Advanced Grignard Synthesis for High-Purity Halogenated Methyl-Benzaldehyde Intermediates
Advanced Grignard Synthesis for High-Purity Halogenated Methyl-Benzaldehyde Intermediates
The pharmaceutical and agrochemical industries rely heavily on the availability of high-quality aromatic aldehydes as critical building blocks for complex molecular architectures. Patent CN101712603A introduces a significant technological advancement in the preparation of halogenated methyl-benzaldehyde through a refined Grignard reaction pathway. This method addresses long-standing challenges in the synthesis of these vital intermediates by offering a route that combines operational simplicity with high product integrity. By utilizing halogenated benzene derivatives and N,N-dimethylformamide (DMF) under controlled conditions, the process achieves product purity levels exceeding 99% with yields consistently surpassing 50%. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this patented technology is essential for securing a stable supply chain for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated benzaldehydes has been plagued by severe operational constraints and safety hazards associated with traditional methodologies. The Gattermann-Koch reaction, often cited in literature such as US6300525, necessitates the use of carbon monoxide under high pressure in the presence of corrosive catalyst systems like hydrogen chloride-aluminum chloride. This not only demands expensive, specialized high-pressure reactor infrastructure but also introduces significant safety risks due to the toxicity of CO and the corrosive nature of the catalysts. Furthermore, the reaction efficiency is highly dependent on the electronic nature of the substrate; it proceeds smoothly only with electron-donating groups, while electron-withdrawing substituents lead to sluggish reaction rates and poor industrial viability. Alternative methods like the Stephen reduction reported in WO2006004903 involve prolonged reaction times of up to 13 hours and the use of stannous chloride, generating substantial heavy metal waste that complicates environmental compliance and increases disposal costs significantly.
The Novel Approach
In stark contrast to these legacy processes, the novel approach detailed in the patent leverages the versatility of the Grignard reaction coupled with Bouveault aldehyde synthesis to circumvent these historical bottlenecks. This method operates under atmospheric pressure, eliminating the need for costly high-pressure equipment and the associated safety protocols required for handling carbon monoxide. The reaction conditions are remarkably mild, with Grignard reagent formation occurring between 10-80°C and the subsequent formylation step proceeding efficiently at temperatures ranging from -40°C to 45°C. By replacing hazardous catalysts with readily available magnesium and using DMF as the formylating agent, the process drastically simplifies the operational workflow. This shift not only enhances the safety profile of the manufacturing facility but also opens the door for a broader range of substrates, including those with electron-withdrawing groups, to be converted into valuable aldehyde intermediates with improved consistency and reliability.
Mechanistic Insights into Grignard-Mediated Bouveault Aldehyde Synthesis
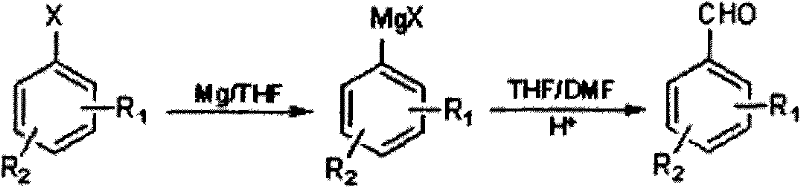
The core of this innovative synthesis lies in the precise generation and utilization of the Grignard reagent, which serves as a potent nucleophile for the subsequent formylation step. The process begins with the activation of magnesium chips in an anhydrous organic solvent, typically tetrahydrofuran (THF), using initiators such as iodine particles or 1,2-dibromoethane to overcome the induction period often seen in Grignard formations. Once the reactive organomagnesium species is generated from the halogenated benzene substrate, it attacks the carbonyl carbon of N,N-dimethylformamide (DMF). This nucleophilic addition forms a stable intermediate complex which, upon acidic hydrolysis, collapses to release the desired halogenated methyl-benzaldehyde. The elegance of this mechanism is its generality; it accommodates various substitution patterns on the benzene ring, allowing for the synthesis of diverse derivatives like 3-chloro-4-bromotoluene conversions without the regioselectivity issues often encountered in electrophilic aromatic substitution reactions.
Controlling impurities is paramount in pharmaceutical intermediate manufacturing, and this protocol incorporates specific measures to ensure high purity standards. The reaction temperature during the DMF addition phase is critically managed, cooling the Grignard reagent to between -40°C and 10°C before the dropwise addition of the DMF solution. This thermal control minimizes side reactions such as Wurtz coupling or over-addition, which could lead to difficult-to-remove byproducts. Following the reaction, the mixture is carefully quenched by adjusting the pH to less than or equal to 2 using acids like hydrochloric acid or ammonium chloride. This acidic workup ensures the complete decomposition of magnesium complexes and facilitates the separation of the organic product from inorganic salts. The final purification via steam distillation further refines the product, removing residual solvents and high-boiling impurities to achieve the reported purity of ≥99%, making it suitable for sensitive downstream applications.
How to Synthesize Halogenated Methyl-Benzaldehyde Efficiently
The practical implementation of this synthesis route requires strict adherence to anhydrous conditions and precise temperature control to maximize yield and safety. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to pilot plant operations without requiring fundamental changes to the reaction chemistry. Operators must ensure that all glassware and solvents are thoroughly dried to prevent premature quenching of the Grignard reagent by moisture. The initiation step is crucial; adding a small portion of the halide solution to the magnesium slurry allows for the exothermic formation of the reagent to be managed safely before the bulk addition proceeds. Detailed standard operating procedures regarding the specific molar ratios of magnesium to halide (1:1 to 1:1.5) and the choice of solvent systems are critical for reproducibility. For a comprehensive guide on executing this synthesis with optimal parameters, please refer to the standardized protocol below.
- Initiate Grignard reagent formation by reacting magnesium chips with halogenated benzene in anhydrous THF using iodine or 1,2-dibromoethane as an initiator at 10-80°C.
- Cool the resulting Grignard reagent to -40 to 10°C and slowly add a solution of N,N-dimethylformamide (DMF) in organic solvent, then warm to 10-45°C for reaction.
- Quench the reaction mixture by adjusting pH to ≤2 with acid, extract with dichloromethane, remove solvent, and purify via steam distillation to obtain the final aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this Grignard-based methodology represents a strategic opportunity to optimize both cost structures and supply reliability. Traditional methods often rely on specialized reagents or extreme conditions that create single points of failure in the supply chain; for instance, reliance on high-pressure carbon monoxide limits the number of qualified contract manufacturers capable of performing the synthesis. By shifting to a process that utilizes commodity chemicals like magnesium turnings, THF, and DMF, the barrier to entry for production is significantly lowered. This diversification of potential manufacturing partners enhances supply chain resilience, ensuring that disruptions at one facility do not halt the entire production line. Furthermore, the elimination of heavy metal catalysts like tin or aluminum chloride reduces the burden on waste treatment facilities, leading to substantial cost savings in environmental compliance and hazardous waste disposal.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction infrastructure and the reduction in raw material costs. By avoiding the need for high-pressure reactors and specialized corrosion-resistant equipment required for Gattermann-Koch reactions, capital expenditure for production facilities is drastically reduced. Additionally, the use of inexpensive and widely available initiators like iodine or 1,2-dibromoethane replaces costly and hazardous catalytic systems. The streamlined workup procedure, which avoids complex chromatographic separations in favor of extraction and distillation, further lowers operational expenses by reducing solvent consumption and processing time. These factors combine to deliver a more cost-effective manufacturing route without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust, commercially available starting materials that are not subject to the same regulatory restrictions as toxic gases or heavy metals. Halogenated benzenes and DMF are produced on a massive global scale, ensuring a stable supply even during market fluctuations. The moderate reaction conditions also mean that the process can be run in a wider variety of standard chemical manufacturing plants, increasing the pool of potential suppliers. This flexibility allows procurement teams to negotiate better terms and secure longer-term contracts, mitigating the risk of price volatility. The ability to produce a wide range of substituted benzaldehydes using the same core platform technology also simplifies inventory management and reduces the need for multiple specialized production lines.
- Scalability and Environmental Compliance: From an environmental perspective, this method offers a cleaner alternative to traditional oxidation or reduction pathways that generate significant amounts of heavy metal waste. The primary byproducts are magnesium salts, which are relatively benign and easier to treat than tin or chromium residues. The process operates at near-atmospheric pressure, reducing the energy footprint associated with compression and high-temperature maintenance. Scalability is inherently supported by the batch-wise nature of the Grignard formation, which is a well-understood unit operation in the fine chemical industry. This allows for seamless scale-up from kilogram to tonne quantities, ensuring that the supply can grow in tandem with the demand for downstream pharmaceutical products while maintaining strict adherence to modern environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Grignard-based synthesis for halogenated benzaldehydes. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety profiles, and substrate scope. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific project needs.
Q: What are the primary advantages of this Grignard method over traditional Gattermann-Koch reactions?
A: Unlike the Gattermann-Koch reaction which requires high pressure and hazardous catalysts like hydrogen fluoride-aluminum chloride, this Grignard-based method operates under atmospheric pressure with milder conditions, significantly improving operational safety and equipment requirements while maintaining yields above 50%.
Q: How does this process ensure high product purity for pharmaceutical applications?
A: The process utilizes a controlled acidic workup (pH ≤ 2) followed by extraction and steam distillation, which effectively removes magnesium salts and unreacted starting materials, consistently achieving product purity levels greater than or equal to 99% suitable for sensitive API synthesis.
Q: Is this synthesis route scalable for industrial production of diverse halogenated substrates?
A: Yes, the patent demonstrates successful application across various substrates including 3-chloro-4-bromotoluene and 2-methoxy-4-fluorobromobenzene, utilizing common solvents like THF and standard initiators, indicating strong potential for commercial scale-up without specialized high-pressure reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Methyl-Benzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the success of your drug development pipelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our expertise in Grignard chemistry and aldehyde synthesis allows us to optimize the process described in CN101712603A, guaranteeing consistent quality and reliable delivery schedules for your most demanding projects.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific application. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for cost reduction and supply chain security in the competitive landscape of fine chemical manufacturing.
