically conducted between -20°C and 40°C. This precise thermal control is crucial for preserving the stability of the cyano group, effectively eliminating the hydrolysis risks associated with high-temperature legacy methods. By utilizing activated acid derivatives such as acid chlorides, esters, or mixed anhydrides for Formula I, the reaction proceeds rapidly and cleanly. The subsequent cyclization step is equally optimized, utilizing simple alcoholic solvents to close the triazole ring with high fidelity. This combination of mild conditions and high-yielding steps ensures that the process is not only chemically superior but also operationally simpler, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Low-Temperature Amidation and Cyclization
The chemical elegance of this synthesis lies in the careful manipulation of reactivity to favor the desired transformation while suppressing side reactions. The process begins with the nucleophilic attack of hydrazine hydrate on 4-cyanopyridine to generate the amidrazone species (Formula II). This intermediate is highly nucleophilic at the terminal nitrogen, poised to attack the electrophilic carbonyl carbon of the activated acid derivative (Formula I). The choice of activating group 'R' in Formula I—whether it be a chloride, an alkoxy group, or an imidazole moiety—dictates the specific reaction kinetics and conditions required. For example, when using acid chlorides, the reaction is fast and exothermic, necessitating the use of acid-binding agents like triethylamine to scavenge generated HCl and drive the equilibrium forward.
Following the formation of the acylated intermediate (Formula III), the system undergoes an intramolecular cyclization to form the 1,2,4-triazole ring. This step involves the nucleophilic attack of the internal hydrazine nitrogen onto the nitrile carbon, followed by tautomerization and aromatization. The patent specifies that this cyclization is best performed in alcoholic solvents at elevated temperatures (50°C to 150°C). The mechanistic pathway ensures that the cyano group, which was vulnerable in previous high-temperature cyanation steps, is now consumed constructively to build the heterocyclic core. This intrinsic protection of the functional group throughout the synthesis minimizes the formation of carboxylic acid byproducts, thereby simplifying the impurity profile and facilitating easier purification to meet stringent purity specifications required for pharmaceutical grade materials.
How to Synthesize Topiroxostat Efficiently
Implementing this advanced synthesis route requires a clear understanding of the operational parameters defined in the patent. The process is divided into three distinct stages: the preparation of the amidrazone, the coupling reaction to form the triazole precursor, and the final ring closure. Each stage has been optimized to maximize yield and minimize waste, providing a clear roadmap for process chemists aiming to replicate these results. The flexibility of the method, allowing for various activated acid derivatives, offers manufacturers the ability to tailor the process based on available raw materials and existing infrastructure, further enhancing its adaptability for different production scales.
- Perform nucleophilic reaction between 4-cyanopyridine and hydrazine hydrate to generate the key amidrazone intermediate (Formula II).
- Conduct low-temperature amidation between the activated acid derivative (Formula I) and Formula II to form the triazole precursor (Formula III).
- Execute thermal cyclization of Formula III in alcoholic solvents to finalize the Topiroxostat structure with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route presents a multitude of strategic benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the manufacturing workflow. By consolidating multiple low-yielding steps into a high-efficiency sequence, the overall throughput of the production line is significantly enhanced. This improvement in process efficiency directly correlates to a reduction in the consumption of raw materials and solvents, leading to substantial cost savings in the procurement budget. Moreover, the elimination of toxic reagents like sodium cyanide removes the need for expensive hazard mitigation measures and specialized waste disposal contracts, further driving down the operational expenditure associated with producing this critical API intermediate.
- Cost Reduction in Manufacturing: The high step yields reported in the patent, often exceeding 90%, mean that less starting material is required to produce a kilogram of the final product compared to legacy routes with yields below 50%. This improved atom economy translates directly into lower variable costs per unit. Additionally, the ability to perform reactions under mild conditions reduces energy consumption for heating and cooling, contributing to a leaner manufacturing cost structure. The simplified purification requirements, driven by the high purity of intermediates, also reduce the load on downstream processing units, allowing for faster batch turnover and better asset utilization.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable raw materials, such as 4-cyanopyridine and common carboxylic acid derivatives, mitigates the risk of supply disruptions often associated with exotic or hazardous reagents. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification results. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, ensuring that medication reaches patients without interruption. The process's inherent safety also lowers insurance premiums and regulatory scrutiny, smoothing the path for uninterrupted commercial operations.
- Scalability and Environmental Compliance: The generation of minimal three wastes (waste water, waste gas, and solid waste) positions this method as an environmentally sustainable choice. In an era of increasing regulatory pressure on pharmaceutical manufacturing emissions, adopting a greener synthesis route provides a competitive edge. The process is designed to be easily scalable from pilot plant to full commercial production without significant re-engineering, ensuring that supply can be ramped up quickly to meet market demand. This scalability, combined with a reduced environmental footprint, makes the technology highly attractive for long-term strategic partnerships focused on sustainable growth.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial implications of this synthesis technology. These answers are derived directly from the data and claims presented in patent CN114315800B, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing supply networks and assess the potential return on investment for adopting this advanced manufacturing technique.
Q: How does the new Topiroxostat synthesis route improve upon traditional methods?
A: Unlike prior art methods which suffer from low yields (e.g., 9.5% total yield in CN105130958A) and toxic cyanide usage, the new route utilizes mild low-temperature amidation to prevent cyano group hydrolysis, achieving step yields over 90% and final purity of 99.8%.
Q: What are the safety advantages of this preparation method?
A: The process eliminates the need for highly toxic sodium cyanide typically used in conventional cyanation steps. Furthermore, conducting the amidation reaction at low temperatures (-20°C to 40°C) significantly reduces the risk of exothermic runaway and cyano group degradation.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It uses readily available raw materials, avoids complex purification steps due to high intermediate purity, and generates minimal three wastes, making it economically and environmentally viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Topiroxostat Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing cutting-edge synthesis technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the methodology described in CN114315800B and possesses the technical capability to implement this high-yield route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Topiroxostat intermediate meets the highest international standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can optimize your supply chain and drive value for your organization. Together, we can ensure a secure, cost-effective, and high-quality supply of this vital pharmaceutical intermediate.
