Advanced Cell-Free Biocatalysis for Commercial Scale-Up of Optically Pure Aryl Alcohols
The landscape of chiral synthesis is undergoing a transformative shift with the emergence of advanced biocatalytic strategies that bridge the gap between enzymatic precision and industrial robustness. Patent CN101857887A introduces a groundbreaking methodology for preparing optically pure aryl alcohols through the asymmetric conversion catalyzed by recombinant bacterial cell-free extracts. This technology represents a significant departure from traditional whole-cell fermentation or isolated pure enzyme protocols, offering a unique hybrid advantage that addresses critical bottlenecks in stereochemical manufacturing. By leveraging the intracellular machinery of recombinant E. coli expressing carbonyl reductase SCR1 without the confinement of the cell wall, this invention achieves exceptional reaction kinetics and stereoselectivity. The core innovation lies in the ability to maintain efficient cofactor regeneration cycles without the need for exogenous coupling enzymes, a feature that drastically simplifies the reaction matrix. For R&D directors and process chemists, this patent outlines a pathway to high-value chiral intermediates with optical purities reaching up to 100% e.e., setting a new benchmark for quality in fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional biocatalytic processes for synthesizing chiral alcohols have long been plagued by inherent inefficiencies that hinder large-scale adoption and cost-effectiveness. In whole-cell catalytic systems, the intact cell membrane acts as a formidable diffusion barrier, restricting the transport of hydrophobic substrates into the cytoplasm and slowing down the efflux of products, which often leads to prolonged reaction times ranging from 12 to 48 hours. Furthermore, when utilizing isolated pure enzymes, the economic viability is frequently compromised by the prohibitive cost of stoichiometric amounts of cofactors like NADPH, necessitating the addition of expensive secondary coupling enzymes such as glucose dehydrogenase to sustain the catalytic cycle. These auxiliary enzymes not only increase the raw material costs but also complicate the downstream purification process by introducing additional protein contaminants that must be removed to meet stringent pharmaceutical specifications. The complexity of managing multiple enzyme components and the sluggish mass transfer rates in whole-cell systems create a substantial burden on manufacturing throughput and operational expenditure.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this paradigm by employing a cell-free extract system that retains the catalytic power of the intracellular environment while eliminating the physical constraints of the cell membrane. This cell-free lysate contains the target carbonyl reductase along with endogenous metabolic factors that facilitate cofactor regeneration, thereby obviating the need for adding external coupling enzymes. By simply supplementing the reaction with a cost-effective cosubstrate like glucose and a trace amount of initial NADP+, the system achieves a self-sustaining catalytic loop with total turnover numbers exceeding 3000. This structural simplification of the catalyst system translates directly into enhanced reaction rates, reducing the processing time to a mere 2 to 12 hours, which is a dramatic improvement over conventional methods. The removal of the cell wall barrier ensures rapid substrate accessibility, while the absence of extraneous coupling proteins streamlines the workup procedure, offering a cleaner and more economically attractive route for the production of optically active aryl alcohols.
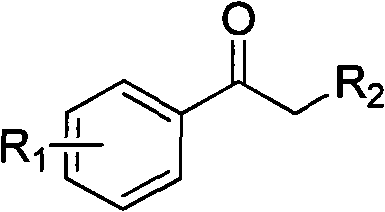
Mechanistic Insights into Cell-Free Extract Catalyzed Asymmetric Reduction
The mechanistic elegance of this biocatalytic system relies on the precise stereochemical control exerted by the carbonyl reductase SCR1 enzyme derived from Candida parapsilosis. Within the cell-free extract, the enzyme maintains its native conformation and active site geometry, which is critical for distinguishing between the prochiral faces of the aryl ketone substrate. The reduction proceeds via a hydride transfer mechanism where the nicotinamide ring of the reduced cofactor NADPH delivers a hydride ion to the carbonyl carbon with absolute spatial specificity, resulting in the formation of the (S)-enantiomer with high fidelity. The presence of various substituents on the aromatic ring, such as chloro, methyl, or methoxy groups, is well-tolerated by the enzyme's active site, allowing for a broad substrate scope without significant loss in enantioselectivity. This robustness is essential for pharmaceutical applications where minor structural variations in the intermediate can dictate the efficacy of the final drug substance.
Furthermore, the impurity profile of the reaction is meticulously controlled by the inherent selectivity of the biocatalyst, which minimizes the formation of side products commonly associated with chemical reduction methods using metal hydrides. The cell-free environment allows for fine-tuning of reaction parameters such as pH and ionic strength, which stabilizes the enzyme and prevents non-specific background reactions. The regeneration of the oxidized cofactor NADP+ back to NADPH is seamlessly integrated into the reaction matrix through the oxidation of the added cosubstrate, ensuring that the catalytic cycle continues uninterrupted without the accumulation of inhibitory byproducts. This closed-loop cofactor economy is a hallmark of the system's efficiency, enabling high substrate loading concentrations up to 80 mM while maintaining consistent optical purity levels above 90% e.e. throughout the conversion.
How to Synthesize Optically Pure Aryl Alcohol Efficiently
Implementing this synthesis route requires a disciplined approach to bioprocess engineering, starting with the cultivation of the recombinant host and the preparation of the active lysate. The protocol involves inducing the expression of the SCR1 gene in E. coli BL21(DE3), followed by cell disruption via ultrasonication to release the soluble protein fraction. This crude extract serves as the biocatalyst, ready to be deployed in either aqueous or organic-aqueous biphasic systems depending on the solubility characteristics of the specific aryl ketone substrate. The detailed standardized synthesis steps, including precise buffer compositions, induction temperatures, and extraction protocols, are outlined in the structured guide below to ensure reproducibility and optimal yield.
- Prepare recombinant E. coli BL21(DE3) expressing carbonyl reductase SCR1, harvest cells, and perform ultrasonication to obtain cell-free extract supernatant.
- Formulate the reaction system with potassium phosphate buffer, cell-free extract, cosubstrate (e.g., glucose), trace NADP+, and the target aryl ketone substrate.
- Incubate the mixture at 20°C for 2 to 12 hours, followed by ethyl acetate extraction and chiral HPLC analysis to confirm optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this cell-free biocatalytic technology offers compelling economic and operational benefits that directly impact the bottom line. The elimination of expensive coupling enzymes and the reduction in cofactor requirements represent a significant decrease in raw material costs, making the production of high-purity chiral intermediates more financially sustainable. Additionally, the drastic reduction in reaction time enhances facility utilization rates, allowing for more batches to be processed within the same timeframe, which effectively increases production capacity without the need for capital-intensive infrastructure expansion. The simplicity of the downstream processing, owing to the absence of whole cells and auxiliary proteins, further reduces solvent consumption and waste generation, aligning with green chemistry principles and lowering environmental compliance costs.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the removal of costly auxiliary enzymes typically required for cofactor regeneration in pure enzyme systems. By relying on the intrinsic capabilities of the cell-free extract and inexpensive cosubstrates like glucose, the direct material costs are substantially lowered. This simplification also reduces the complexity of inventory management, as fewer specialized biological reagents need to be sourced and stored, leading to streamlined procurement operations and reduced risk of supply disruptions for critical catalyst components.
- Enhanced Supply Chain Reliability: The robustness of the recombinant bacterial strain and the stability of the cell-free extract contribute to a highly reliable supply chain for chiral intermediates. The process is less susceptible to the variability often seen in whole-cell fermentations, ensuring consistent batch-to-batch quality and yield. This predictability allows for more accurate demand forecasting and inventory planning, minimizing the risk of stockouts for key pharmaceutical building blocks. Furthermore, the scalability of the fermentation and extraction steps ensures that supply can be rapidly ramped up to meet surging market demands without compromising product integrity.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the cell-free system offers distinct advantages by generating less biological waste compared to whole-cell processes. The absence of cell biomass simplifies the separation of the organic product, reducing the volume of wastewater and the load on treatment facilities. This cleaner process profile facilitates easier regulatory approval and compliance with increasingly stringent environmental regulations. The ability to operate in biphasic systems also enhances the solubility of hydrophobic substrates, enabling higher concentration reactions that improve space-time yields and make the process more attractive for multi-ton commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cell-free biocatalytic technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational parameters and performance metrics. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: How does the cell-free extract system improve upon traditional whole-cell catalysis?
A: The cell-free system eliminates the cell membrane barrier, significantly reducing mass transfer resistance for substrates and products. This results in a drastic reduction in reaction time from 12-48 hours down to 2-12 hours while maintaining high optical purity.
Q: Is additional coupling enzyme required for cofactor regeneration in this process?
A: No. Unlike conventional pure enzyme systems that require expensive coupling enzymes like glucose dehydrogenase, this cell-free extract system utilizes intrinsic mechanisms or simple cosubstrates to regenerate NADPH, simplifying the catalyst composition and lowering costs.
Q: What represents the typical optical purity and yield for this biocatalytic method?
A: The process consistently achieves optical purity ranging from 90% to 100% e.e. with product yields between 50% and 90%, making it highly suitable for the production of high-value chiral pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Pure Aryl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the cell-free extract technology described in CN101857887A for the production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of optically pure aryl alcohol meets the exacting standards required by the global pharmaceutical industry. We are committed to leveraging this innovative biocatalytic approach to deliver superior quality products with unmatched consistency.
We invite forward-thinking partners to collaborate with us to explore the full commercial potential of this technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient synthesis route can optimize your supply chain economics. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the market for high-purity pharmaceutical intermediates.
