Optimizing Rivastigmine Production: A Technical Deep Dive into Ethylmethylcarbamoyl Chloride Manufacturing
Optimizing Rivastigmine Production: A Technical Deep Dive into Ethylmethylcarbamoyl Chloride Manufacturing
The global demand for effective treatments against Alzheimer's disease continues to drive innovation in the synthesis of key pharmaceutical intermediates, specifically ethylmethylcarbamoyl chloride, a critical precursor for Rivastigmine Tartrate. As detailed in patent CN101643439A, a novel manufacturing pathway has been established that fundamentally alters the economic and safety landscape of producing this vital chemical building block. This technical insight report analyzes the transition from legacy multi-step syntheses to a streamlined, two-step protocol that leverages Lewis acid-catalyzed amide reduction. For R&D directors and procurement strategists, understanding this shift is paramount, as it offers a tangible route to cost reduction in pharmaceutical intermediates manufacturing while simultaneously addressing stringent environmental and safety compliance standards required by modern regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of methylethylamine, the immediate precursor to our target molecule, relied on cumbersome and hazardous synthetic sequences. Traditional literature often describes a three-step process starting from benzaldehyde, involving enamination, methylation, and subsequent hydrolysis. A critical bottleneck in this legacy approach is the methylation step, which notoriously utilizes dimethyl sulfate, a substance classified as a potent carcinogen and highly toxic alkylating agent. Alternatively, routes originating from benzenesulfonyl chloride also suffer from excessive step counts and the generation of substantial sulfonamide waste streams. Furthermore, prior art such as US Patent 4131674 describes the subsequent conversion to the carbamoyl chloride using phosgene in the presence of stoichiometric amounts of triethylamine as an acid scavenger. This requirement introduces significant downstream processing burdens, as the resulting triethylamine hydrochloride salt must be separated and disposed of, inflating both material costs and waste treatment expenses.
The Novel Approach
In stark contrast, the methodology disclosed in CN101643439A presents a paradigm shift by utilizing readily available amides—specifically N-methylacetamide or N-ethylformamide—as the starting materials. This strategy collapses the synthesis of the amine intermediate into a single, efficient reduction step, bypassing the toxic methylation reagents entirely. The subsequent conversion to ethylmethylcarbamoyl chloride is achieved through a direct reaction with triphosgene or phosgene, notably performed without the addition of any acid-binding agent. This elimination of the base scavenger is a profound process intensification move, as it prevents the formation of solid salt byproducts during the reaction, thereby simplifying the workup procedure to a mere solvent removal and distillation. 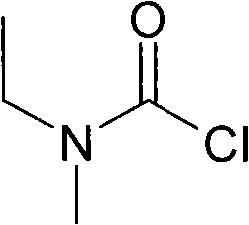
Mechanistic Insights into Lewis Acid-Catalyzed Amide Reduction
The core chemical innovation lies in the activation of the amide carbonyl group towards nucleophilic attack by hydride species. Under standard conditions, amides are notoriously resistant to reduction by mild hydride donors like sodium borohydride due to the resonance stabilization of the nitrogen lone pair with the carbonyl carbon. However, the introduction of a Lewis acid catalyst, such as zinc chloride (ZnCl2), ferric chloride (FeCl3), or aluminum trichloride (AlCl3), coordinates with the carbonyl oxygen. This coordination withdraws electron density from the carbonyl carbon, significantly increasing its electrophilicity and rendering it susceptible to reduction by borohydrides (KBH4 or NaBH4) even at moderate temperatures. The reaction is typically conducted in a mixed solvent system of toluene and tetrahydrofuran (THF), which balances the solubility of the inorganic salts with the organic substrates while allowing for reflux conditions that drive the reaction to completion.
Following the reduction, the resulting methylethylamine is isolated via a pH-swing extraction and distillation protocol. The crude reaction mixture is treated with hydrochloric acid to form the water-soluble amine hydrochloride salt, allowing organic impurities to be removed. Subsequent basification with sodium or potassium hydroxide liberates the free amine, which is then distilled. In the second stage, the purified amine reacts with triphosgene. Unlike previous methods requiring base, this reaction proceeds cleanly because the hydrogen chloride generated is managed through the specific thermal profile and distillation setup, or potentially recycled, rather than neutralized by expensive organic bases. 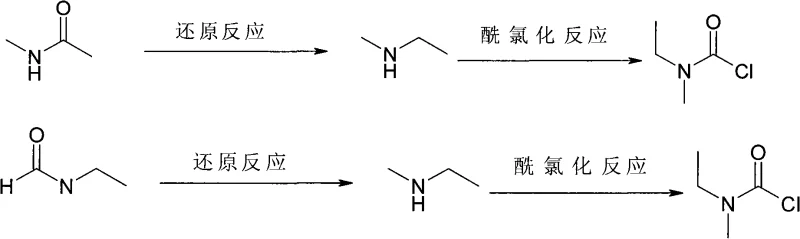
How to Synthesize Ethylmethylcarbamoyl Chloride Efficiently
The operational simplicity of this patented route makes it highly attractive for technology transfer and scale-up. The process begins with the careful addition of the Lewis acid to the solvent mixture under cooling, followed by the controlled addition of the amide substrate and the reducing agent. Temperature management is critical during the exothermic reduction phase to prevent runaway reactions, after which the mixture is heated to reflux to ensure full conversion. Once the amine is isolated, the phosgenation step requires precise temperature control during the addition of the amine to the triphosgene solution to manage gas evolution, followed by a reflux period to complete the acylation.
- Reduction of N-methylacetamide or N-ethylformamide using KBH4/NaBH4 and a Lewis acid (e.g., ZnCl2) in a toluene/THF solvent system to generate methylethylamine.
- Isolation of the amine intermediate via pH adjustment and distillation, collecting the fraction boiling at 50-80°C.
- Reaction of the isolated methylethylamine with triphosgene in dichloromethane under reflux conditions to yield the final carbamoyl chloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into enhanced operational efficiency and risk mitigation. The most significant advantage is the complete avoidance of dimethyl sulfate, which not only removes a major safety hazard for plant operators but also eliminates the need for specialized containment equipment and costly hazardous waste disposal protocols associated with sulfur-based toxins. Furthermore, the starting materials, N-methylacetamide and N-ethylformamide, are commodity chemicals with robust global supply chains, ensuring supply chain reliability and shielding production schedules from the volatility often seen with specialty reagents.
- Cost Reduction in Manufacturing: The elimination of triethylamine or other acid scavengers in the final step represents a direct material cost saving. In large-scale production, the cost of stoichiometric bases and the subsequent waste treatment of the resulting salts can be substantial. By removing this requirement, the process reduces the overall mass intensity of the reaction, leading to lower raw material consumption per kilogram of product. Additionally, the shortened synthetic sequence reduces utility costs associated with heating, cooling, and agitation across fewer unit operations, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: Relying on bulk commodity solvents like toluene, THF, and dichloromethane, alongside widely available reducing agents like potassium borohydride, minimizes the risk of supply disruptions. Unlike complex chiral catalysts or custom-synthesized intermediates that may have single-source suppliers, the inputs for this process are standardized industrial chemicals. This diversification of the supply base ensures that production can be maintained continuously, supporting the commercial scale-up of complex pharmaceutical intermediates without the bottleneck of sourcing obscure reagents.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard unit operations such as reflux, distillation, and liquid-liquid extraction that are easily modeled from laboratory to pilot to commercial plant scales. The reduction in hazardous waste generation, particularly the absence of sulfate waste and amine salt sludge, aligns with increasingly strict environmental regulations. This 'green chemistry' aspect not only reduces disposal fees but also future-proofs the manufacturing site against tightening emissions and effluent standards, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for feasibility assessments.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: This method eliminates the use of highly toxic dimethyl sulfate required in traditional methylation routes and removes the need for acid-binding agents like triethylamine during the phosgenation step, significantly simplifying purification and waste treatment.
Q: Which reducing agents are compatible with this Lewis acid-catalyzed protocol?
A: The patent specifies a range of effective reducing agents including potassium borohydride (KBH4), sodium borohydride (NaBH4), lithium aluminum hydride (LiAlH4), and even catalytic hydrogenation systems, offering flexibility based on available infrastructure.
Q: How does this process impact the purity profile of the final API intermediate?
A: By avoiding complex multi-step sequences involving enamination and hydrolysis, the impurity profile is drastically simplified. The direct reduction of stable amide precursors minimizes side reactions, leading to higher crude purity before final distillation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethylmethylcarbamoyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this Lewis acid-catalyzed route are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of ethylmethylcarbamoyl chloride meets the exacting standards required for Alzheimer's therapeutic applications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits for your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a secure and efficient supply of this critical pharmaceutical intermediate.
